Chapter 30:
Potential sources for a return of smallpox
INTRODUCTION
The eradication of smallpox was defined by successive WHO expert groups (WHO Scientific Group on Smallpox Eradication, 1968; WHO Expert Committee on Smallpox Eradication, 1972) as “the elimination of clinical illness caused by variola virus”. An important corollary of this definition was that it did not involve the extinction of variola virus, as some experts had urged. The procedures for the certification of smallpox eradication described in Chapter 24 were therefore designed to ensure that no human cases had occurred in the countries concerned for at least 2 years—i.e.,that the human-to–human chains of transmission of smallpox had been interrupted. Careful follow-up of rumours of suspected smallpox by national authorities and by WHO, described in Chapter 28, has failed to confirm a single case of smallpox in a field situation since the last case was recognized in Somalia in October 1977, although there were 2 laboratory associated cases in Birmingham, England, in August-September 1978. The correctness of the conclusions that the Global Commission for the Certification of Smallpox Eradication arrived at in its deliberations in December 1979 (World Health Organization, 1980) has been borne out by 10 additional years of freedom from the disease.
As the expert groups which defined eradication recognized, the absence of cases of smallpox is not synonymous with the extinction of variola virus, and if the virus is not extinct it is possible that further cases of smallpox could occur. This chapter is concerned with the known and hypothetical ways in which variola virus could be preserved and enter again into chains of human-to-human transmission. By far the most important, because it would be the most difficult to eliminate or circumvent, would be the continuing transmission of the virus in some wildlife reservoir or reservoirs. Indeed, such a situation would have made the permanent global eradication of smallpox an impossibility. This was therefore a matter that greatly exercised the staff of the WHO Smallpox Eradication unit from the outset of the global eradication programme (see Chapters 10 and 29). The lack of evidence of infection of humans from an animal host in Europe, North America and Australia, in which the disease no longer occurred, did not exclude this possibility for parts of Africa and Asia in which smallpox was still endemic until the 1970s. Although there was no documented evidence that this had taken place, it was conceivable that infection of man from an animal source might have occurred in the past without having been detected, as indeed proved to be the case with human monkeypox. Because of the reported isolations in laboratories in Bilthoven (Netherlands) and Moscow (USSR) of variola-like viruses (“whitepox” viruses) from animal tissues (see later in this chapter), investigation of this possibility remained a major concern of virologists involved in research associated with the eradication programme throughout the 1970s and into the early 1980s.
Material stored by variolators was another potential source for the recurrence of cases of smallpox which concerned the WHO Secretariat and members of the Global Commission, especially in countries such as Afghanistan in which low temperatures prevailed for much of the year. However, the most obvious source of virus that might cause human infection at some time in the future consists of laboratory stocks of variola virus, from which, indeed, the last known outbreak in the world originated (see Chapter 23). These could be stocks of virus known by WHO to be held in microbiologically secure laboratories, or they could be specimens in the deep-freeze storage facilities of any laboratory that had ever worked with the virus. Finally, stocks of virus may be held secretly, for possible use in microbiological warfare.
Other hypothetical sources of smallpox could be from virus released by reactivation in a human subject who had had the infection, or release of viable virus long preserved in scabs, on old clothes, or even in coffins. For the sake of completeness, the possibility that another species of Orthopoxvirus might be “transformed” into variola virus (already discussed in Chapter 2) needs to be briefly reconsidered here, as a possible source for the return of smallpox.
IS THERE AN ANIMAL RESERVOIR OF VARIOLA VIRUS?
The presence of an animal reservoir of variola virus is in theory the most important potential source for a return of smallpox. In discussing the origin of variola virus in Chapter 2, we suggested that conditions suitable for its perpetuation as a specifically human pathogen have existed for a few thousand years at the most. Variola virus was therefore probably derived from some closely related orthopoxvirus that survived in nature by circulation in an animal that occurred in large numbers at the time of early man and had a much shorter life-span. In this chapter two possibilities will be discussed: that there is an animal reservoir of variola virus as we know it, or that some other animal orthopoxvirus could be “transformed” into variola virus by a few mutational steps or perhaps by chance recombination with another animal orthopoxvirus. The great importance of this question, from the point of view of smallpox eradication, can be illustrated by reconsidering briefly the history of the first deliberate attempt to eradicate a human disease, yellow fever, which has already been mentioned in another context in Chapter 9.
The Example of Yellow Fever
In 1915 the International Health Commission of the newly established Rockefeller Foundation agreed to help with the global eradication of yellow fever, an undertaking that General W. C. Gorgas, Surgeon-General of the United States Army and the hero of disease control during the construction of the Panama Canal, considered eminently feasible. Yellow fever was regarded as an urban disease, and its global eradication was based on a simple epidemiological concept:
” . . . that the disease could be acquired only through the bite of an A. aegypti mosquito that had become infected by feeding on a human being sick with yellow fever; that there were certain endemic centers of the disease that served as seedbeds ; that these foci of infection were few in number, and that if they were destroyed, yellow fever would disappear forever .” (Warren, 1951.)
It is a matter of history that this concept was too simple. As early as 1907 Franco et al. (1911) had recognized the existence in Colombia of another epidemiological situation—the contracting of yellow fever in the forest, rather than in urban areas. Another outbreak of yellow fever occurred in the same locality in 1916, but since Aedes aegypti could not be found there, it was assumed by the Gorgas Commission that the diagnosis was erroneous (Gorgas, 1917). Eventually Soper (1935, 1936) drew attention again to the paper of Franco et al. and described what he called “jungle yellow fever”, which he postulated could be due to maintenance of the virus in another vertebrate host or perhaps long persistence in invertebrate vector. At about the same time surveys of forest monkeys of many species in both Africa and South America revealed the presence of neutralizing antibodies in their sera. With the discovery of a vertebrate reservoir other than man, it was clear that the global eradication of yellow fever was impossible, although urban yellow fever could be eliminated by ridding towns and cities of Aedes aegypti.
The significance of the yellow fever experience was not lost on those who contemplated the global eradication of smallpox and investigations into the possibility that there might be an animal reservoir of variola virus in Africa or Asia were initiated soon after the Intensified Smallpox Eradication Programme was launched (see Chapter 10).
Smallpox in Apes and Monkeys
Experimental observations with variola virus had demonstrated that, unlike vaccinia and cowpox viruses, both of which have a wide host range, few laboratory animals could be infected with variola virus, except under unusual conditions (see Chapter 2). However, it had been demonstrated during the latter part of the 19th century that monkeys (probably Macaca mulatta) could be infected with variola virus (Zuelzer, 1874 ; Copeman, 1894), and later studies (Hahon, 1961) showed that many species of monkeys and apes were susceptible. Noble (1970) found that 3 species of New World monkeys that he tested were insusceptible to variola minor virus, although they reacted serologically but without symptoms to experimental infection with variola major virus.
Reported infections of primates in nature
Arita & Henderson (1968) reviewed the published accounts of supposed smallpox in primates as well as other naturally occurring epidemics of “pox” infections among monkey populations. Only 8 such episodes are known and only 4 of these occurred during the present century (Table 30.1). In only 2 instances was laboratory confirmation available; in each of these there had been close association between the primate concerned and cases of human smallpox .
