Chapter 19:
Eastern Africa: Kenya Uganda, United Republic of Tanzabia, Rwanda, Burundi
INTRODUCTION
This chapter describes the development of smallpox eradication programmes in 5 contiguous countries of eastern Africa—Burundi, Kenya, Rwanda, Uganda and the United Republic of Tanzania’ (Fig. 19.1). In 1970 their combined population amounted to 42 million and their total land area to 1 818 000 square kilometres. In comparison, adjacent Zaire to the west had half as large a population in 1970 but an area 25% greater.
The infrastructure of transport and communication services was better developed than in the countries of western and central Africa; moreover, government and private health centres, clinics, dispensaries and hospitals were more numerous. Vaccination, with liquid vaccine, had been practised more or less extensively throughout most parts of these countries for more than 50 years, most vaccinations being given when outbreaks of smallpox occurred. Some health facilities offered vaccination to any patients attending them, but the vaccine was generally stored at room temperature and its potency was therefore presumably low or nil.
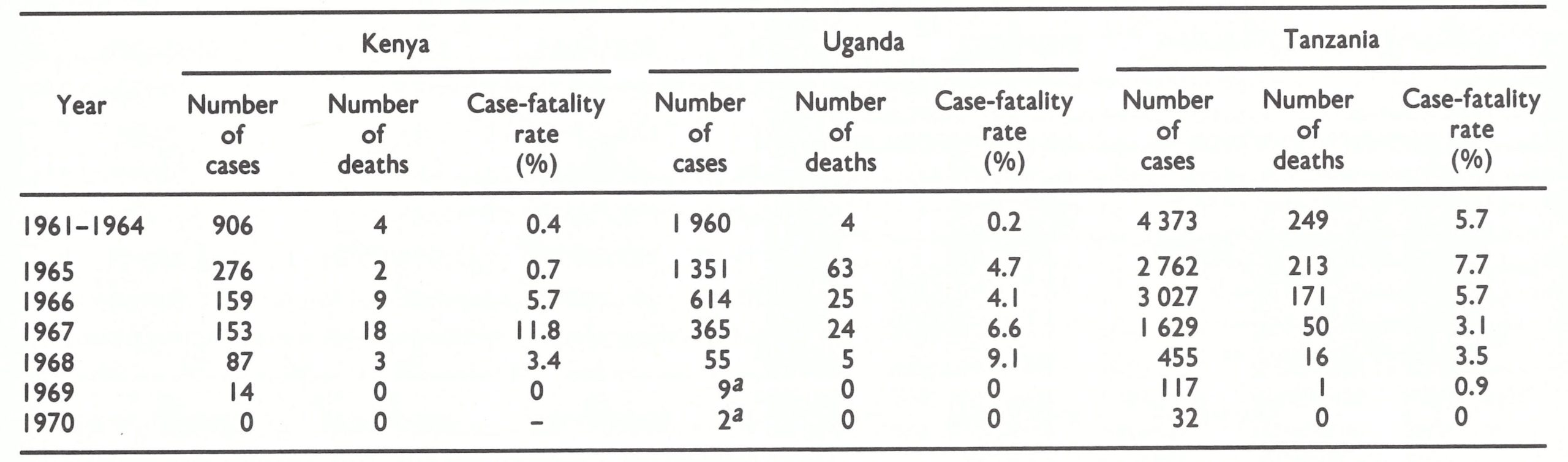
Among the many health problems in these countries, smallpox in the early 1960s had not been of high priority except in Tanzania. Epidemics of variola major had occurred in previous decades but, more recently, in all countries except Tanzania, variola minor with a low case-fatality rate prevailed. For example, during the period 1961-1965, 1182 cases with only 6 deaths were reported from Kenya, and between 1961 and 1964 Uganda recorded 1960 cases and 4 deaths. However, Uganda in 1965 and Kenya in 1966 began to experience an increasing number of deaths due to smallpox and consequently took a greater interest in controlling the disease. In Tanzania, a more severe form of smallpox had been prevalent, with recorded case-fatality rates of 5-7%. Even so, the problem was not large when viewed against the greater threats posed by malaria, tuberculosis and several other infectious diseases. Vaccination campaigns, however, were an integral part of public health activities because of the fear of recurrence of epidemics of the virulent variola major. With substantial commerce and travel between eastern Africa and Asia and the generally held belief that severe variola major prevailed elsewhere in Africa, there was reason to maintain adequate levels of vaccinial immunity.
After 1967, when the Intensified Programme began, all 5 countries agreed to undertake eradication programmes which would include systematic national vaccination campaigns employing, for the first time, freeze-dried vaccine of acceptable quality. WHO provided each country with advisers, all the requisite transport, equipment and supplies (including vaccine) and reimbursed the governments for the costs of petrol and vehicle maintenance. The countries made available a small cadre of national personnel, but little else. Each conducted effective national smallpox vaccination campaigns, which, in Kenya, Rwanda and Tanzania, were carried out in conjunction with the administration of BCG (antituberculosis) vaccine to children under 15 years of age.
Smallpox vanished quickly, transmission being interrupted by the end of 1970, well before the systematic vaccination campaigns reached most areas and, indeed, before effective programmes of surveillance and containment had been developed. After 1970, importations from the Sudan occurred along Uganda’s northern border, and importations from Ethiopia and Somalia along Kenya’s north-eastern borders. The outbreaks were all in sparsely populated areas; none resulted in large numbers of cases or persisted for long periods.
The mass vaccination campaigns differed somewhat in character from one country to the next, as each was adapted to the nature of the health services and the political structure of the country concerned. Information about the epidemiological characteristics of smallpox in eastern Africa is scarce, however, because little was done to improve the national reporting systems or systematically to investigate suspected cases until after 1970, when transmission had been interrupted.
The rapidity and ease with which smallpox transmission was interrupted in these countries, as well as many others in Africa, were surprising. At the time, the situation served to engender an unwarranted confidence that smallpox eradication could be readily achieved with only a modest incremental investment of resources, even in programmes in which performance was marginal. In Africa itself—notably in Ethiopia, Somalia and the Sudan—this belief later proved to be unfounded.
1 The United Republic of Tanzania was formed in 1964 by the union of Tanganyika, Zanzibar and Pemba . Although its official name is used elsewhere in this book, it is, for brevity, referred to as "Tanzania" in the text and tables of this chapter. The islands of Zanzibar and Pemba experienced no smallpox and conducted no special eradication programme.
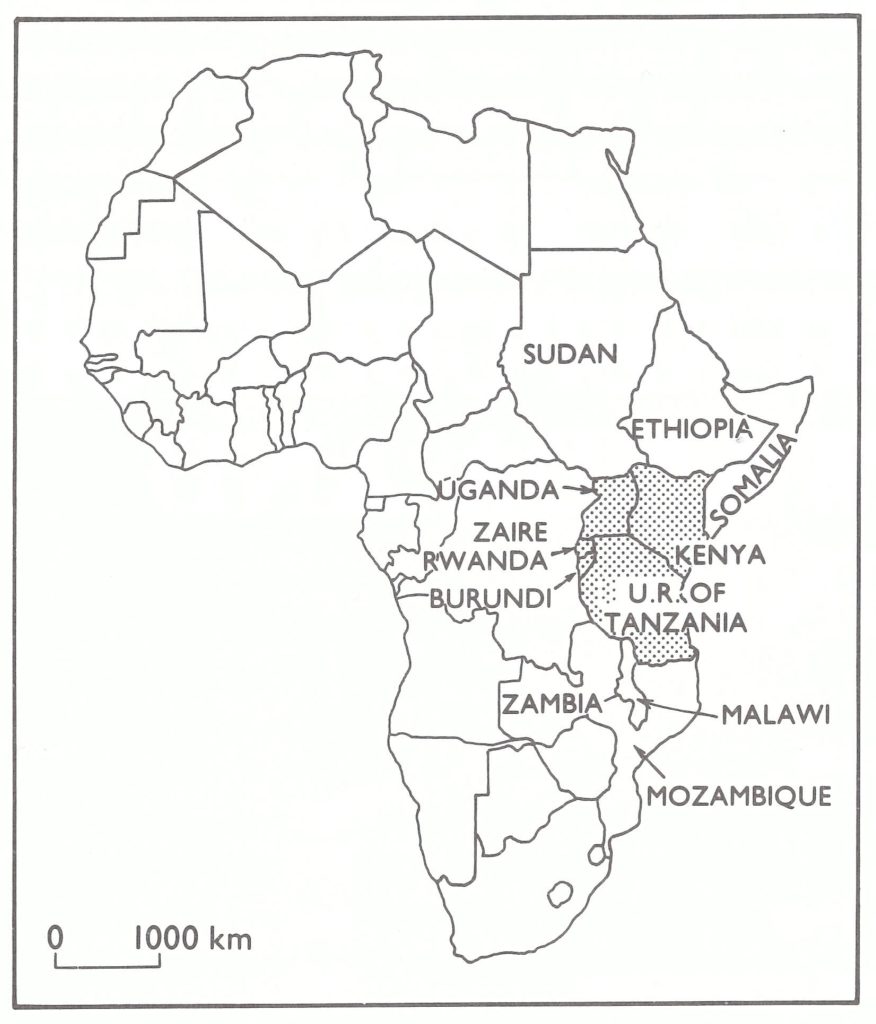
Fig. 19.1. Eastern Africa and adjacent countries.