“Smallpox” infection in a monkey population in the forests of southern Brazil was reported by Bleyer (1922), who noted that carcasses of Mycetes seniculus and Cebus capucinus were found under the trees, the dead animals having fallen from the tree-tops. Sick as well as dead monkeys were covered with numerous pustules like those seen in human smallpox and the mortality was extremely high in certain districts . Anderson (1861) reported that a “smallpox” outbreak observed in 1841 in monkeys was followed by a smallpox outbreak in a human population in Panama. Two monkeys that he examined were covered with “perfectly formed” pustules. Schmidt (1870) mentioned 3 other episodes, 2 in France and 1 in Trinidad.
An outbreak of a vesiculo-pustular disease was observed by M. A. Rahman (quoted by Arita & Henderson, 1968) in rhesus monkeys in Bengal, India, in 1936 . Many deaths were observed among monkeys which lived in mango groves near the town and visited the town frequently in search of food and water. The sick monkeys were quiet and lethargic and had pustular lesions, particularly on the face, palms and soles . When they died they fell from the trees and roof tops, and the carcasses were disposed of without any particular precautions being taken. Despite the poor vaccinial immunity status of the population, no human pox-like illnesses were observed.
All these episodes must be regarded with caution so far as their significance as evidence of an animal reservoir of variola virus is concerned. They may have been instances of the infection of primates from human cases of smallpox or they may have not been due to variola virus or indeed to any kind of poxvirus. However, 2 episodes have been well documented which demonstrate that primates in close contact with smallpox-infected humans may have become infected with variola virus. Gispen (1949) observed 2 orang-utans in the Jakarta Zoo which contracted a pox infection at the time of a smallpox epidemic in the area. Both animals were in the same cage and demonstrated typical lesions on the face, hands, and soles of the feet; Bras (1952a) performed an autopsy on the one that died (Plate 30.1). Variola virus was isolated on the chorioallantoic membrane of the chick embryo from specimens taken from both affected animals. Other monkeys in the zoo, none of which had been previously vaccinated, remained unaffected. The other episode occurred in India, where Mack & Noble (1970) recorded an unusual situation in which 1 or possibly 2 performing monkeys (Macaca mulatta) appear to have been infected with variola virus by intimate household contact with human cases of smallpox.

Plate 30.1. Orang-utan (Pongo pygmaeus) in the Jakarta Zoo, which was infected during the 1949 epidemic of smallpox and died.
Throughout the global smallpox eradication campaign attempts were made to identify the source of infection in all outbreaks, and as the incidence of smallpox fell to low levels in each endemic country, this became a matter of high priority, so that chains of transmission could be traced. Thousands of such outbreaks in Africa and south-eastern Asia were investigated by skilled epidemiologists. In the vast majority, infection could be traced to a case of smallpox. Very rarely fomites (e.g., laundry items or burial shrouds) were implicated. In a few instances it was not possible to determine the source of the outbreaks. Some of them may have been due to infection acquired from mild unrecognized cases, or the source remained undetected through faulty or incomplete investigation. But the possibility of infection from an animal source was always present in the minds of epidemiologists in the rare instances in which no human source could be clearly identified . Never during the global smallpox eradication campaign could smallpox be traced back to an animal source. Finally, although monkeys are common in many of the countries of Africa and Asia in which smallpox was once endemic, sometimes living in close contact with man or captured by hunters, no case of smallpox has been found in any of these countries since eradication.
Experimental transmission of smallpox between primates
More significant than isolated examples of smallpox in monkeys or the infection of animals by inoculation are 2 sets of observations on the natural transmission of variola virus from one primate to another. Noble & Rich (1969) showed that serial infection could be maintained for as many as 6 successive passages in Macaca irus (cynomolgus) monkeys placed in contact with other monkeys during the period of rash, but transmission then failed (Fig. 30.1). Among chimpanzees, Kalter et al. (1979) observed that 2 animals situated in cages near a chimpanzee experimentally inoculated with variola virus contracted smallpox, one suffering a severe illness. One other animal in the group escaped infection. Clearly, primates of several species are susceptible to variola virus, get a rash when infected, and can transmit the disease to other primates in contact with them. However, in Macaca virus, the only primate in which adequate studies were conducted, the infection persisted with some difficulty and then died out.
As was discussed in Chapter 2, smallpox could not be maintained in isolated human populations numbering under 200 000. Unless smallpox in non-human primates produced a situation in which recurrent infectivity could occur years after the primary infection, it is likely that populations of any particular species of monkey would be too small to maintain the disease. This is certainly true of chimpanzees and orang-utans, in which infection with variola virus causes a severe disease not unlike smallpox in man.
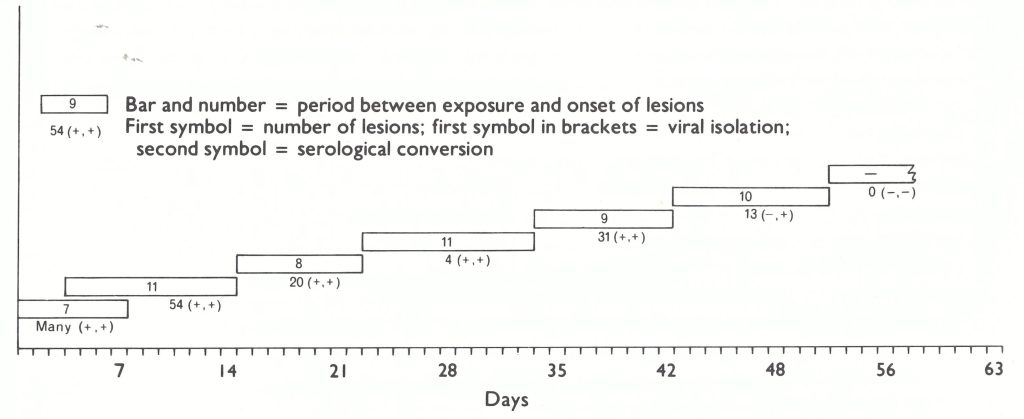
Fig. 30.1. Five successive contact infections of Macaca virus philippinensis before failure in transmission. The first monkey was infected by intranasal inoculation and after 7 days (indicated in bar) developed many lesions, from which variola virus was recovered (first “+” symbol) and subsequently showed serological conversion (second ” + ” symbol). The second monkey was placed in the same cage 3 days after the inoculation and developed 54 lesions II days after exposure; it was then placed in contact with the third monkey, and so on. The penultimate monkey in the series developed 13 lesions, but virus was not recovered from the crusts, although antibodies developed. The last monkey failed to develop lesions or convert serologically. (Based on Noble & Rich, 1969.)