SMALLPOX CONTROL ACTIVITIES BEFORE 1969
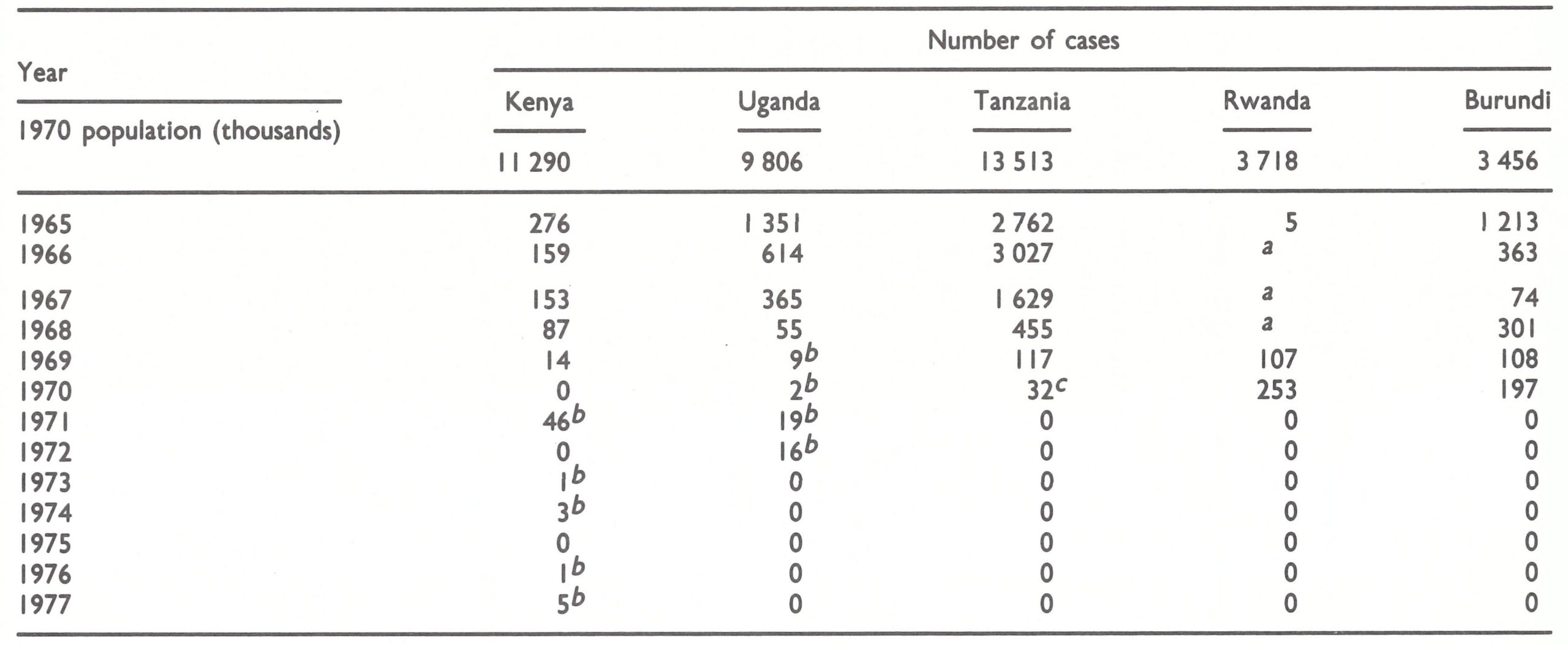
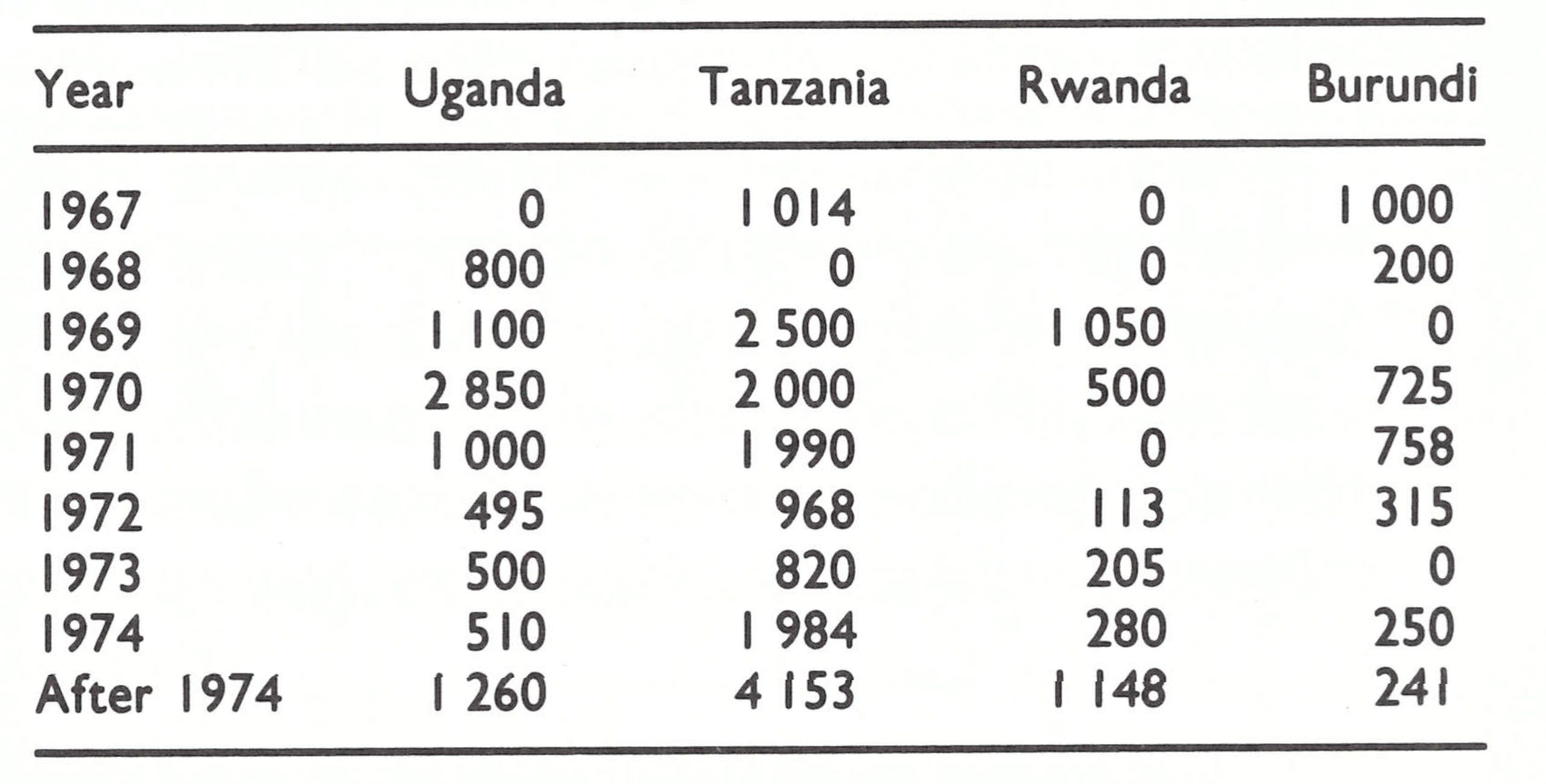
In 1967, when the Intensified Smallpox Eradication Programme began, the countries of eastern Africa reported a total of only 2221 cases (Table 19.1). In a population which then numbered almost 38 million, this figure was not large. Moreover, the number of recorded cases was diminishing rapidly, from 5607 in 1965 to 4163 cases in 1966. Of the 5 countries, only Rwanda reported no cases whatsoever during the period 1966-1968 but it was provisionally categorized as an endemic country, since it was difficult to believe that a country completely surrounded by heavily infected areas could be free of smallpox. Not until 1969 was it learned that it was government policy to report only cases that were confirmed by laboratory examination. Because no specimens were examined, no cases were reported.
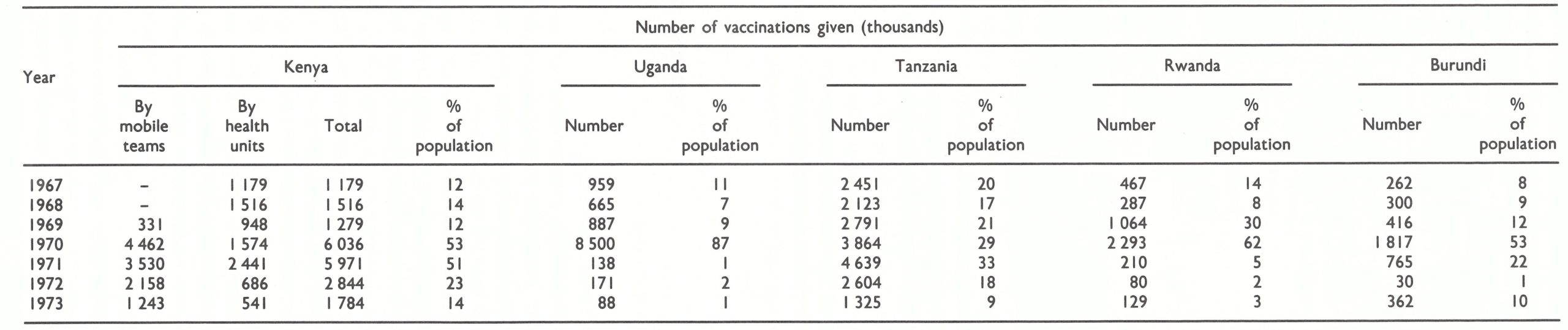
Vaccination was provided routinely in all countries, the number performed annually being equivalent to about 10-20% of the population (Table 19.2). Undoubtedly many of the vaccinations were unsuccessful because of the use of vaccine that was subpotent at the time of administration. Kenya, Tanzania and Uganda used liquid vaccine produced at the Medical Research Laboratory in Nairobi or at the Lister Institute, England. Although usually stored under refrigeration in the capital city, the vaccine was exposed to ambient temperatures during shipment and rarely refrigerated at any of the intermediate distribution points or in health centres. This was a problem, because liquid vaccine becomes inactive within 3 days or less at ambient temperatures when it is not refrigerated. No data are available regarding the proportion of successful vaccinations because vaccinees were not examined to determine whether their vaccinations had taken. Until 1965, Rwanda and Burundi also used liquid vaccine, produced at a laboratory in Rwanda or imported from Belgium. In 1965, the Rwanda laboratory began producing a reasonably satisfactory freeze-dried vaccine, but the vials in which it was distributed were so large that the amount of vaccine each contained was sufficient, after reconstitution, for 600 doses or more. This was not much of an improvement, because freeze-dried vaccine, after reconstitution, loses its potency within 24 hours unless it is refrigerated, and few health centres had refrigerators which functioned properly. Rarely did any health unit have cause to vaccinate more than a few persons a day but the staff continued to use the reconstituted vaccine until each vial was empty. In 1968, for example, a WHO smallpox eradication adviser observed at a hospital in Rwanda that a vial containing vaccine reconstituted 3 months earlier was still in use.

Plate 19.1. In Rwanda and throughout eastern Africa, mass vaccination had traditionally been used to control the spread of smallpox. Villagers willingly assembled at collecting points to be vaccinated.
In each of the countries, mass campaigns were conducted from time to time in specific areas when epidemics occurred. For such campaigns, vaccine was shipped to the field direct from the laboratories and was more likely to have been of adequate potency at the time of administration than vaccine which had been kept for weeks or even months at health centres. Only one country-wide mass vaccination campaign—in Uganda—had been carried out since the 1950s. In 1965, the number of reported smallpox cases and deaths in Uganda increased. The Ministry of Health, fearing epidemic variola major, launched a national campaign. Between August 1965 and February 1966, 7.5 million persons were vaccinated, a total equivalent to more than 90% of the country’s population at the time. The cost of vaccine alone, most of which was purchased from the United Kingdom, amounted to more than US$200 000. A survey conducted by Ladnyi in 1966 revealed vaccination scars in 88% of children in primary schools, but in only 61% of people attending clinics at 6 hospitals. The examination of 33 patients with smallpox revealed that 8 had been unsuccessfully vaccinated 1-3 months before. It was apparent that the campaign had been far less successful than the coverage might suggest.
The impression that a more severe form of smallpox had appeared in Uganda is borne out by the available records. Up to 1965, deaths from smallpox had been infrequent in Uganda, but in 1965 the case-fatality rate increased to nearly 5 % and remained high until transmission was interrupted (Table 19.3). During the following year, the casefatality rate in Kenya also increased, with both cases and deaths occurring primarily in areas adjacent to Uganda. Kenya, too, undertook a mass campaign—in Nyanza Province, which borders on Uganda. Between March and April 1966, 670 000 persons were vaccinated. In the same year, 500 000 people in Burundi and 6 million in Tanzania were vaccinated during mass campaigns.
From 1966 onwards, the case-fatality rates in Uganda and Kenya were characteristic of those observed with the African strain of variola major. This strain had been prevalent in neighbouring Tanzania and Zaire, from which it had presumably been introduced. Nothing can be said of the status of Rwanda or Burundi at this time, because no cases were officially recognized by Rwanda and deaths were not registered in Burundi.
AGREEMENTS TO UNDERTAKE ERADICATION PROGRAMMES
funds for eradication; they were, in fact, the first WHO regional smallpox eradication staff to be appointed. Ladnyi immediately embarked on a series of visits to each of the countries, beginning in 1966, with Burundi, Kenya, Malawi, Tanzania, Uganda and Zambia.
At the start, he could do little except offer advice, since the resources allocated to the smallpox eradication programme by WHO were so limited. Moreover, because most countries did not consider smallpox an important problem, few were interested in developing programmes, or, indeed, were able to do so without special assistance.
The above-mentioned decision of the Nineteenth World Health Assembly to allocate funds for smallpox eradication from the Organization’s 1967 regular budget made it possible to launch many national programmes. Surprisingly, however, except for those from Zaire and Zambia, no requests for assistance were received by WHO from any of the countries in central, eastern or southern Africa. Meanwhile, during 1966 and 1967, programmes throughout western Africa began to develop with assistance provided by the USA (see Chapter 17). Smallpox eradication staff from WHO Headquarters asked the regional office to contact the eastern and southern African countries to ascertain their interest in developing eradication programmes. However, the regional office pointed out that, as a matter of policy, it responded only to requests for assistance initiated by the countries themselves. In part, this policy derived from the views of the Director-General of WHO. In meetings with his regional directors in 1966, he had urged extreme caution about imposing another mass programme on Member states, after the experience with malaria eradication. The regional directors, in turn, had conveyed this to the WHO representatives in the countries themselves. Because the DirectorGeneral regarded smallpox eradication as improbable, he believed that such a programme would only divert national resources from more important activities (see Chapter 10). The WHO smallpox eradication staff in Geneva, on the other hand, maintained that the decision to undertake global smallpox eradication had received the unanimous approval of the World Health Assembly; funds had been appropriated for this purpose and thus it seemed only reasonable to contact the endemic countries to assess their intentions and needs. To expect the African countries themselves to respond spontaneously to new programme directions seemed unrealistic. All were newly independent, beset with problems and struggling to bring order into chaotic bureaucracies. These arguments were, however, of no avail.
Ladnyi, as a member of the regional office staff, had his hands tied; there was little he could do to overcome the apparent impasse in the initiation of eradication programmes in central, eastern and southern Africa, without directly flouting the policy of the regional office. The problem of how to approach the countries was resolved when the regional office agreed that a medical officer from Headquarters, Dr Stephen Falkland, accompanied by Ladnyi, could visit a number of African countries early in 1967 so that the Director-General could report to the World Health Assembly in May of that year on each country’s smallpox status and intended activities, if any, in accordance with the Assembly’s request of 1966. Dr Falkland was specifically directed that under no circumstances should he propose to any country that it should undertake a programme. In the event of a display of interest, however, he carried with him a draft letter requesting assistance, prepared for the signature of the minister of health, as well as a draft plan of operations which could be readily adapted to the specific conditions of each country. He made brief visits to Burundi, Kenya, Rwanda, Tanzania and Uganda, all of which expressed interest in initiating smallpox eradication programmes and which forthwith sent formal requests to WHO for assistance.