Monkeypox
It had been known since 1958 that monkeys in laboratory colonies occasionally suffered epizootics due to another orthopoxvirus—monkeypox virus (Magnus et al ., 1959; see Chapter 29). Known outbreaks of monkeypox were reviewed by Arita & Henderson (1968), and early in 1969 the WHO Smallpox Eradication unit convened an Informal Group on Monkeypox and Related Viruses (see Chapter 29) to advise it on matters relating to the problem of an animal reservoir of variola virus. The following year one of the members of the Informal Group (Dr Svetlana Marennikova) recognized that the virus recovered from a suspected case of smallpox that had occurred in Zaire 2 years after the last outbreak of smallpox in the area (Ladnyj et al., 1972) was indeed monkeypox virus (Marennikova et al.1972b). Subsequently, human monkeypox has come to be recognized as a rare zoonosis apparently confined to villages in tropical rain forests in central and western Africa (see Chapter 29). The significance of human monkeypox in the present context lies in observations made in the course of ecological surveys in Zaire designed to elucidate the natural history of monkeypox virus . These resulted in a series of reports on what came to be called “wild whitepox” and ultimately “whitepox” viruses.
“Whitepox” Virus Isolations
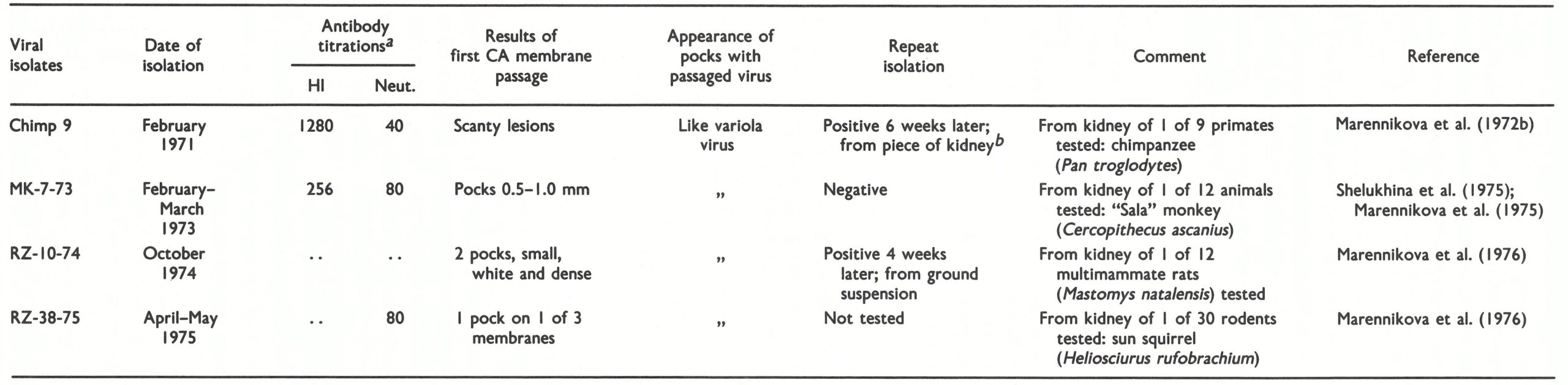
Three sets of isolations of “whitepox” viruses have been made which are best dealt with separately and chronologically. The first set (“Netherlands isolates”) comprises 2 strains, designated 64-7255 and 64-7275, apparently recovered from normal cynomolgus monkey kidney cell cultures at the National Institute of Public Health at Bilthoven (Gispen & Kapsenberg,’ 1966 ; Gispen & Brand-Saathof, 1972). The second set (“Zaire isolates”) comprises 4 strains of virus reported by workers at the Moscow Research Institute for Viral Preparations to have been recovered from the tissues of 4 different species of wild animal captured in Zaire between 1971 and 1975 (Table 30 .2). The third set (“white clones”) comprises several isolates made from preparations of monkeypox virus maintained at the Moscow Research Institute for Viral Preparations (Marennikova & Shelukhina, 1978 ; Marennikova et al ., 1979).
Table 30.2: Details of the circumstances of the recovery in Moscow of “whitepox” viruses from wild animals captured in Zaire
 a Varlola virus Isolated.
a Varlola virus Isolated.
a HI=haemagglutinin-inhibition, Neut,= neutralization of vaccinia virus on chorioallantonic (CA) membrance; figures indicate reciprocal of titre;..=data not recorded.
b Special precautions to prevent contamination with variola virus were taken at time of reisolation (personal communication from S.S. Marennikova).
White Pock Mutants, "Whitepox" Viruses and White Clones (Variants) of Monkeypox Virus
The appearance of pocks produced on the chorioallantoic membrane by different species of Orthopoxvirus varies from bright red (cowpox virus), through a greyish ulcerated appearance with a haemorrhagic centre (monkeypox virus and some strains of vaccinia virus including rabbitpox virus) to a dense white pock with no sign of ulceration (variola virus, some strains of vaccinia virus and camelpox virus). All viruses that produce haemorrhagic pocks also yield a small proportion of non-haemorrhagic pocks; these are the "white pock mutants" of cowpox, rabbitpox, neurovaccinia or monkeypox virus, which have been used for genetic studies of orthopoxviruses.
In 1966 Gispen & Kapsenberg reported the recovery of orthopoxviruses from apparently normal cynomolgus monkey kidney cells used for viral diagnostic work in the National Institute of Public Health, Bilthoven. One of them was vaccinia virus; the other 3 were regarded as monkeypox virus. However, when studying these "monkeypox" virus isolates, Marennikova et al. (1971) showed that one of them resembled variola virus and was clearly distinguishable from monkeypox virus. Gispen & Brand-Saathof (1972) confirmed this result with 2 of the 3 "monkeypox" virus isolates and in the same paper observed that a white pock mutant that they recovered from monkeypox virus resembled the parental virus in most properties and was clearly distinguishable from variola virus. To discriminate between them, the variola-virus-like strains were called "wild white" poxvirus, or "whitepox" virus.
Between 1971 and 1975 Marennikova and her colleagues at the Moscow ResearchInstitute for Viral Preparations reported that they had recovered white-pock- producing orthopoxviruses from the tissues of 4 species of animals shot in Zaire (chimpanzee, monkey, sun squirrel and multimammate rat). All isolates resembled Gispen's "wild white" poxvirus (and thus variola virus) in all the biological properties that they could test. For purposes of reference, the term "whitepox" virus was used to include all six of these viruses (Arita & Henderson, 1976).
In 1978-1979 Marennikova and her colleagues reported that they had obtained "whitepox" by inoculating monkeypox virus on the chorioallantoic membrane and in hamsters. They called these isolates "stable white clones" or "white variants" of monkeypox virus.
In this way two terms came to be used by virologists associated with the smallpox eradication programme:
(1) White pock mutants—of rabbitpox, cowpox and monkeypox viruses; and
(2) "Whitepox" viruses—from normal cynomolgus monkey kidney cells, from apparently normal wild animals shot in Zaire, or found as "stable white clones" or "white variants" of monkeypox virus.
Netherlands isolates
During 1964 and 1965 4 orthopoxvirus isolations were made from cynomolgus monkey kidney cells that were being used for the isolation of viruses from human material in the diagnostic laboratory at the National Institute of Public Health, Bilthoven (Gispen & Kapsenberg, 1966). One strain (64-9411) was eventually diagnosed as monkeypox virus (see Chapter 29) and one strain (65-3993) was vaccinia virus, recovered together with herpes simplex virus from vesicle fluid (J. G. Kapsenberg, personal communication, 1981). The other 2 strains (64-7255 and 64-7275) were first regarded as o monkeypox virus (Gispen & Kapsenberg, 1966); subsequently Marennikova et al. (1971) reported that strain 64-7275 differed from monkeypox virus in a number of, A biological characteristics but could not be differentiated from variola virus. This finding was confirmed by Gispen & Brand Saathof (1972), who later showed that these 2 strains differed from monkeypox virus and resembled variola virus in tests for virus. specific antigens (Gispen & Brand-Saathof, 0 1974). Subsequently Esposito et al.(1978) and Dumbell & Archard (1980) demonstrated E that the DNA electropherograms and restrico tion endonuclease maps of these 2 isolates, while differing slightly from those of some other strains of variola virus, identified them a unequivocally as members of the variola virus species.
Although in a comment at the International Symposium on Smallpox Vaccine, Bilthoven, in 1972 Gispen (1973) stated, correctly, that “there was no known possible contact of these animals [from which the kidney cell cultures infected with whitepox virus had been derived; our italics] with pox viruses”, it subsequently transpired that on 2 occasions in September 1964, material from smallpox patients from Vellore, India, had been handled in the diagnostic laboratory in which the cynomolgus kidney cells were being used. Skilful detective work by Dr Kapsenberg identified the passage transfer of cell cultures during which viral contamination could have occurred; there was a temporal coincidence with manipulation of the cynomolgus cells in question and cells infected with material from the smallpox cases. That laboratory contamination was the explanation was demonstrated with a high degree of confidence by the fact that the biological properties and restriction endonuclease electropherograms of strains 64-7255 and 64-7275 were identical with those of 1 of the 2 variola viruses isolated from the Vellore cases, which were slightly different from those of mosother strains of variola virus (Dumbell & Kapsenberg, 1982).
Zaire isolates
Five strains of poxvirus were recovered from the kidneys of wild animals captured in Zaire and processed in the Moscow Research Institute for Viral Preparations. Four were identified as “whitepox” viruses (Table 30.2), and the fifth (MK-10-73) as vaccinia virus.
Pocks were always scanty on the membranes on which they were first observed. All subsequent investigations, including detailed DNA mapping carried out independently in 2 laboratories (St Mary’s Hospital, London, England, and the Centers for Disease Control, Atlanta, USA), have confirmed that the 4 strains of “whitepox” virus are indistinguishable from variola virus.
A priori it is difficult to believe that a virus which has such a narrow host range as variola virus should be recovered from the tissues of 2 species of healthy rodents that are common in and near tropical forest villages in Central Africa, the rat Mastomys natalensis (syn.coucha) and the sun squirrel Heliosciurus rufobrachium. This view is reinforced by the observation that variola virus and “whitepox” virus strain RZ-38-75, said to have been recovered from Mastomys natalensis, multiplied very poorly after intraperitoneal inoculation in that animal (T. Kitamura, personal communication, 1978).
Since the Moscow Research Institute for Viral Preparations functioned as one of the two WHO collaborating centres that carried out laboratory diagnosis of smallpox for the Intensified Smallpox Eradication Programme (see Chapter 10), specimens of variola virus were constantly being handled in the laboratories in which the “whitepox” virus isolates were made. The universal experience of laboratories which have handled orthopoxviruses, including the experience of the National Institute of Public Health in Bilthoven, just described, attests to the difficulty of avoiding occasional cases of laboratory contamination.
Laboratory Contamination with Viruses
Bacteriologists are accustomed to the idea that unwanted organisms may occasionally find their way into culture media and multiply there. Indeed, the discovery of penicillin resulted from one such episode (Fleming, 1929). Similar accidents may occur with viruses, especially if manipulations are of the kind that produce aerosols. Only a few cases of the contamination of cultures have been reported (e.g., Andrewes et al., 1944). Another kind of environmental contamination is the accidental infection of laboratory workers (see, for example, Pike, 1979; Wedum et al., 1972), which is an indicator of the possibility of infection of laboratory cultures.
H. Mahnel (personal communication, 1984), concerned about the occasional occurrence of vaccinia virus plaques on "uninfected" cell monolayers, carried out tests with vaccinia and monkeypox viruses under conditions simulating those in the laboratory. The infectivity of tissue culture fluid dried on coverslips and stored in Petri dishes in the shade in the laboratory (temperature 20-23°C) dropped from 10 5 .8 plaque-forming units per ml to 10 2 .8 plaque-forming units per ml in 12 days, but the dried material did not completely lose infectivity until the 6th week. He believes that the principal source of laboratory contamination of cultured cells arises from droplets of infected fluid drying on bench tops and in the dust of the laboratory.
Although the risk of cross-infection can be reduced by good microbiological technique, there is an ever-present possibility of the contamination of tissue cultures or eggs. Almost every laboratory manipulation is associated with some risk of producing aerosol particles that could contain virus. Dimmick et al. (1973) have drawn up a table listing the "spray factors" of various laboratory operations and accidents. Assuming that a viral suspension contains 10 7 pock-forming units per ml, it is possible to calculate the number of infectious aerosol particles released as a result of normal laboratory operations or minor accidents.
| Operation | Spray Factor | Virions per m³ of Working Area |
|---|---|---|
| Blender opened 5 minutes after stop | 10-5.3/min | 50 |
| Pipetting, minimal bubbling | 10-6.3/min | 5 |
| Intranasal inoculation of mice | 10-7.3/min | 0.5 |
| Drop spilled from a height of 100 cm | 10-6.3 | 5 |
| Removal of plug from test tube | 10-8.5 | 0.03 |
The only certain way of avoiding cross-infection with poxviruses is to use only one isolate at a time, within a glovebox facility or a biocontainment hood, and thoroughly sterilize the working area before handling another strain of virus.
Without special markers and laborious experiments, it was very difficult to "prove" that laboratory contamination had occurred, as Dumbell & Kapsenberg (1982) were able to do, but every experienced laboratory scientist who has worked with poxviruses can recall instances for which he or she believed that contamination was the most likely cause of an unexpected result.

Plate 30.2. Farida Huq (b. 1942) undertook graduate studies with Dr K. R. Dumbell in London before assuming responsibility for the smallpox laboratory in Dhaka, Bangladesh. Besides carrying out diagnostic work she studied the viability of variola virus in scabs under tropical conditions.
Two features caution against too ready an acceptance of this explanation for all the Moscow “whitepox” virus isolates: the positive orthopoxvirus antibody titres found in 3 of the 4 animals whose tissues apparently contained virus, and the reported reisolation of virus from part of the stored kidney of Chimp 9 (from a chimpanzee) and from a ground-up suspension of kidney tissue of RZ38-75 (from a sun squirrel). However, investigations of the ecology of monkeypox in Zaire, reported in detail in Chapter 29, suggest that there may be several orthopoxviruses circulating among wild animals in the forests of Zaire ; the antibody results signify prior infection with an orthopoxvirus, not necessarily variola virus or even monkeypox virus. Reisolation is the standard procedure for confirming the validity of suspicious isolates, but to be acceptable it must be carried out with material that had not been processed in the laboratory at the time the first isolation was made. This situation did not apply to the material from Heliosciurus, but it did apply to the kidney of the chimpanzee (S.S. Marennikova, personal communication, 1983), a species which is known to be susceptible to infection with variola virus (Kalter et al ., 1979). However, Chimp 9 was one of the two “whitepox” viruses tested by Dumbell & Huq (1986), who showed that it resembled Asian rather than African strains of variola virus in its biological characteristics (see below).