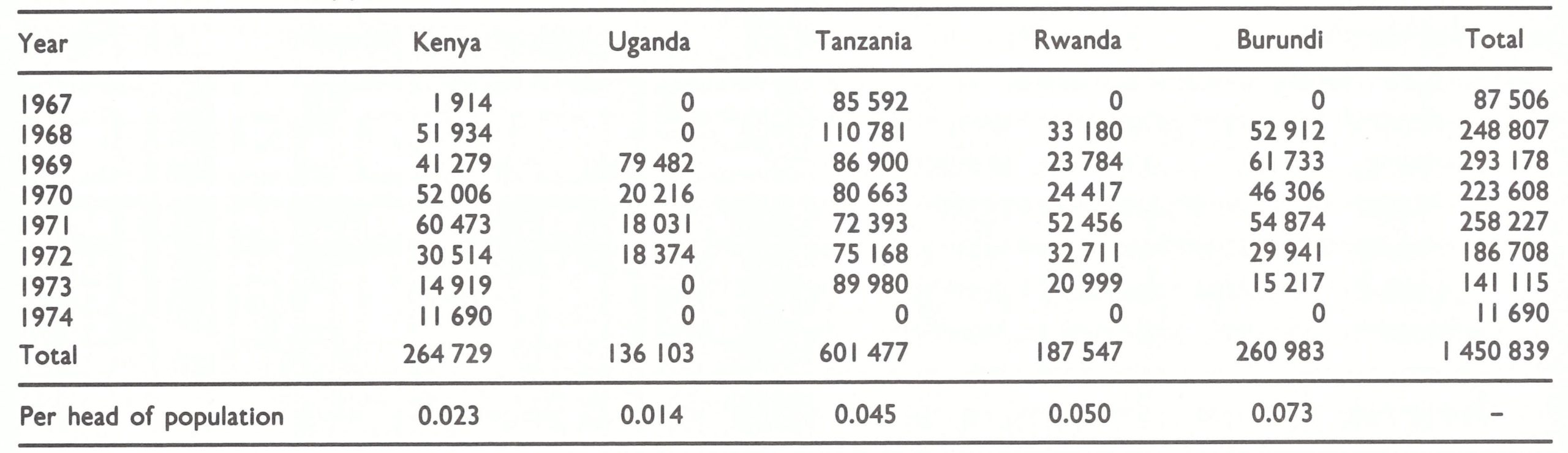
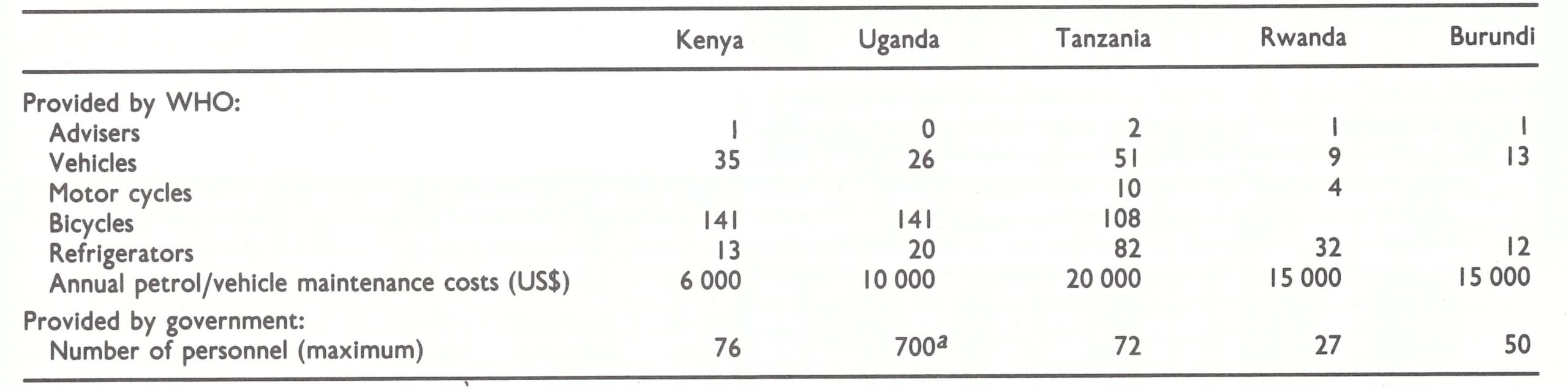
Of the 5 countries dealt with in the present chapter, only Tanzania viewed smallpox as an important problem. However, to undertake eradication programmes, it was unnecessary for the governments concerned to commit substantial national resources. For each programme WHO agreed to provide an adviser, vehicles (including in some instances, motor cycles and bicycles), refrigerators, camping equipment, vaccine and bifurcated needles, as well as a sum of money to cover the costs of petrol and vehicle maintenance. The country’s obligation was primarily to provide a small cadre of national staff. To offset the additional expenditures necessary for salaries of these workers, WHO made available vaccine and transport, which hitherto had been at the charge of the government. In all, the Organization provided US$1 450 839 in support of programmes in eastern Africa, a sum equivalent to US$0.01-0.07 per head of population (Table 19.4). The individual governments disbursed no more—and in most cases less—to eradicate smallpox than they had spent on controlling the disease.
PROVISION OF FREEZE-DRIED VACCINE
The acquisition of adequate supplies of freeze-dried vaccine and their distribution presented a problem to WHO throughout most of the global eradication programme (see Chapter 11). In the countries of eastern Africa, as well as many of those in central and southern Africa, there were unique difficulties which required special solutions.
Attempts were made in all endemic areas to promote the local production of vaccine, but it was not practicable for every country to do so because of the considerable cost and effort needed to establish and maintain a laboratory. For some 50 years, the Kenyan Medical Research Laboratory had been producing liquid vaccine, which it sold to Tanzania and Uganda. Thus, it seemed logical to supply this laboratory with equipment and consultant assistance so that it might serve as a regional resource for the production of freeze-dried vaccine. UNICEF provided US$20 000 for the necessary equipment and in January 1967 WHO made available the services of a consultant. By April 1967, the laboratory, directed by Dr Geoffrey Timms, had succeeded in producing 4 experimental batches of freeze-dried vaccine and by October 1968 was producing sufficient vaccine of a consistently high quality to meet all Kenya’s needs. As production increased, it became possible to supply neighbouring countries, but herein lay a quandary. Kenya needed to charge for the vaccine to recover production expenses. WHO’s stock of vaccine, however, was being provided through voluntary contributions and then distributed free of charge to all endemic countries to encourage its use. Unless this had been done, many governments, lacking reserves of convertible currency, would have continued to use locally produced liquid vaccine. The purchase of vaccine from Kenya by WHO was not feasible, because several producers in Europe were anxious to sell vaccine and such a move might have endangered the donation programme (see Chapter 11); thus, WHO had adopted the policy of purchasing no vaccine whatsoever. The problem was ultimately solved by supplying Kenya with almost all the materials needed for vaccine production, thus enabling it to produce additional vaccine which could be donated to WHO for use in other African countries. In all, WHO provided some 31 million doses of vaccine for use in the countries of eastern Africa (Table 19.5), of which 15 million doses were produced in Kenya.
The distribution of vaccine in a timely manner to the numerous African countries was a factor that had to be taken into account. Vaccine was in short supply throughout most of the eradication programme, and it was therefore not possible to stockpile large reserves in the endemic countries. With the uncertain schedule of vaccination campaigns, the shipment of vaccine had to be planned in advance. Vaccine donated to WHO during the early years came primarily from Europe or North America and was maintained in a coldstorage warehouse in Geneva. With the extensive air transport network centred in Geneva, it was not difficult to deliver vaccine anywhere in the world at 48 hours’ notice. Vaccine donated by Kenya, however, was shipped direct to neighbouring countries to minimize transport costs. Distribution systems were thus in place for national programmes to be sustained without large stockpiles.
A problem which had to be tackled was that of communication through the official channels of WHO. For an African country to obtain vaccine, the standard procedure called for a request to be directed to the WHO Regional Office for Africa in Brazzaville, which would forward it to Geneva. Communication between African countries, however, was often much slower than communication between Africa and Europe, and inevitably there were delays in processing the request within the regional office. A telegraphic request through official channels sometimes took 4-8 weeks before being received in Geneva. Meanwhile, field programmes came to a halt. The difficulty was resolved simply by sending 2 telegraphic messages—an official one to the regional office in Brazzaville and an unofficial one to Geneva, on which action was promptly taken.
Freeze-dried vaccine which had been contributed to WHO was made available to several of the countries for use in their health services even before their formal mass vaccination campaigns began (Table 19.5). This vaccine was first used by Burundi and Tanzania at the end of 1967 and by Uganda early in 1968. Kenya began using locally produced freeze-dried vaccine in October 1968. Tanzania reverted briefly to the distribution of liquid vaccine during November 1968, when it received a bilateral donation of 300000 doses, but fortunately no further contributions of this type were received. Rwanda continued to use its own locally produced freeze-dried vaccine until early 1969, when the government was persuaded to stop production and to destroy all existing stocks.
THE VACCINATION CAMPAIGNS
Except for a pilot programme in Tanzania in 1968, during which 350 000 persons were vaccinated, none of the vaccination campaigns began until 1969. Meanwhile, the number of reported cases of smallpox in Kenya, Tanzania and Uganda fell sharply (see Table 19.1), from 3800 in 1966 to 2147 in 1967 and to only 597 in 1968. Rwanda, during this period, did not officially recognize cases of smallpox; and little can be said about trends in Burundi, whose reporting system was less adequate than the others. During these years, little had been done to change the reporting systems and there was no organized surveillance-containment activity. However, the trend in incidence was steadily downward and systems which had detected many cases in prior years were now reporting very few. The mass campaigns in Uganda, Burundi, parts of Tanzania, and in Nyanza Province, Kenya, in 1965-1966 were undoubtedly responsible in part for the decrease in the number of reported cases. Perhaps of greater importance was the provision of freeze-dried vaccine for use by the health services in the control of outbreaks and in the limited routine vaccination programmes in which some health centres were engaged.
However, as we shall see, the vaccination campaigns in Kenya and Uganda began after transmission had apparently been interrupted; in Tanzania, only 88 additional cases occurred after the beginning of the campaign, of which 23 are known to have resulted from an importation from Zaire (Table 19.6). The endemic spread of smallpox stopped only 13 months after the programme had begun in Rwanda and after 17 months in the case of Burundi.
The most heavily infected regions in 1967-1968 lay on the shores and in the vicinity of Lake Victoria (Fig. 19.2) and in the southeastern area of Tanzania near the border with Mozambique. The region around Lake Victoria was also one of the most densely populated. As programmes began in 1969, Kenya and Tanzania concentrated their efforts specifically in these areas (Fig. 19.3).
Table 19.6: Eastern Africa: number of reported cases of smallpox, by month, 1969-1970
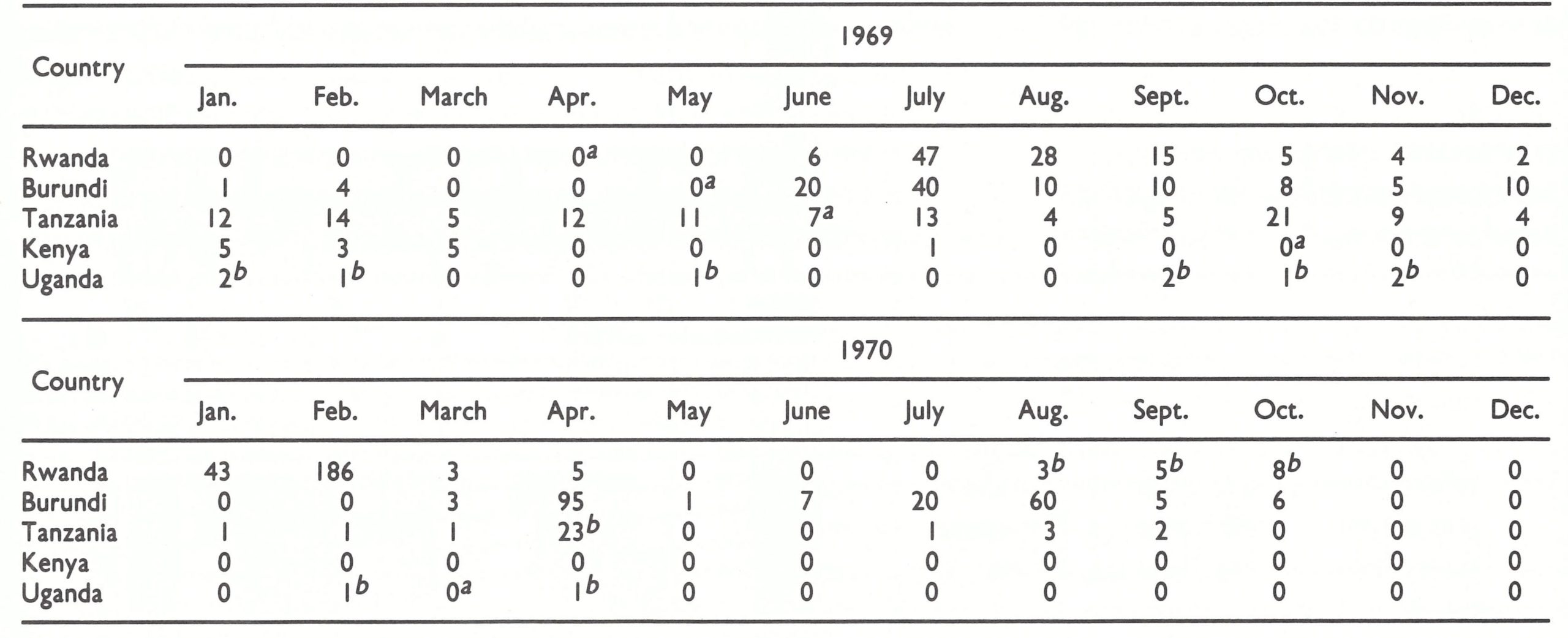 Note: cases are shown by month of detection; many of the patients concerned experienced the onset of illness one to several months earlier.
Note: cases are shown by month of detection; many of the patients concerned experienced the onset of illness one to several months earlier.
a Denotes beginning of mass vaccination programme.
b Cases resulting from Importations.
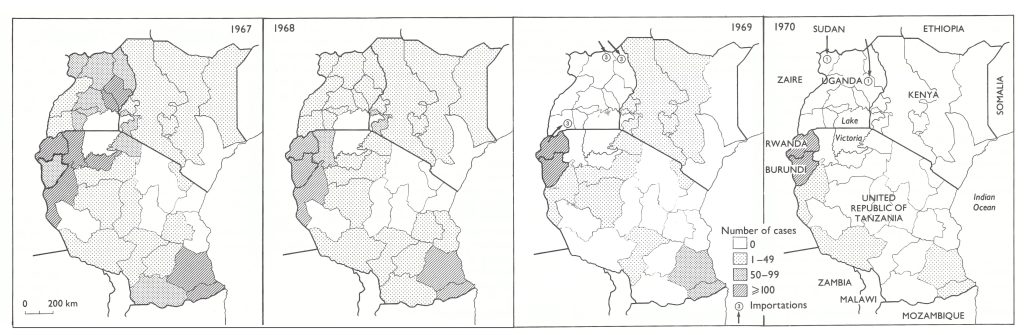
Fig. 19.2. .Eastern Africa: number of reported cases of smallpox by administrative area, 1967- 1970. The number of cases in Rwanda in 1967 and 1968 is estimated.
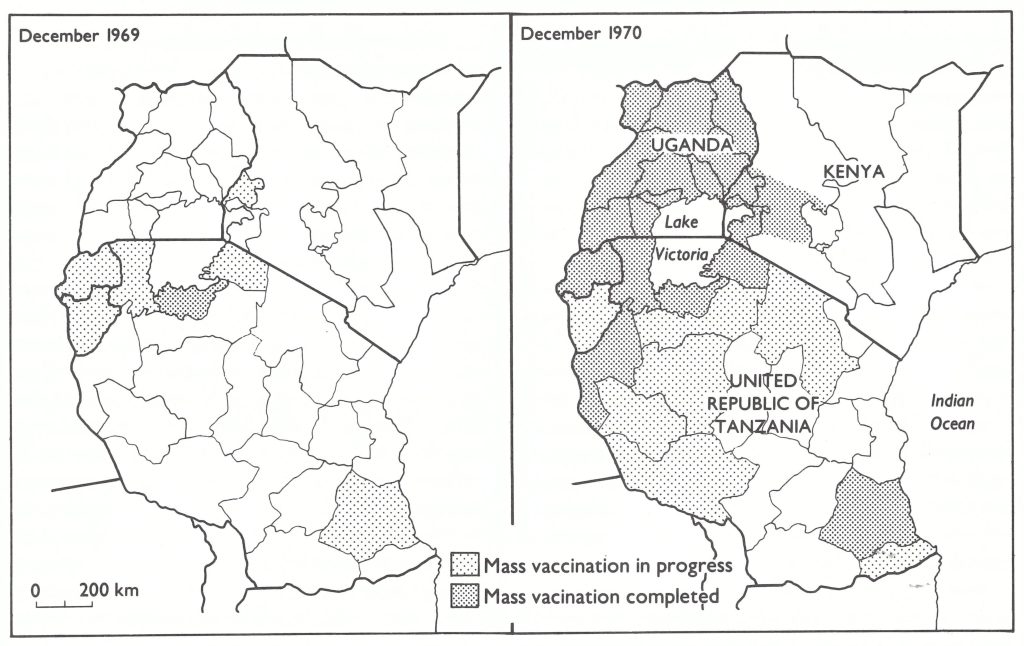
Fig. 19.3. Eastern Africa: status of vaccination campaigns, by administrative area, December 1969 and December 1970.
Kenya
A WHO technical officer from Belize, Mr Henry Smith, was recruited to assist in the development of the Kenya programme. He arrived in September 1968 and continued with the programme until February 1974. From October 1968 to the end of May 1969, Mr Smith visited all provincial and district headquarters, beginning with those in the west, whence cases were being reported, to demonstrate the use of the new bifurcated needle and the freeze-dried vaccine. At the time of his visit, stocks of liquid vaccine were replaced with freeze-dried vaccine. Staff in the districts were then given responsibility for training those in peripheral units. In Kenya at that time there were 163 health centres and 350 dispensary clinics; their staff reported having vaccinated 1.5 million persons in 1968 and 948 000 in 1969—about 25% of the population in all.
The beginning of the mass vaccination campaign had to await the delivery of vehicles, which usually arrived 12-18 months after they were ordered. The vehicles were delivered in the spring of 1969 and the programme began in October in Western Province, one of two small but densely populated south-western provinces inhabited by 40% (4.5 million) of Kenya’s population. Kenya’s last known case of endemic smallpox occurred 4 months before the programme began.
Initially, the staff consisted of 2 supervisors assigned from the health services and 18 Kenyan National Youth Service Volunteers, who had completed elementary school and had undergone a 1-month training course. The team progressed from district to district. Three weeks before it was to begin work, Mr Smith, or sometimes the national programme director, met the district medical officer and health inspector to plan the campaign. Within the district, vaccinations were given to the inhabitants of one sublocation after another—a sublocation being a unit ranging in population size from a few hundred to 4000 and administered by a subchief. An assistant health inspector who worked in the area contacted each subchief to explain the purpose of the programme and to select a place of assembly for each sector at which approximately 300 persons would be expected to forgather. In the interests of economy of operation, the entire team worked in only one sublocation or a few adjacent sublocations each day. Assembly points included hospitals, health centres, dispensaries, schools, markets, chiefs’ houses, shops, bus stops and wellheads. A few days before vaccinations were to be carried out, the subchief informed people of the schedule and the location of assembly points. Teachers were asked to pass on the details to parents and pupils. On the appointed day, 2 vaccinators (or more, if attendance was expected to be sizeable) were dispatched to the selected locations by Land Rover. One performed vaccinations while the other kept a simple tally by age group of the vaccinees. The vaccination centres were kept open throughout the entire day in order to give people sufficient time to attend, some being obliged to walk as far as 5 kilometres.
Because of the small number of staff, the vaccination campaign progressed so slowly that, by the spring of 1970, it was estimated that 10 years would be required to complete it. Accordingly, it was agreed that more National Youth Service Volunteers should be recruited and more supervisors assigned. By the end of 1970, the staff complement had grown to 50 vaccinators, 18 drivers and 8 health inspectors. In addition, it was agreed that when a team was in an area, local health staff who could be spared would work with it. The resources provided to the programme are shown in Table 19.7. With the increased complement of personnel, it was eventually possible to work in 10 districts simultaneously.

Plate 19.2. Henry C. Smith (b. 1935) served as a WHO technical officer for smallpox eradication in Kenya, 1968-1974, before joining the programme in India.
In May 1970, it was decided to administer BCG vaccine concurrently with smallpox vaccine to children under 15 years of age, and from then on the vaccinators worked as 2-man units. One vaccinator administered smallpox vaccine in one arm, using the bifurcated needle, and one administered BCG vaccine in the other arm, using a needle and syringe, and tabulated the numbers vaccinated. With this staff, between 400 000 and 500000 smallpox vaccinations were performed monthly. By the end of 1970, the vaccination schedule had been completed in Nyanza and Western Provinces as well as in portions of the neighbouring Rift Valley Province (see Fig. 19.3). In other parts of Kenya the population was vaccinated during 1971 and, finally, the coverage was extended to the numerous inhabitants of Nairobi and its surroundings in 1972. In all, 10.5 million vaccinations were performed by the mobile teams and 5.6 million by the established health units during this period.
The only departure from the routine occurred in March and April 1971, when 2 outbreaks, one of 45 cases and one of a single case, occurred in the north-eastern part of Kenya as a result of importations from Ethiopia (see Fig. 19.4). The mass vaccination campaign in this area had been scheduled to begin in April and last throughout June 1971, but its date of commencement was advanced to March. Subsequently, 6 vaccination/casesearch rounds were conducted in this and 5 other northern districts that bordered on Ethiopia and the Sudan and were considered to be at high risk.
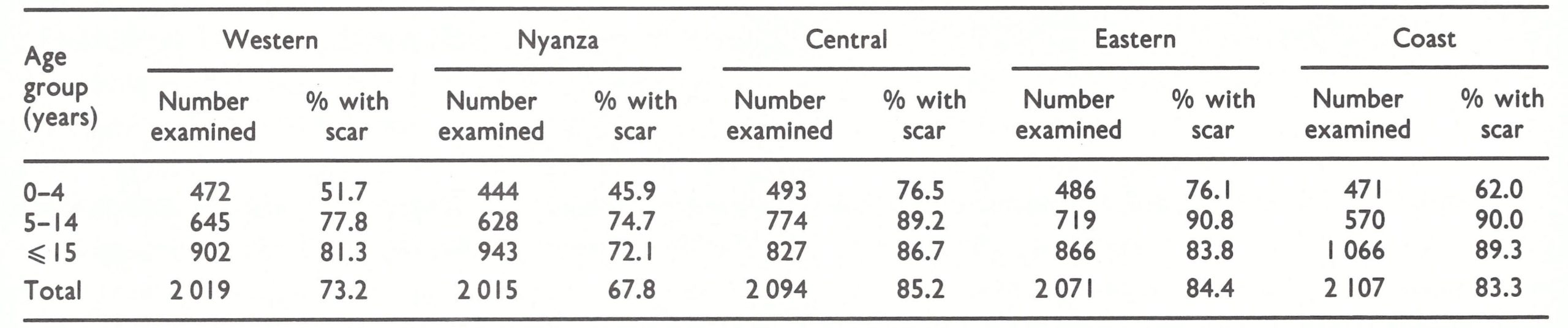
Except for Rwanda, none of the 5 countries followed the programme for assessment recommended in the WHO Handbook for Smallpox Eradication Programmes in Endemic Areas (SE/67.5 Rev.1). The handbook called for an assessment team to visit a 10% random sample of villages 1-2 weeks after vaccination to determine coverage and the proportion of people successfully vaccinated. As was the case in a number of countries, health officials resisted the diversion of personnel and vehicles from the vaccination campaign itself. To determine coverage in Kenya, the number vaccinated was simply compared with the estimated population in the area—an unsatisfactory procedure in a country where census data were so imprecise. Approximately 100 children were examined one week after vaccination to determine whether a sufficient proportion had been successfully vaccinated. Definitive information about the degree of success of the vaccination campaign did not become available until the conclusion of the programme, when, in 1972, 2 WHO staff from the WHO Epidemiological Surveillance Centre in Nairobi undertook a carefully designed cluster sample survey. Approximately 2000 persons were examined in each of 5 different provinces. The results are shown in Table 19.8.
The vaccination coverage of individuals aged 5 years and over was satisfactory in the Central, Eastern and Coast Provinces but not in Western and Nyanza Provinces, in which the programme had first been launched and had been completed in 1970. In these latter two provinces, the vaccination coverage of children under 5 years of age was substantially below expected levels; this was partly accounted for by the fact that health centres were vaccinating only about 25-30% of those born subsequent to the 1970 campaign.