The number of strains of variola virus whose DNAs have been mapped by restriction endonuclease cleavage is too small to allow any conclusions to be drawn about the relationship of these “whitepox” isolates to other strains of variola virus from western or central Africa. However, comparison of 4 biological characteristics of 32 variola virus strains originating from the Indian subcontinent, western Africa or Zaire with those of the “whitepox” strains Chimp 9 and MK-7-73 (purportedly recovered from primates captured in Zaire) showed that these “whitepox” isolates had the same group of biological characteristics as the great majority of strains of Asian variola major and were quite unlike 12 strains of variola virus derived from western Africa or Zaire (Table 30.3). Dumbell & Huq (1986) concluded that these two “whitepox” viruses (Chimp 9 and MK-7-73) “did not enter into the chains of transmission which maintained human variola in West Africa.
In summary, and in view of the results set out in the next section, it seems likely that the “whitepox” virus isolates made in Moscow were due to laboratory contamination with Asian strains of variola virus.
Table 30.3: The distribution of 4 independent biological properties characteristic of Asian variola major virus according to the geographical origin of the strains testeda
 a Varlola virus Isolated.
a Based on Dumbell & Huq (1986).
a Varlola virus Isolated.
a Based on Dumbell & Huq (1986).
b The characters were: chick embryo virulence; ceiling temperature; haemadsorption on human embryo fibroblast monolayers; and haemagglutinin production in HEp 2 cells.
c Chimp 9 and MK-7-73 (see Table 30.2).
“White clones” (“variants”) of monkeypox virus
Seeking an explanation for the recovery of whitepox virus strains from the tissues of several species of wild animals from Zaire, Dr Marennikova postulated that they may have arisen as “white clones” of monkeypox virus, which was known to infect non-human primates in that country (see Chapter 29). Several investigators (Bedson, 1964; Gispen & Brand-Saathof, 1972) had previously isolated white pock mutants of monkeypox virus, but found them to resemble the parental virus rather than variola virus in properties such as species-specific antigenicity (Gispen & Brand-Saathof, 1974) and the pattern of their intracellular polypeptides (Harper et al.,1979). In this section attention will be focused on two reports of what Marennikova and her colleagues call “stable white clones” recovered from stocks of monkeypox virus that had been maintained for several years in the Moscow laboratory. All these “white clones”, recovered either as pocks on the chorioallantoic membrane (Marennikova et al.,1979) or from organs of hamsters that had been inoculated some weeks earlier with the monkeypox virus material (Marennikova & Shelukhina, 1978), were identical; they were indistinguishable by biological tests from the Zaire and Bilthoven “whitepox” virus isolates, which have now been shown to be variola virus.
In view of the importance of this claim as to the origin of “whitepox” viruses, from the point of view of the feasibility of smallpox eradication, the WHO Smallpox Eradication unit organized a series of studies to determine whether these results could be independently confirmed. The studies in question were conducted in the WHO collaborating centres in London and Atlanta. Dumbell & Archard (1980) reported on 17 white pock mutants recovered on the chorioallantoic membrane from their own stocks of the Copenhagen strain of monkeypox virus and compared their biological characteristics and DNA maps with those of wild-type monkeypox virus, the Harvey strain of variola virus, the 2 Netherlands “whitepox” isolates, 2 Zaire “whitepox” viruses and 2 “white clones” recovered from monkeypox virus by passage through hamsters (Marennikova & Shelukhina, 1978). All the “whitepox” viruses and the 2 “white clones” had DNAs that were virtually identical with that of variola virus (Table 30.4). The monkeypox white pock mutants differed among themselves in both genome maps and biological properties (Table 30.5). Some, which had been recovered after serial passage of the monkeypox virus stock at high concentrations, showed complex genome changes, with deletions and transpositions; some showed minor differences from wildtype monkeypox virus, others were indistinguishable from it. In all cases the DNA maps were readily distinguishable from those of variola and “whitepox” viruses (Fig. 30.2).
Table 30.4: The derivations of various viral strains and the nature of their genomes as determined by restriction endonuclease analysisa
 a Based on Dumbell & Archard (1980) and Esposito et al. (1985).
a Based on Dumbell & Archard (1980) and Esposito et al. (1985).
b var = DNA pattern like that of variola virus; mp = DNA pattern like that of monkeypox virus; mp, del = DNA pattern like that of monkeypox virus, with terminal deletion; mp,mod = DNA pattern like that of monkeypox virus but with complex modifications.
Table 30.5: Phenotypic characteristics of white pock mutants of monkeypox virus
 a Mutants described by Dumbell & Archard (1980).
a Mutants described by Dumbell & Archard (1980).
b Numbers of mutants in each group. There were differences (not shown) within groups in the appearance of the white pocks, the size of the lesions produced In rabbit skin and the extent of growth in pig embryo kidney cells.
c Mutants described by Esposito et al. (1985).
d Not tested.
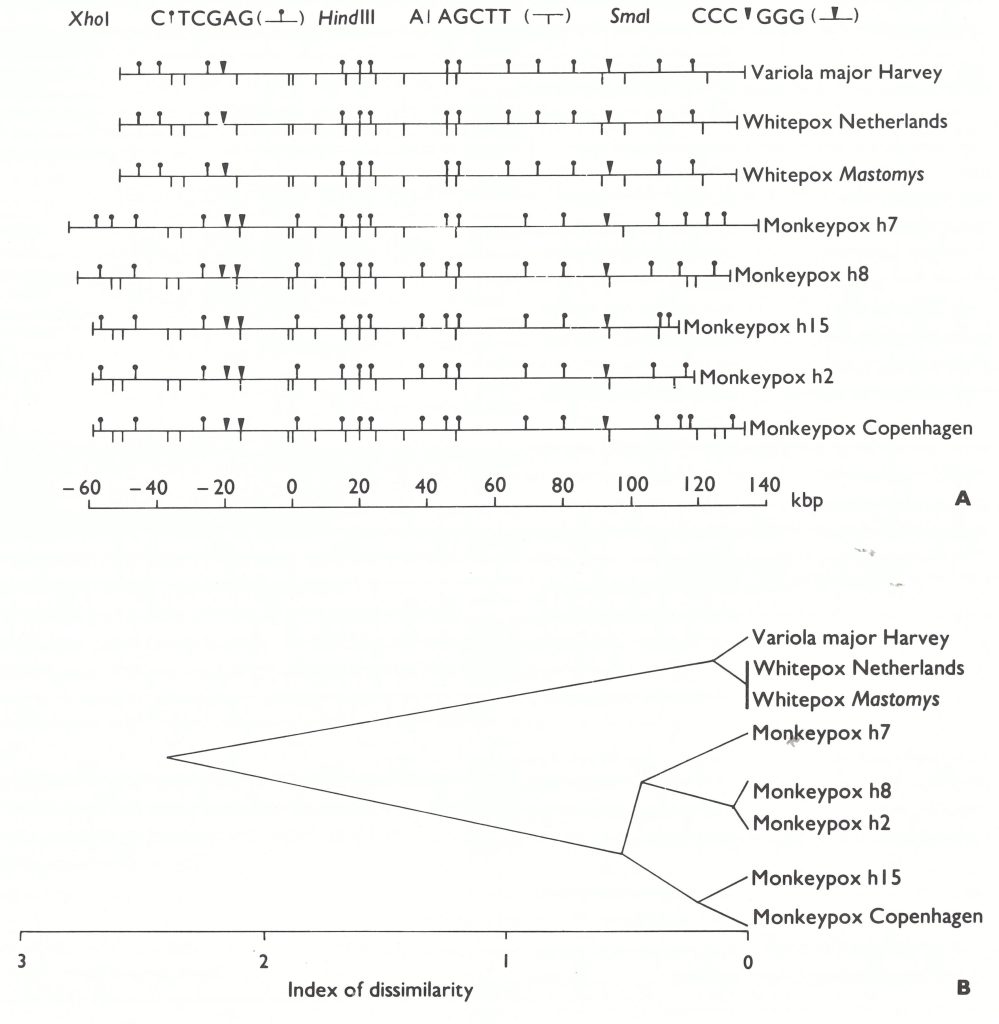
Fig. 30.2. A: Physical map locations of Hindlll, Xhol and Smal cleavage sites in DNA from variola, “whitepox” and monkeypox viruses: Harvey strain of variola major, Netherlands “whitepox” 64-7255, “whitepox” Mastomys (= Moscow RZ-10-74), Monkeypox Copenhagen, and 4 white pock mutants derived from Monkeypox Copenhagen (Monkeypox h2, h7, h8 and h IS. (Based on Dumbell & Archard, 1980.) B: Dendrogram illustrating the similarities and differences between these DNAs. Presence, absence or impossibility (because DNA molecules were too small) of coincidence of the cleavage sites illustrated was determined after aligning all maps on a common Hindlll cleavage site about 60 kilobase pairs from the left-hand end of the molecule; the results were then analysed as described by Gibbs & Fenner (1984), using the squared Euclidean metric (number of attributes= 60). The “index of dissimilarity” has no absolute value but illustrates the close resemblances between the DNAs of variola virus and the 2 “whitepox” viruses, and between the DNAs of monkeypox virus and the 4 mutants derived from it.