At the conclusion of the campaign, in July 1972, 22 sectors were identified, to each of which a team with a vehicle was assigned. The teams were to proceed from one sublocation to the next, vaccinating those eligible for vaccination and inquiring at schools, markets and health units about possible cases that might have occurred during the preceding 3 months. Each team was expected to complete a tour of its sector once a year; in high-risk areas, the schedule was intensified to once every 3-6 months. The teams were called surveillance teams, but in fact their principal responsibility was to provide maintenance vaccination. The system remained in operation for little more than a year
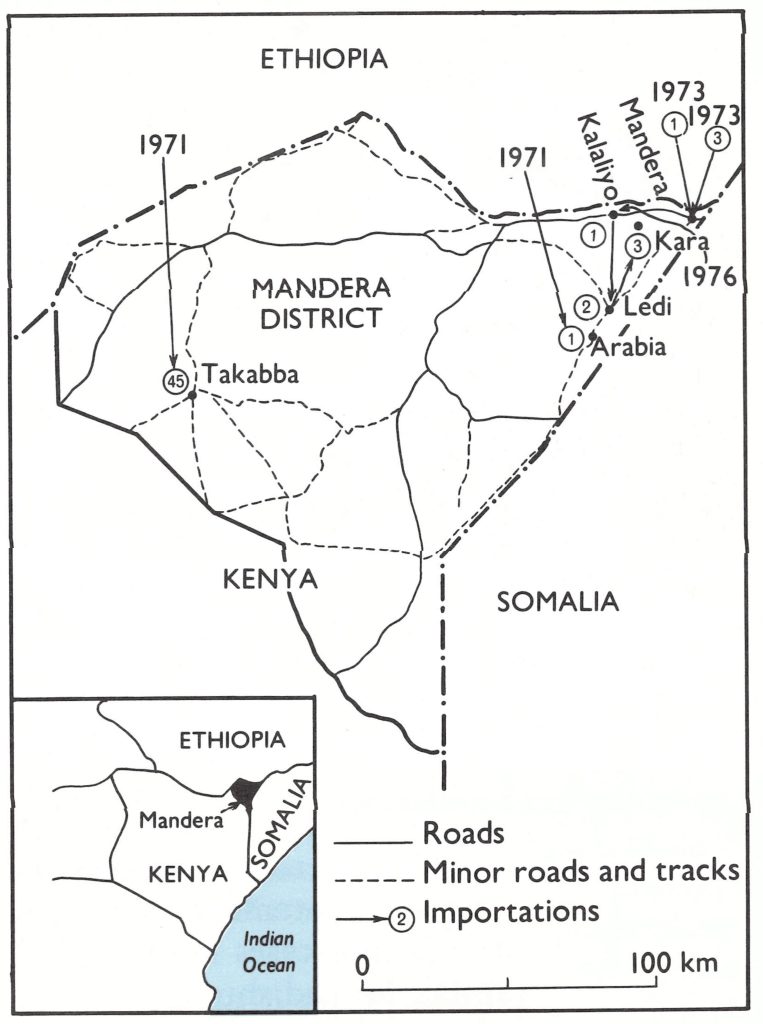
Fig. 19.4. Kenya: sources of importations of smallpox, 1971-1976.
Uganda
Like Kenya, Uganda had a reasonably extensive network of health centres and clinics and a complex of roads reputed to be the best in eastern Africa. A mass vaccination campaign using liquid vaccine was completed in 1966, following which the number of cases of smallpox diminished to only 365 in 1967 and 55 in 1968. Between 600 000 and 900 000 persons were being vaccinated annually in the various health establishments, and in 1969 freeze-dried vaccine supplied by WHO was introduced for use throughout the country. Because liquid vaccine had been employed in the previous vaccination campaign and because it was known that many of the vaccinations had not been successful, Uganda agreed in mid-1968 to undertake a special 3-year national vaccination campaign. WHO provided the necessary vehicles and equipment. However, the government later decided to abandon this schedule in order to conduct, in 1970, a national mass vaccination campaign to be completed in just 6 months. To help to prepare the work, Dr Georgij Nikolaevskij, of the WHO Headquarters Smallpox Eradication unit, was detailed to Uganda for a 5-month period. He assisted Dr Yurij Rikushin, a WHO medical officer originally assigned to Uganda for BCG vaccination. District health inspectors in each district organized the programme, which required the population to assemble for vaccination at collecting points. Vaccination staff were divided into groups of 3 or 4 persons who moved from subcounty to subcounty. In all, some 700 public health staff were engaged in the campaign. Between March and August 1970, 8.5 million vaccinations were performed in a population then estimated to be 9.8 million. There was no special assessment of the campaign to measure the prevalence of vaccination scars after it had been completed, but most believed that it had been generally well executed. Like the programme in Kenya, however, it was begun many months after smallpox transmission had been interrupted.
Following the campaign, vaccination continued to be performed by the various health centres in the country but only 100 000-200 000 persons were vaccinated annually, a figure equivalent to perhaps half the number of children born each year. In addition, sporadic campaigns which involved the vaccination of about 100 000 persons were conducted in areas bordering on the Sudan following the importation of cases from that country.
Such knowledge as was available regarding the level of vaccinial immunity at the time was provided by 2 surveys carried out in different parts of northern Uganda (Table 19.9).
Both surveys were conducted in areas convenient of access by road, immediately after special area-wide mass campaigns had taken place. One must assume that vaccinial immunity in more remote areas of these districts and in other parts of Uganda was substantially below the levels shown in Table 19.9.

Plate 19.3. Yurij P. Rikushin (b. 1923), a WHO medical officer working in tuberculosis control in Uganda, 1965-1971, also acted as the smallpox eradication adviser there and was instrumental in organizing Uganda’s 6-month intensive mass vaccination campaign.
Tanzania
Of the 5 countries, Tanzania was the only one which had expressed genuine enthusiasm about undertaking a programme. A thousand or more cases of smallpox had been reported almost every year since the 1950s, and in many outbreaks case-fatality rates of 5-10% had been recorded. Vaccination with liquid vaccine had been provided in health centres throughout the country, but no special programme had been conducted until 1965-1966, when the number of reported cases increased to 2762 in 1965 and to 3027 in 1966. During these 2 years, a mass campaign was conducted in which 6 million vaccinations were given and, subsequently, some 2-2.5 million vaccinations, with the lowpotency liquid vaccine, were performed annually in health units, which then consisted of 105 hospitals, 54 rural health centres and 1442 dispensaries. However, 1629 cases were reported in 1967—and reporting in Tanzania was considered to be less complete than in Kenya or Uganda.
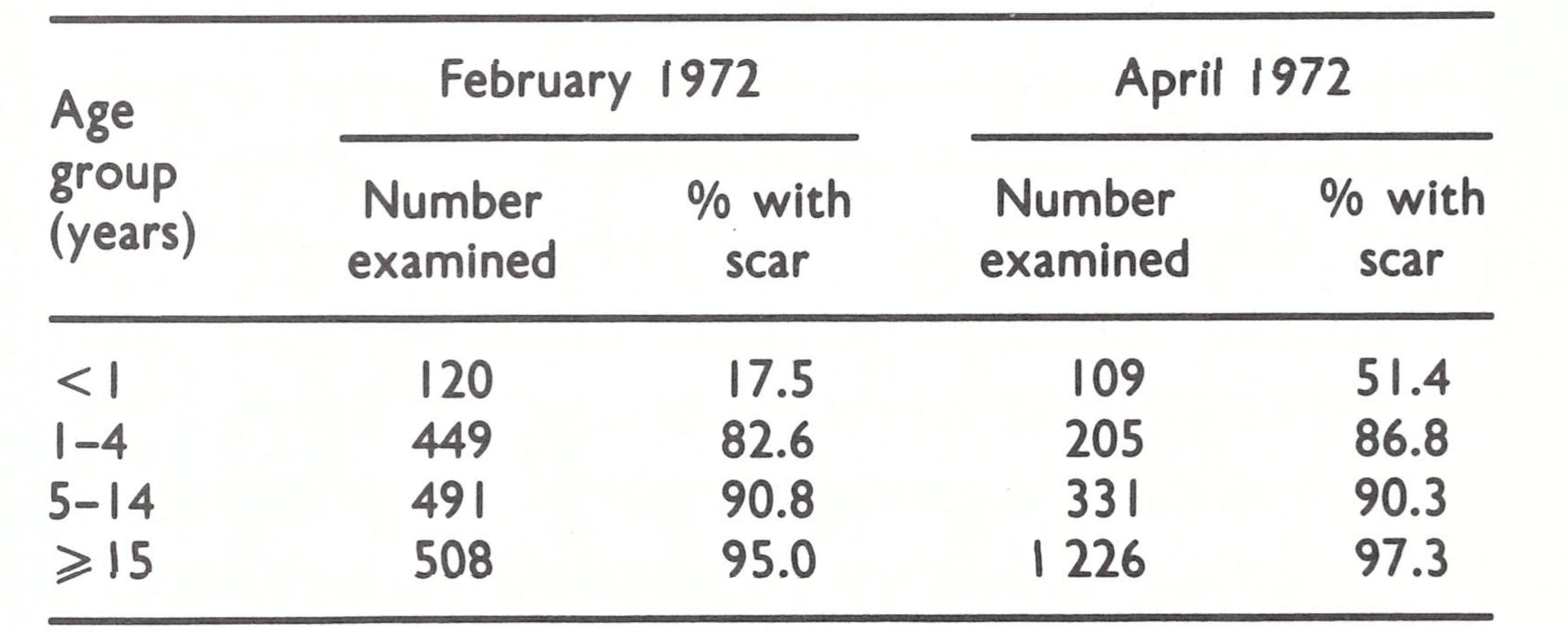
In August 1967, WHO and the government agreed on a plan of operations which provided for transport, equipment and supplies of freeze-dried vaccine, as well as the Table 19.10. Tanzania, Geita District: results of assignment of a WHO medical officer, Dr vaccination scar survey, by age group, R onne , and a technical office , Mr. 1969 G. Michalatos. The WHO staff arrived in the late spring of 1968, and in July 1968 a pilot project was begun in Geita District (population, 385 000) in northern Tanzania. This district had reported the highest incidence of smallpox of any in the country in 1966-1967 and was considered to be one of the most difficult in which to work because of poor roads and extensive swamps. During a 5-month period, a team of 24 vaccinators, a supervisor and 4 drivers performed vaccinations equal in number to more than 90% of the population.