Studies at the Centers for Disease Control (CDC), in Atlanta, confirmed the results obtained in London. Four white pock mutants—2 derived in Atlanta from CDC stocks of a monkey strain of monkeypox virus (Copenhagen) and 2 from their stocks of a human strain (Congo)—resembled monkeypox virus in both their DNA maps (Table 30.4) and their biological characteristics (Table 30.5). On the other hand, Esposito et al. (1985) reported that isolations of white pock mutants made by a member of the staff of the Moscow laboratory working in the CDC, but not using the glovebox facility, had biological properties and DNA maps identical with those of variola virus. On the basis of a detailed analysis of all the data, these authors concluded that “the spontaneous production of whitepox from monkeypox virus would be genetically impossible”, and that “the whitepox viruses recovered from monkeypox virus stocks had originated exogenously”.
This work was carried out in 1978 and 1979, and in an assessment of the situation in 1980, on the eve of the declaration of smallpox eradication by the Thirty-third World Health Assembly, Fenner et al. (SE/80.154) concluded that “the variety of mutants derivable from monkeypox in the laboratory does not present any greater threat to the success of smallpox eradication than does the existence in nature of orthopoxvirus species other than variola.
Conclusion: There is No Animal Reservoir of Variola Virus
The evidence reviewed in the foregoing pages suggests that smallpox was indeed a specifically human disease. Variola virus is a distinctive species; DNA analysis of strains of widely differing virulence for man (variola major and variola minor viruses) shows that their DNAs are very similar, but quite different from those of any other species of orthopoxvirus or from those of any viral mutants that have been recovered under conditions that precluded the possibility of laboratory contamination. Variola virus has a narrow host range; besides man the only animals in which serially transmissible infection is known to have occurred are chimpanzees, orang-utans and one species of monkey. The “whitepox” viruses isolated from a variety of animals between 1964 and 1975 are strains of variola virus. The most acceptable explanation is that all of them were laboratory contaminants.
The laboratory evidence must be seen in both epidemiological and evolutionary perspectives. Apart from known contaminants (the Bilthoven viruses), the 4 “whitepox” viruses were recovered from areas of Zaire in which human monkeypox occurred and continues to occur more frequently than anywhere else in the world. The isolation rate in Moscow for the recovery of a virus from wild animals was high (4 out of 61 animals from the areas that were tested, compared with none of 930 animals from the area tested by the Centers for Disease Control, Atlanta, primarily in a search for monkeypox virus). Since human monkeypox is a zoonosis and intensive surveillance in selected areas of Zaire between January 1982 and December 1984 revealed 210 cases of human monkeypox, of which 144 were thought to have an animal source (see Chapter 29), there is clearly the kind of contact between humans and animals in these tropical rain forests that would make infection of humans with variola virus likely, if indeed it occurred in the animals there. However, all cases of “suspected smallpox” in western and central Africa since 1970 that were due to orthopoxvirus infection have been shown by epidemiological and laboratory investigation to have been caused by monkeypox virus.
MATERIAL STORED BY VARIOLATORS
Variolation was a matter of concern as a source of outbreaks of smallpox during the operations of the Intensified Smallpox Eradication Programme in Afghanistan, Pakistan and Ethiopia (see Chapters 14 and 21). In addition, it was known that an outbreak in northern Yunnan Province in China in 1958 was due to variolation, 126 cases having occurred a year after the last known case in that subregion. Late in 1984 it was learned from Dr Jiang Yutu, in a personal communication, that there had been other outbreaks of smallpox in North China (Nei Monggol Autonomous Region (Inner Mongolia) and 2 nearby counties in Shanxi Province) which were ascribed to the activities of variolators and the first of which occurred some 6 years after the last reported cases of smallpox in these regions (see Chapter 8, Table 8.13; and Chapter 27). The explanation for these outbreaks (Jiang Yutu, 1985; and personal communications, 1984, 1987) appears to be that variolators in Shanxi and adjacent parts of Inner Mongolia continued to keep variolation material, stored with honey in sealed jars, and maintained its potency by annual passage in susceptible family members and by the addition of fresh material to the jars. The 1962-1964 and 1965 outbreaks were initiated by such variolated persons but the chains of transmission have not been elucidated. The occurrence of these outbreaks long after smallpox had been eradicated from China led to measures that eliminated this source of the disease and no case of smallpox has been recorded in the country since 1965.
Apart from these episodes in China, which did not come to the attention of WHO until 1984, major concern with the danger of variolators’ material causing outbreaks of smallpox after the interruption of transmission centred on Afghanistan. Tests for viable virus in material collected from variolators there gave positive results in one sample 9 months after the material had been collected from a patient; all the others were negative long before this (see Chapter 14, Table 14.15). All the variolators who were questioned about their mode of operation said that, if it was available, they preferred to use fresh material (from a recent case), and as smallpox became less common they usually added fresh scabs or pustule fluid to their stored material whenever possible. One sample tested contained herpesvirus particles, doubtless derived from a misguided attempt to keep viable stocks for variolation.