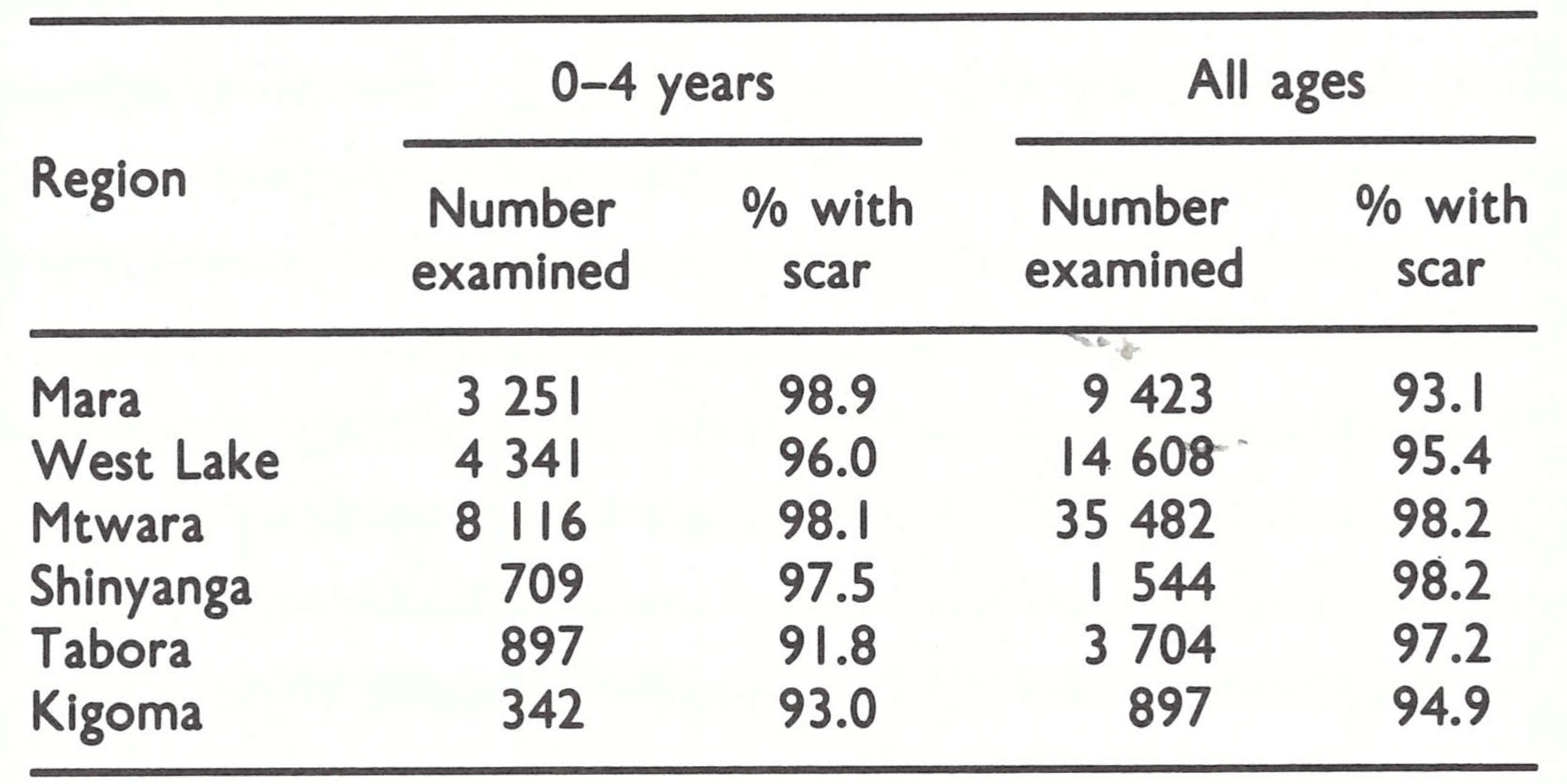
Lack of government funds then resulted in the suspension of the campaign for 7 months, until July 1969, when it was resumed in the endemic north-western districts (see Fig. 19.2) and at the end of the year in 2 endemic south-eastern districts. Three mobile teams, each consisting of 23 persons, conducted the campaign under the direction of each area’s district health inspector. The teams were subdivided into 4 units, each comprising a supervisor and 4 or 5 vaccinators and equipped with a motor vehicle, 2 bicycles and a kerosene-operated refrigerator. Political leaders, called “ten-cell chairmen,” played an important role. Each was contacted one or two weeks before the team was due to arrive and was asked to prepare a list of all residents in the area for which he was responsible . When the team arrived, the residents were assembled and each was summoned by name for vaccination. The system might appear cumbersome and time-consuming, but in fact the vaccinators consistently averaged more than 500 vaccinations a day. They continued to achieve this norm even when, in 1970, they undertook the additional task of administering BCG vaccine to children under 15 years of age. The system was an unusual one but it worked well in Tanzania, in which the political structure was highly organized. An independent cluster sample survey conducted in Geita District by a team from the WHO Epidemiological Surveillance Centre in Nairobi revealed that 94% of the persons examined had vaccination scars at the conclusion of the programme (Table 19.10).
Later surveys, conducted during 1970 at the conclusion of campaigns in 6 regions, revealed comparable results (Table 19.11).
Although by the end of 1969 the vaccination campaign had been completed in only 1 district and had just begun in 3 others, the number of reported cases of smallpox that year decreased to 117. Reporting was still inadequate, since little had been done to improve the national reporting system; however, this was the lowest total of cases ever recorded. Even if the reporting system had not improved, it certainly did not appear to have deteriorated. Finally, in 1970, the investigation of some cases began. Of 3 outbreaks examined, 1 (in April) had resulted from an importation from neighbouring Zaire. Cases were reported from only 4 regions (Fig. 19.2), and in September the last case was found.
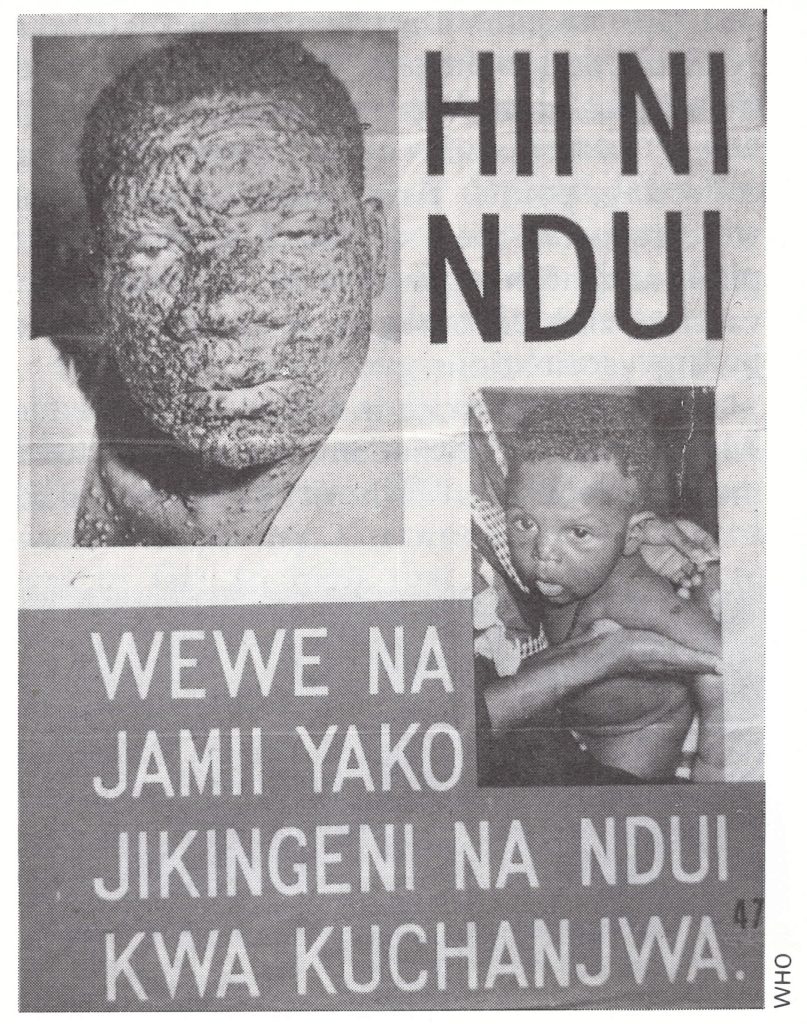
Plate 19.4. Tanzanian posters in Swahili were widely distributed in 1969, reading: “This is smallpox. You and your family protect yourselves by getting vaccinated”.
The programme continued, region by region and district by district, at length concluding with the vaccination of the population of the then capital, Dar es Salaam, in August 1973. While in most countries the residents of the capital city were the first to be vaccinated, they were left until last in Tanzania, since Dar es Salaam, situated on the coast, was far away from the known endemic areas. In all, 19.8 million persons were vaccinated against smallpox between 1967 and 1973 and 3.7 million under 15 years of age received BCG vaccine. After September 1970, vaccinators during their visits reported 23 suspected cases but none proved to be smallpox.
On completion of the programme, mobile teams continued to vaccinate and to search for cases in remote and border areas ; the health services vaccinated about a million persons each year thereafter but, as in Kenya and Uganda, interest in the programme diminished sharply.
Rwanda and Burundi
The programmes in these densely populated countries, each with some 3.5 million inhabitants, were a study in contrasts . In both, the responsibility for operating the programme was largely delegated to their respective WHO smallpox advisers. One of these, Dr Celal Algan, was an energetic worker, well acquainted with conditions in the field, who, with the help of Rwandese staff, conducted one of the most effective programmes in Africa. The other, who was assigned to Burundi, rarely travelled to the field and had little apparent capacity for organization; the programme in Burundi reflected these shortcomings.
In both countries, the population lived in small hamlets and hillside villages scattered over a tropical and subtropical upland plateau. Roads were few and often impassable during the February-May rainy season. Hospitals, health centres and dispensaries, some operated by the government and some by missionaries or other private voluntary organizations, were comparatively numerous. In 1971 there were 157 hospitals, health centres and dispensaries in Rwanda and 104 hospitals and dispensaries in Burundi. Vaccination was offered at all these health units; about 10-15% of the population were vaccinated each year. The vaccine used was a liquid product which had been manufactured by a laboratory in Butare (Rwanda) since 1953. Beginning in 1965, this laboratory also produced some freeze-dried vaccine which, as has been mentioned earlier, was prepared in 600-dose vials. These vials contained such a large quantity of vaccine that it was often kept, o unrefrigerated, for many days or even weeks, with a consequent loss of potency after the first day.
Rwanda
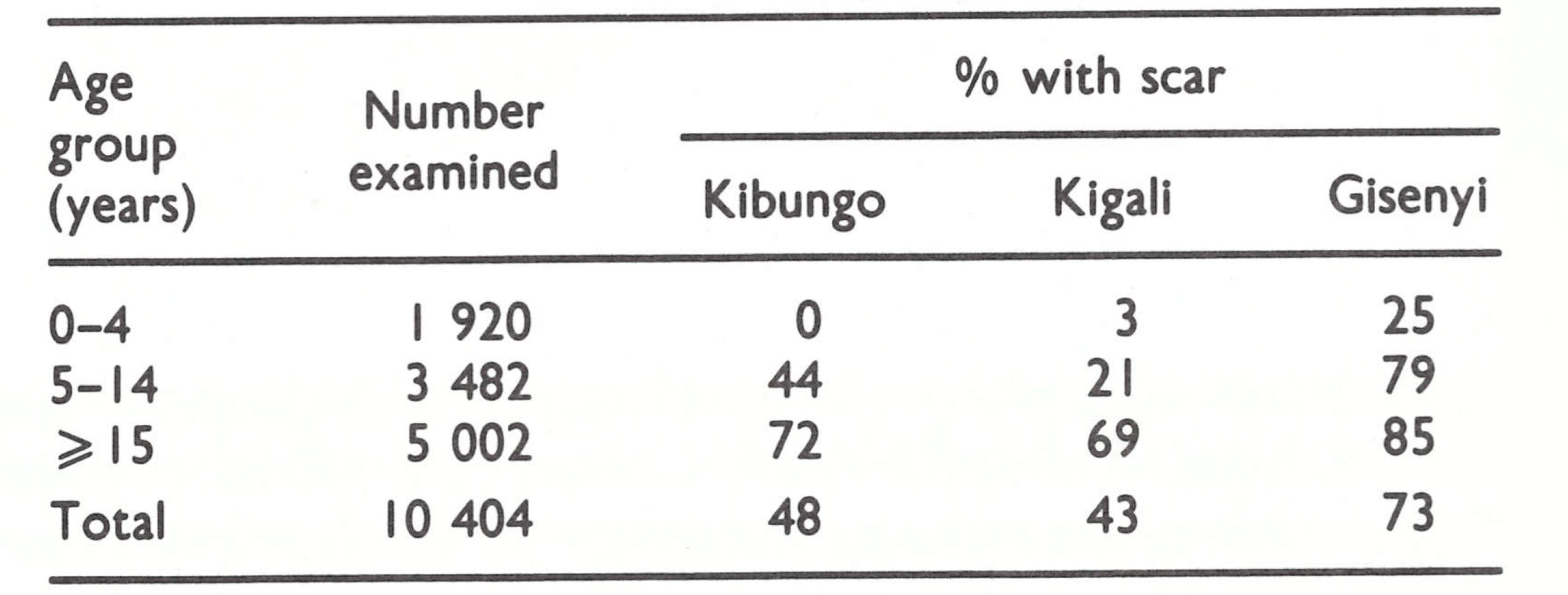
Vaccination in Rwanda had been performed primarily by 2 teams, each consisting of 3 persons, who shared a vehicle and travelled regularly to 400 vaccination points established at health facilities and schools. A sample scar survey conducted in 1968 of 10 404 persons in 3 different prefectures showed a generally low level of vaccinial immunity, especially among children (Table 19.12).
An agreement was signed with WHO in April 1968 which provided for the assignment of a WHO medical officer and the supply of vehicles and equipment, vaccine and needles, as well as for the costs of petrol and vehicle maintenance to be met by WHO. However, at the end of 1968, the Ministry of Health and WHO staff working in the country re-evaluated the plan and decided to combine the field activities with those of a BCG vaccination campaign then under way. Dr Algan, at that time the WHO adviser for tuberculosis control in the country, volunteered to assume responsibility for both activities.
In mid-April 1969, the programme began operations with a supervisor and 10 vaccinators who worked in pairs, one administering BCG vaccine and the other smallpox vaccine. Only a single vehicle was available, and so the team moved as a group to the various administrative units (prefectures), where vaccination was performed at collecting points. Areas from which cases were reported were attended to first. Hospitals and health clinics were supplied with vaccine and encouraged to participate in giving vaccinations.