Within scabs, out of sunlight and in cool surroundings, viable variola virus can survive for several years. However, variolators’ material was never held under such “ideal” conditions, and tests showed that it rarely contained viable virus for as long as one year. The major factor that now further reduces the risk presented by variolation material as a source for the recurrence of smallpox is the passage of time. The last cases of smallpox in countries in which variolation was widely practised during the 20th century occurred in 1965 (China), 1973 (Afghanistan), 1974 (Pakistan) and 1976 (Ethiopia). In Ethiopia, in which smallpox was the most recently endemic, variolation was usually carried out with fresh pustular material when an epidemic threatened, and material was rarely stored (see Chapter 21). The lapse of 10-20 years since the last cases of smallpox in the other countries has reduced the incentive to practice variolation, and even under the best storage conditions (short of refrigeration) the viability of variola virus gradually falls off. The absence of any recurrence of smallpox due to variolation for 10 years or more gives reason to believe that this practice will never again initiate an outbreak of smallpox.
LABORATORY STOCKS OF VARIOLA VIRUS
While the existence of an animal reservoir was without doubt the most serious risk to the achievement of the permanent eradication of smallpox, the existence of stocks of variola virus in laboratories constituted and, while such stocks of variola virus are retained, will continue to constitute a real though remote risk that further cases of human smallpox could occur.
Changes in Attitude towards Work with Variola Virus
The handling of variola virus has been subject to progressively stricter control over the years. In Jenner’s day variolation was extensively practised, with no control or supervision of the way in which the virus was handled or administered and usually little or no check on the movements of the inoculated subject. Jenner, and vaccinators after him, did not hesitate to test the efficacy of vaccination by challenge inoculation with variola virus. Variolation, which involved the uncontrolled distribution of variola virus, was not made illegal until 1840 in Great Britain and 1870 in British India, and, as has been noted above, it was widely practised in Afghanistan up to the early 1970s and in Ethiopia until as late as 1976.
Pathologists who were interested in smallpox and vaccinia experimented with variola virus as a matter of course and with few precautions, secure in the protection afforded by a previous attack of smallpox or by repeated vaccination. The attitude of virologists towards handling the virus in the 1950s is revealed in the report on experiments on viability by Wolff & Croon (1968), who stored scabs from a patient in an envelope in a cupboard in their laboratory in 1954, and tested one scab annually for the presence of variola virus. At that time reliance was placed on vaccination and revaccination of all laboratory workers, good laboratory technique, and careful disposal and sterilization of infected material and glassware. Biohazard hoods had not then been invented and work with variola virus was carried out on open benches, although special cubicles were sometimes used when the virus was handled on a large scale. In countries in which smallpox was still endemic-for example, in India, Africa and South America-material for smallpox diagnosis was regarded, with reason, as being less hazardous to handle in the laboratory than, for example, material containing the tubercle bacillus.
Safety Regulations in Laboratories Holding and Handling Variola Virus
Recommendations made in 1969 and 1974
The first comment by WHO on precautions to be taken when handling variola virus in the laboratory appeared in the publication Guide to the Laboratory Diagnosis of Smallpox for Smallpox Eradication Programmes (World Health Organization, 1969a), which was prepared by 2 virologists with extensive experience in handling variola virus (Dr A. W. Downie and Dr J. Noble) and 2 epidemiologists (Dr I. Arita and Dr A. S. Benenson). The guide suggested that methods that ensured high standards of microbiological safety should be employed and that everyone who might have occasion to enter the laboratories should be vaccinated annually . However, recommendations on physical containment were relatively simple (Fig. 30.3A)—a separate room entered via a vestibule, but no provision for sterilization of the ambient air, for biosafety hoods or for 2-way autoclaves.
Following the accidental infection of a laboratory worker in London in 1973 (see Chapter 23), virologists in the United Kingdom interested in smallpox drew up a code of safety practice (Cox, 1974). This code formalized arrangements for vaccination and revaccination and for the first time suggested the use of safety cabinets and emphasized the risk of aerosol production during laboratory manipulations. It recommended that animal inoculation should be performed only in “institutes with specially designed facilities”.
During the middle and late 1970s laboratory safety in general became a much more prominent issue. There were several reasons for this, including deaths from Lassa and Marburg fevers contracted in the laboratory, the invention and development of sophisticated methods for the physical containment of infectious agents, and the widespread concern for laboratory safety engendered by the introduction of recombinant DNA technology.
Safety measures recommended by WHO in 1977
Because of this change in attitude towards laboratory safety, and the imminent achievement of the global eradication of smallpox, the WHO Smallpox Eradication unit in August 1977 organized a meeting of experts to discuss safety measures in laboratories retaining variola virus. The expert group included senior staff from the WHO collaborating centres for smallpox research in London, Tokyo, Moscow and Atlanta, and Dr J. H. Richardson of the Office of Biosafety at the Centers for Disease Control, Atlanta. Recommendations for physical containment and the packaging and shipping of specimens were drawn up (SME/77.2). Although smallpox laboratories in the WHO collaborating centres had to a greater or lesser degree already installed the physical facilities outlined in this report, it was the first official statement by WHO prescribing the kinds of physical containment necessary for holding 1335 and handling dangerous pathogens, with strict control of entry, facilities for air and biowaste sterilization, gloveboxes (see Plate 30.3), 2—way autoclaves and biological safety cabinets (see Fig. 30.3 B), as well as the usual stipulations about vaccination and regular revaccination.
The laboratory-associated outbreak in Birmingham, England, in August-September 1978 (see Chapter 23) caused serious concern among the general public and the health authorities of many countries. Reacting to this concern, the WHO Smallpox Eradication unit arranged a meeting in Geneva in April 1979 to study the safety measures adopted in laboratories then retaining stocks of variola virus and to discuss the destruction or transfer of the virus if the laboratory concerned was not using it for research. Government officials from the 7 countries and scientists from the 8 laboratories that then retained variola virus attended the meeting and reviewed the situation (WHO/ SE/79.137), with special reference to the role of national authorities in maintaining safety standards. They agreed on modifications to the WHO recommendations for physical containment in accordance with further experience with such facilities; these were issued as WHO document SME/77.2 Rev. 1 and later reproduced in Annex 9 to the final report of the Global Commission (World Health Organization, 1980). Eventually, in March 1983, the WHO Committee on Orthopoxvirus Infections, noting that the Organization, through its Special Programme on Safety Measures in Microbiology, had published a book entitled Laboratory Biosafety Manual (World Health Organization, 1983) which provided a standard reference for a maximum containment laboratory, recommended that WHO document SME/77.2 Rev. 1 should be withdrawn.
Over the several years after the 1977 meeting steps were taken to ensure that the facilities in the WHO collaborating centres in Atlanta and Moscow were upgraded, and new laboratories were built in each centre. It is not widely appreciated how difficult it is to build a highly secure biocontainment laboratory and maintain it properly, particularly in relation to airflow. For various reasons it took more than 3 years, to construct and test the new facilities in both centres, and all research work with variola virus ceased during this period, although studies with variola virus DNA, which is not infectious, continued in Atlanta. Between August 1981 and March 1983 urgent diagnostic work on specimens from cases of suspected smallpox or monkeypox was carried out in the Special Viral Pathogens Laboratory at the Centers for Disease Control in Atlanta. Work with variola virus in other laboratories that held the virus in 1979 (see Table 30.6) ceased at various times between 1979 and 1982, when their stocks were transferred to a variola virus storage facility in the Centers for Disease Control.