As was the practice in other countries, Dr Algan and his Rwandan counterpart held meetings with local authorities beforehand to explain the programme and to identify the most suitable collecting points and work out the best schedule. Local political leaders were given the responsibility for convening the people on the appropriate day and, later, for the reporting of smallpox cases. Concurrent assessment of the campaign was by random sample evaluation to ascertain take rates.

Plate 19.5. A: Celal Algan (b. 1926), previously the WHO medical officer assigned to Rwanda for tuberculosis control, assumed responsibility in 1968 for organizing the smallpox eradication work in a campaign combining the administration of smallpox and BCG vaccines. The well-planned, rigorously supervised operation completed its 36-month work plan in half that time, while simultaneously developing an effective surveillance programme. Subsequently, as the WHO regional adviser on smallpox eradication in Africa (1975-1980), Algan organized the extensive activities required for the certification of eradication. B: Land Rovers move out for field work towing trailers with camping equipment. C: Rwandan vaccination team.
From the beginning, the team worked conscientiously and well, initially averaging 750 vaccinations per vaccinator per day and later as many as 1500 vaccinations per vaccinator per day. The productivity reported was in fact so high that the figures were viewed with scepticism by Ladnyi and WHO Headquarters staff. Ladnyi, however, confirmed the validity of the data during a special visit, noting in passing that the vaccinators complained only of sore fingers and wondered whether thimbles could be provided.
In October 1969, the vehicles intended for the programme were delivered and, with additional transport available, the complement of staff was increased to 27 (2 supervisors and 25 vaccinators, 5 of whom served as drivers); the personnel were divided into 3 teams, of which 2 were occupied with vaccination and the third, headed by Dr Algan, with surveillance-containment. By the end of 1969, 989000 persons had been vaccinated against smallpox by the mobile teams and 54 000 children had been given BCG vaccine.
During 1970, the investigation of 18 reported cases led to the discovery of 235 additional cases. After April, only 3 outbreaks, consisting of 3, 5 and 8 cases respectively, were discovered; each had resulted from importations from Zaire.
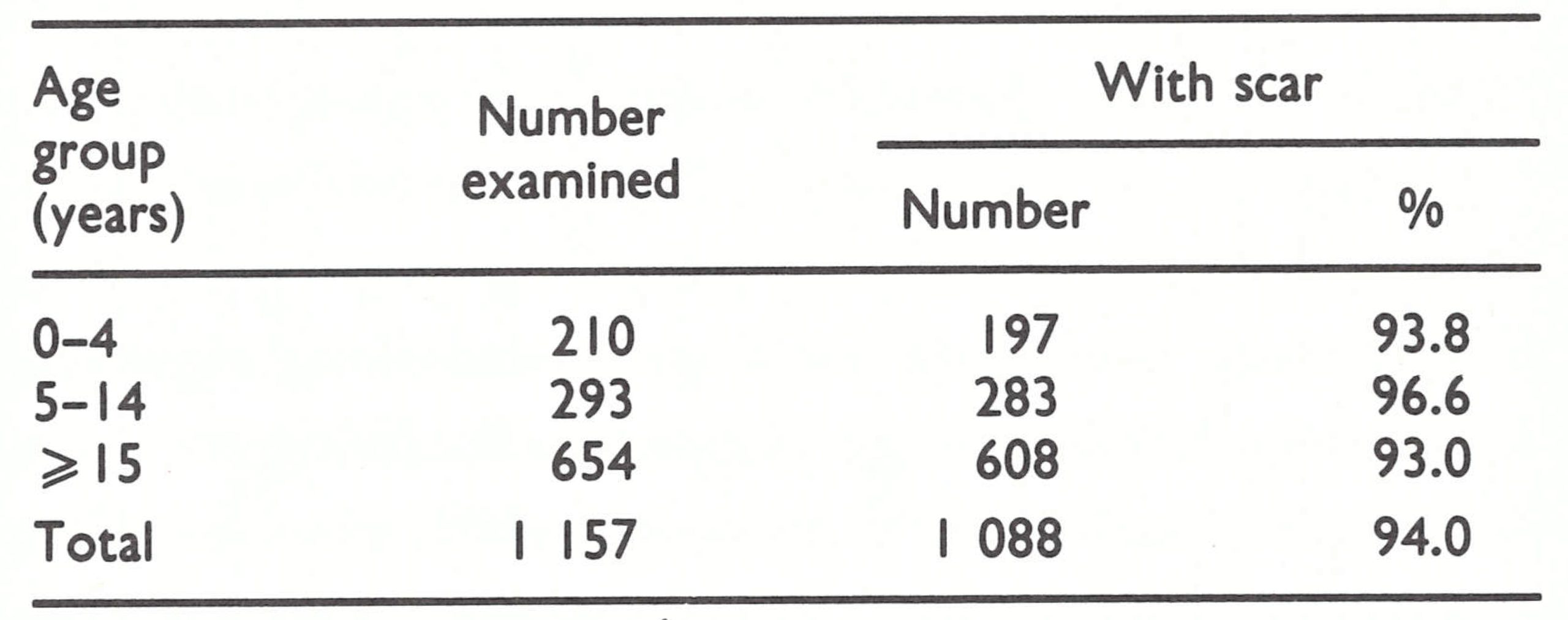
By the end of October 1970, only 18 months after the vaccination campaign began, the programme had been completed. During this period, the teams had vaccinated more than 3 million persons. A scar survey conducted in 1971 showed that, of those examined, 87% of children aged 0-4 years had vaccination scars, as did 97% of those aged 5-14 years and 93% of persons aged 15 years and over.
After the programme had been completed, hospitals and clinics continued to administer smallpox and BCG vaccines, while 8 teams, each consisting of 3 vaccinators, travelled throughout the more remote areas, especially those bordering Zaire, to search for cases and to vaccinate.

Plate 19.6. The recording form used in vaccination centres in Rwanda. The numbers of smallpox vaccinations and revaccinations administered could be quickly and easily noted by age group.
Burundi
Vaccination in Burundi had traditionally been performed by health units. In 1966, however, 500 000 persons were vaccinated during a special mass vaccination campaign undertaken after the occurrence of 1213 reported cases of what was said to have been variola minor. Sample surveys carried out by Ladnyi in 3 prefectures of Burundi in June 1968 revealed a remarkably high level of vaccinial immunity in all age groups in 2 of the areas in which the campaign had been conducted, and satisfactory levels in individuals aged 5 years and over in the third area (Table 19.13).
A plan of operations for an eradication programme was agreed on and signed in December 1967 by WHO and the government. How much smallpox might be present was unknown, since reporting was very incomplete. Only 74 cases were recorded in 1967, but the number rose to 301 in 1968.
In December 1968, a WHO medical officer arrived on assignment, along with supplies and equipment. Field activities began in May 1969 with 2 (later 4) mobile vaccination teams, each consisting of a supervisor, 4 vaccinators, an enumerator and a driver. The number of staff increased to 31 by mid-1969 and to 50 by 1971. The method of work was similar to that in Rwanda although never characterized by the same degree of careful planning and diligence. During 1969, only 415 705 vaccinations were performed, a number far lower than in Rwanda. Ladnyi, who visited the programme in February 1970, discovered that none of the vaccine was being kept under refrigeration, that vaccinators were issuing each vaccinee with a signed and officially stamped certificate of vaccination and that vehicle maintenance was virtually nil. The existing stocks of vaccine were destroyed and new supplies were flown in; the time-consuming practice of issuing individual certificates of vaccination was stopped; but a proposal to replace the WHO smallpox adviser was rejected. Fortunately, a new WHO country representative, Dr Christos Karamustakis, arrived at this juncture. An intelligent, industrious person who had worked previously in Zaire and knew the smallpox programme there, he assumed on a part-time basis an overall supervisory role and, thereafter, the programme proceeded better. However, despite a far greater complement of personnel than in Rwanda, and despite the absence of the requirement for simultaneous administration of BCG vaccine, the mass vaccination campaign in Burundi took more than 2 years to complete and did not conclude until June 1971.
In all, 2 997 500 vaccinations were performed between 1969 and 1971. No concurrent assessment of coverage was performed, nor were vaccinations checked to find out whether they were successful. Following the campaign, vaccinial immunity was not high, as was revealed in a 1973 sample survey conducted by a team from the WHO Epidemiological Surveillance Centre in Nairobi (Table 19.14).
The local health services were expected to continue the vaccination campaign but little had been done to obtain their cooperation and thus the number of vaccinations performed in 1972 dropped to only 30 000. Other WHO staff were brought in to help to organize an effective reporting and maintenance vaccination programme, and by 1973 reporting had begun to improve and the number of vaccinations to increase.
SMALLPOX OCCURRENCE, 1969-1970
Only 355 cases were reported from the 5 countries in 1969 and 484 cases in 1970. How accurately this reflects the true incidence of smallpox is difficult to assess. The eradication programmes consisted essentially of the traditional mass vaccination campaigns of the past, few of which incorporated either concurrent assessment of coverage or a special surveillance—containment element.
A national surveillance programme which served to ensure that all suspected cases were investigated and the outbreaks contained was a difficult concept for the countries to grasp and to implement. This was especially true of the former colonies of the United Kingdom, since their health services tended to be much more decentralized, the primary responsibility for health programmes being vested in district medical officers. This policy of decentralization of authority and responsibility offered many advantages in implementing a variety of programmes but not for the development of a national surveillance programme. District medical officers considered the control of smallpox to be specifically their own responsibility and resented the intrusion of a national presence. Where district medical officers were competent and diligent, this was not a problem; but many were not, and for all of them smallpox was but one of an array of problems and not always of high priority. A report of the occurrence of smallpox evoked the typical response that cases occurred in the province from time to time.
Ladnyi struggled to try to improve reporting and surveillance but, with responsibility for 19 countries, could devote little time to any single country. Moreover, he had been instructed by the regional office that he was not to undertake travel unless a visit was specifically approved in advance by the country and by the regional office, a process which he found to require at least 6 months. He ultimately decided to sidestep this restriction by sending a telex to the regional office informing it of his proposed travel plans and indicating that he would proceed unless instructed otherwise within the next 2-3 weeks. Rarely did the regional office reply in less than 4-8 weeks to any communication. A pattern therefore evolved in which Ladnyi proposed a trip and, receiving no reply, went ahead with his travel plans and eventually returned to Nairobi. After his return, he often received a telex advising him not to undertake the proposed trip.
In 1969, with so few cases being reported in eastern and southern Africa, it seemed that transmission would be interrupted quickly whatever the status of the vaccination campaigns. Accordingly, on 4 June 1969, Henderson wrote to the regional office and to Ladnyi in the following terms:
“I believe that it is absolutely, vitally and unequivocally essential that provision be made for a responsible person, preferably a medical officer, to investigate personally each and every case to verify the diagnosis and to assist with or carry out himself the necessary investigations and containment activities. Any plan not incorporating this particular feature is doomed to failure . . . Experience throughout the world both in endemic and in non-endemic areas has clearly shown that when full responsibility is entrusted to the district health authorities, the results obtained are highly irregular and frequently unsatisfactory .”
Interestingly, eastern Africa in particular was to disprove Henderson’s sweeping prediction of failure. The status of smallpox in 1969 was anything but clear. Kenya ceased to detect cases that year. Burundi recorded 108 cases and Tanzania 117 cases, but in neither country was there an adequate surveillance programme. In Rwanda, on the other hand, efforts were made to improve reporting and to investigate outbreaks. There, Dr Algan sought to obtain reports from the health units and, when outbreaks were found, redirected the programme to conduct mass vaccination in the infected areas. Virtually all cases in Rwanda were those detected by the special teams. It is probable that reporting was also somewhat more complete in 1969 in Burundi and Tanzania because vaccination teams worked in infected areas and detected cases in the process. However, it is likewise probable that some of the reported cases were misdiagnosed cases of chickenpox. Uganda, for example, originally reported 19 cases in 1969, but when these were investigated by a WHO consultant, only 9 were found to have been smallpox.
In 1970, Kenya reported no cases and Uganda only 2; the latter, when investigated by a WHO consultant, were found to have been infected in the Sudan and Rwanda respectively. Tanzania detected only 32 cases, the last in September. Twenty-three of the cases occurred following an importation from Zaire; the others were not investigated. In Burundi vaccination teams found 197 cases, about which nothing more is known ; the last of them occurred in October. Rwanda reported 253 cases in 1970, of which 18 were notified by the health services and the rest detected by the surveillance team. Transmission in Rwanda was interrupted in April 1970, although 3 outbreaks resulting from importations from Zaire occurred between August and October. Data regarding the age distribution of cases throughout 1969-1970 are available only from Rwanda (Table 19.15). Virtually all the cases were in children under 15 years of age, almost half of them under 5 years old.
Increased confidence that reporting was reasonably complete in 1970 was provided by the expanding vaccination campaigns, which served both to improve vaccinial immunity and to detect any cases that were present. In October 1970, the last cases were reported from these 5 countries, but not until nearly a year later could it be stated with confidence that transmission had actually been interrupted.
The rapid disappearance of smallpox from this area was surprising considering the perfunctory surveillance and containment activities. Levels of vaccinial immunity varied widely from place to place but, except where the systematic vaccination campaigns had been conducted, they were not generally high . Indeed, smallpox had disappeared from all but the densely populated areas of Burundi, Rwanda and Tanzania before the campaigns began. In retrospect, a combination of factors was probably responsible: (1) a generally scattered population with few large urban centres; (2) limited travel by most of the population, many of whom respected the numerous tribal boundaries; (3) prevalent, although not universal, tribal traditions which called for patients to be isolated in a separate hut and cared for by someone who had had smallpox; (4) a comparatively extensive network of health centres which, when freeze-dried vaccine was made available, successfully immunized a large proportion of people; and (5) the presence of a form of smallpox which spread less readily than did variola major in Asia and against which successful vaccination provided a longer-term immunity.
After 1970, outbreaks were reported only in Kenya and Uganda, in the former following importations from Ethiopia and Somalia, and in the latter from the Sudan.
IMPORTION INTO UGANDA
It is probable that transmission in Uganda was interrupted in 1968. Only 9 cases were detected in 1969, and although the sources of these outbreaks were not clearly traced, 6 occurred in Sudanese refugee camps in its northern districts and the other 3 in the south among refugees from Rwanda. In 1970, 1 case occurred in the north in a refugee infected in the Sudan and 1 case in a person infected in Rwanda. During 1971-1972, Uganda recorded 35 cases, all in its northern districts among Sudanese refugees and members of the local population who were in contact with them. These are shown as imported cases in Table 19.1.
Juba Province in the southern Sudan lay just across Uganda’s northern border and there a protracted civil war had been in progress. Tens of thousands of Sudanese refugees had taken up residence in northern Uganda and many continued to move back and forth between the two countries. The health services in Juba had been devastated by the war, vaccinial immunity was low, and, throughout 1972, smallpox was widely prevalent.
In October 1970, when Uganda had completed its mass vaccination campaign, it reverted to its previous pattern of operations, whereby vaccination was performed at clinics and health centres; district health inspectors assumed responsibility for surveillance activities and the containment of any outbreaks that were found. WHO recommended that a national surveillance team should be constituted to improve reporting and to participate in the investigation of any reported outbreaks, but this advice was not followed.
In 1971, 19 cases were reported from the northern districts of Acholi and Madi, 15 of which occurred in Sudanese immigrants; 4 local residents were infected by them. In 1972, 16 further cases were detected in this same area, primarily among Sudanese nationals. WHO staff who visited the area discovered that containment measures were not promptly taken, the investigation of outbreaks was perfunctory, containment was poorly executed and a number of cases reported locally were not notified to the national authorities. Fortunately, in this sparsely settled, reasonably well vaccinated area, the spread of smallpox was even less efficient than the performance of the staff.
In early 1973, a special training programme in surveillance-containment was conducted for the staff in northern Uganda, and in April a central surveillance team was constituted to investigate all suspected cases. A number of suspected cases were competently investigated and specimens obtained, but none proved to be smallpox. Meanwhile, Uganda’s only remaining endemic neighbours, Zaire and the Sudan, succeeded in interrupting transmission—Zaire in June 1971 and the Sudan in December 1972. Thus, the surveillance team began work in Uganda after the risk of importations had all but ceased, ironically paralleling the execution of its national vaccination campaign, which began after smallpox transmission had been interrupted.
IMPORTION INTO KENYA
Kenya, in contrast to Uganda, initially took more vigorous steps to improve reporting and established a national surveillance team in 1970. Six suspected cases were investigated in that year, all of which proved to be chickenpox. In March 1971, a nomadic herdsman from Ethiopia, travelling while ill, crossed the border and infected residents in a village some 70 kilometres south of the border. Subsequently 44 more cases occurred in 3 generations of transmission, 5 of the cases being fatal. The large proportion of deaths was unusual for outbreaks in the KenyaEthiopia area at this time, but whether the outbreak was due to variola major rather than variola minor is unknown. Mass vaccination had not yet been conducted in this area but, because of the outbreak, teams were quickly transferred to the district. Six thousand persons were vaccinated in and around the village, and the teams subsequently searched and vaccinated the entire district. No further cases were discovered.
In April 1971, another case was reported from a village 150 kilometres east of the first (Fig. 19.4). The patient was an Ethiopian adult visitor who resided just 14 kilometres north of the border. More than 95% of the village people had already been vaccinated and no further cases occurred.
The national mass vaccination campaign concluded in June 1972 and, at that time, the country was divided into 22 sectors, each with a mobile surveillance team which was supposed to be responsible for surveillance and for the vaccination of nomadic and other groups considered likely to import smallpox. By then, Ethiopia was Kenya’s only neighbour with endemic smallpox.
In Kenya the surveillance programme proved to be little better than in the other countries. In part, this can be ascribed to the fact that no surveillance teams had been established before transmission was interrupted, and thus they had no experience either in the investigation or in the containment of outbreaks. Moreover, the absence of cases understandably led to complacency. Sustaining interest in smallpox eradication after transmission had apparently been interrupted was difficult in all countries and especially difficult in Kenya. Following the outbreaks in March and April 1971, no further cases were detected for more than 2 1/2 years.

Plate 19.7. The strategy for search activities in Kenya during 1977 was worked out by Jean-Paul Ryst (b. 1939), a WHO consultant, Ziaul Islam (b. 1931), a WHO medical officer for epidemiological surveillance in Africa, 1971-1982, and Wilfred Koinange (b. 1939), Director of Health Services of Kenya.
On 30 December 1973, a 25-year-old man with smallpox was found in Mandera in north-east Kenya at the border with Ethiopia and Somalia. He had travelled 150 kilometres from an area in Ethiopia in which smallpox was known to be occurring. Containment vaccination was begun, and during the campaign a second case was discovered. The patient was a 23-year-old woman, who developed rash on 27 January and subsequently infected 2 other people. She had arrived in Mandera only 2 days before becoming ill and thus represented a second importation. The source was said to be a Kenyan village near the Ethiopian border, some 80 kilometres to the west. The fact that no investigation of this area was undertaken (although no further cases were reported) is indicative of the quality of the surveillance.
Again, 2 years were to elapse without cases. Meanwhile, Ethiopia reported its last cases in August 1976. In late September, however, Somalia reported its first cases for almost 2 years, in the capital, Mogadishu, supposedly resulting from importations from Ethiopia. As at the end of December, 39 cases had been reported, all in Mogadishu (see Chapter 22). When the report of cases in Somalia was received, a Kenyan team was sent to the border area to undertake an active search for cases. This team then represented virtually the only surveillance unit within 150 kilometres or more, since because of fighting in the area, Ethiopian teams had to remain at least that distance from the Kenyan border. Somali staff, if they were finding cases outside Mogadishu, were not reporting them.
That smallpox did not spread into Kenya must be attributed more to good fortune and to a sparse population than to the work of the inexperienced surveillance team. In December 1976, smallpox was again introduced into Mandera District (see Fig. 19.4). From the available, and somewhat conflicting, reports, it appears that a 40-year-old Kenyan man, who had been studying in an Islamic school near Mogadishu, became ill with smallpox on 26 December. He was then staying with his brother in a village, Kalaliyo, 29 kilometres west of Mandera town; 3 days later, he travelled some 35 kilometres south to a village, Ledi, to stay with his sister, and soon thereafter he returned to Somalia. The health officer in Mandera, informed that a case resembling smallpox had been seen in Ledi, sent a health inspector and a vaccinator. On arrival, they were told that the patient had left; they returned without vaccinating anyone. On 13 January 1977, a subchief reported that the patient’s sister had developed a rash. This time, the surveillance team went to Ledi, obtained a specimen and departed. Eleven days later, a report was received from a Nairobi laboratory that the specimen contained smallpox virus. On 26 January, the team again went to Ledi and found that the family had departed for a village (Kara) some 50 kilometres to the north-east. Another day elapsed before they could be found. Vaccination was then begun but 2 daughters became ill with smallpox on 22 and 27 January and another daughter (still unvaccinated) on 7 February. The patients were advised to stay in the house, but it was learned later that the mother regularly left the house to obtain water from a nearby waterhole frequented by a number of people.
Additional staff were sent from Nairobi to assist, and on 17 February 1977 WHO epidemiologists well acquainted with surveillance and outbreak investigation arrived. Mass vaccination carried out by 6 teams with vehicles in villages and towns accessible by road had commenced on 28 January. By midFebruary, the proportion of the population vaccinated reached 80% in the main villages, but only 30% in more remote villages. In fact, the 16 inhabitants of a village located just 2 kilometres from the outbreak were found, in early March, not to have been vaccinated at all. Meanwhile, because of heavy rains in the area, many nomads began moving through the area from Ethiopia as well as Somalia. At the end of April, a survey of 2256 persons in a nearby division revealed that only 527 (23%) had vaccination scars. By the end of February, a search had been organized throughout the eastern part of Mandera District which, by assessment, was reasonably effective. A reward of 200 Kenya shillings (US$24) was offered to anyone reporting a case. Gradually, monthly searches were extended to other districts bordering on Ethiopia and Somalia, but because of mud, vehicle breakdowns and scarcity of staff, probably not more than a half to two-thirds of the districts were satisfactorily searched. The searches continued until March 1978, some 6 months after the last known case occurred in Somalia.
It was feared that more cases would be found, but only 1 additional case was discovered by the search teams, in a 25-year old man who had become ill on 16 December 1976. The source of infection was not determined although it was considered to be unrelated to this outbreak. Meanwhile, efforts were made to find the original case in Somalia but it was never discovered.
CONCLUSION
The circumstances attendant on the eradication of smallpox from these 5 countries were wholly unlike those encountered in Asia or South America. In Asia, levels of vaccinial immunity comparable with, and in many instances better than, the levels observed in eastern Africa failed to stop transmission. Only when a fully effective surveillancecontainment programme was introduced could transmission be interrupted. In Brazil, surveillance and containment played a notably less important role in eradication, but there the vaccination campaign was carefully executed with specially trained assessment teams evaluating the performance of the vaccination teams on a daily basis. In Kenya, Uganda, and Tanzania, smallpox incidence was diminishing rapidly by the time the Intensified Programme began. In all but limited areas of Tanzania, and in Rwanda and Burundi, the introduction of freeze-dried vaccine of good quality was all that was required to interrupt transmission; the extensive infrastructure of health services did the rest. Mass vaccination campaigns, with neither assessment nor surveillance (except in Rwanda), served to eliminate smallpox in the other areas.
Whatever the deficiencies of the programmes, smallpox transmission was rapidly interrupted at little cost, with the addition of only a few experienced health staff, a small cadre of minimally trained vaccinators and encouragement and support from WHO. In retrospect, many factors account for this success: the substitution of freeze-dried for liquid vaccine; the ready, even enthusiastic, acceptance of vaccination by most of the population; the good cooperation of village leaders, missionaries, teachers and others; the presence of milder strains of variola virus which spread less easily; the generally sparse population and the infrequency with which they travelled over long distances; and an extensive network of health centres and clinics, many of which undertook to control outbreaks when these were discovered.
Of particular note in these countries was the extraordinarily high productivity of the staff compared, for example, with that of their counterparts in the Indian subcontinent. Whereas in the latter group of countries, the performance of 25 vaccinations by a vaccinator in one day was considered to be a worthy accomplishment, African vaccinators regularly vaccinated on average 500 or more individuals a day, the record being achieved in Rwanda, where during a particular month, the vaccinators, using bifurcated needles, averaged 1508 vaccinations per vaccinator per day. In the Indian subcontinent, it was considered necessary to have at least 1 smallpox vaccinator for every 5000-8000 or 20 000 persons, according to circumstances. In the countries of eastern Africa, on the other hand, the total smallpox eradication staff, at maximum strength, numbered only 1 per 148 000 persons in Kenya, 1 per 180 000 in Tanzania and 1 per 136 000 in Rwanda.
The rapidity with which success was achieved in these and other African countries, at such little cost and with minimum effort, provided enormous encouragement to the global eradication programme. To have interrupted transmission in countries in which health services were considered to be less effective and extensive than in Asia was an important factor in motivating Asian governments to make a more concerted attack on the problem.