Plate 30.3. Glovebox facility (Class III biological safety cabinet) in use at the WHO collaborating centre in the Centers for Disease Control, Atlanta, GA, USA. To eliminate the risk of laboratory contamination, only a single specimen was handled at a time and the cabinet was sterilized with peracetic acid before another specimen was introduced.
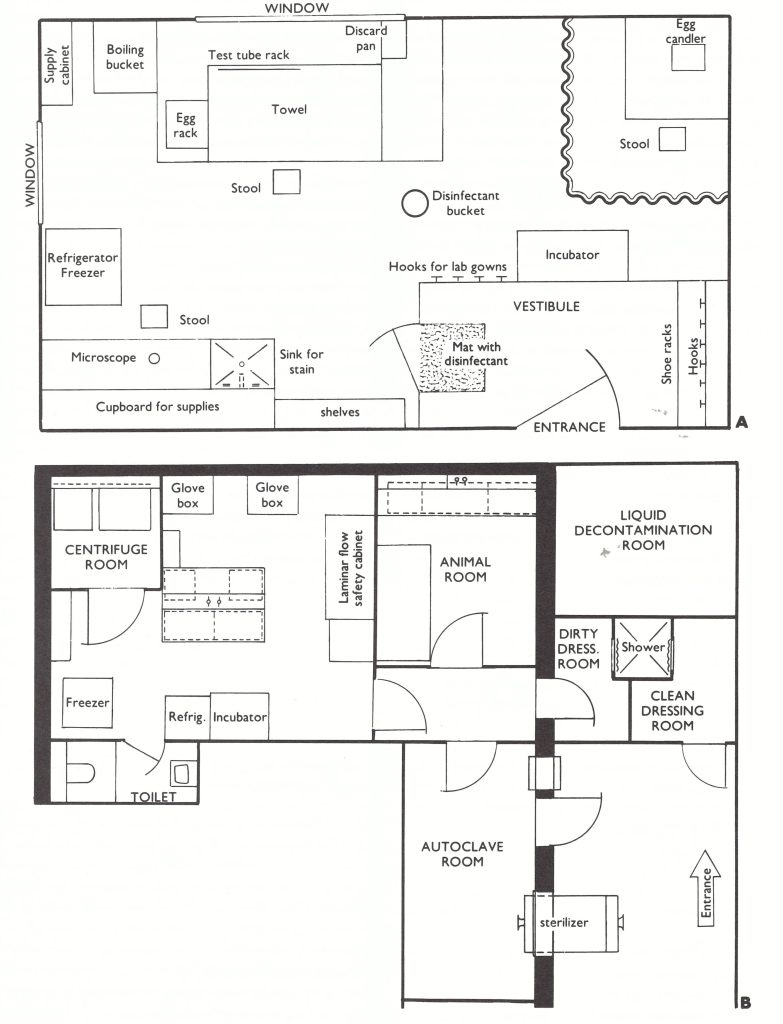
Fig. 30.2. A: Suggested layout for a smallpox diagnostic laboratory as conceived in 1969. (From World Health Organization, 1969a.) B: Plans of part of the smallpox laboratory at the Centers for Disease Control, Atlanta, GA, USA, drawn in 1979. Note the much more stringent physical containment, with access and exit of personnel through a shower and access of materials through a decontamination box and exit via a two-way autoclave. Effluent air is passed through a HEPA (high-efficiency particulate air) filter and liquid and solid wastes are sterilized by heat treatment. Within the laboratory, virus is handled within a glovebox or under a laminar flow hood and special precautions are taken with centrifugation and other laboratory manipulations.
The Maximum Containment Laboratory
The 1977 report and the Laboratory Biosafety Manual recommended the use of a maximum containment laboratory for the handling of dangerous pathogens. In essence, this is a room or a few rooms (or, for veterinary laboratories handling exotic viruses, a complete laboratory building) in which special measures are taken for the physical containment of dangerous pathogens, to minimize the risk of their escape outside the laboratory. The principal features of a maximum containment laboratory are:
- Controlled access. The entry and exit of personnel and supplies are through airlock systems. On entering, personnel put on a complete change of clothing, and they shower on exit before changing back into their street clothes.
- Controlled air system. Negative pressure is maintained by an individual supply and exhaust air mechanical ventilation system with HEPA (high-efficiency particulate air) filters in the exhaust (and in the intake when necessary).
- Decontamination of effluents. All effluents from the maximum containment laboratory are rendered safe, including the shower water.
- Sterilization of waste and materials. A double-door pass-through autoclave is provided.
Properly installed, operated and maintained, these arrangements should prevent the escape of dangerous pathogens from such a laboratory. To protect personnel from infection and to minimize the risk of contamination of apparatus and cultures, an additional facility can be operated within the maximum containment laboratory. This is called the primary containment facility, of which there are 3 forms:
- A fixed “Class III” biosafety cabinet, in which material is handled via glove ports and all effluent air is passed through a HEPA filter.
- A flexible-film plastic equivalent of a Class III biosafety cabinet.
- A positive-pressure ventilated suit, operated only in a special laboratory which provides a decontamination shower for personnel leaving the risk area.
The WHO collaborating centre in Atlanta handled all specimens of known or suspected variola virus within a Class III biosafety cabinet. One specimen was handled at a time and the cabinet sterilized with peracetic acid before another specimen was introduced. This practice enabled that laboratory to avoid completely any cases of laboratory contamination of cultures by its personnel, throughout its operations in smallpox diagnosis and research.
Two of the 19 recommendations of the Global Commission (World Health Organization, 1980), all of which were accepted by the Thirty-third World Health Assembly in May 1980, dealt with the inspection of WHO collaborating centres approved to hold and handle variola virus. It was recommended that each laboratory that still held variola virus (whether or not a WHO collaborating centre) should report annually to WHO on its safety measures and should be inspected periodically by WHO. The implementation of these recommendations has been described in Chapter 28.
Justification for the Retention and Use of Variola Virus
The potential danger of experimentation with variola virus in a laboratory not equipped with full microbiological security facilities was dramatically demonstrated by the Birmingham outbreak, discussed in Chapter 23, some 10 months after the last known case of naturally transmitted smallpox had been reported in Somalia. In response to the concern expressed by many WHO Member States, a consultation of public health and virological experts who were not themselves involved in laboratory work with variola virus was convened in Geneva in February 1979. Its report (WHO/SE/79.135) was subsequently used by the Global Commission for the Certification of Smallpox Eradication in formulating its final report (World Health Organization, 1980). In essence, the consultation recommended that research with variola virus was justified, at least for the next 3 years, because of the problem then posed by reports of the isolation of “whitepox” viruses and the work on DNA mapping that provided a method of at least partially solving this problem. In January 1979, the WHO Executive Board had asked the Secretariat to recount what measures were being taken in response to the Birmingham outbreak. The Smallpox Eradication unit was able to inform the Board of the proposed consultation and the anticipated meeting on laboratory safety measures, mentioned in the previous section, which were designed to deal with the essence of the problem—namely, whether further research with variola virus was necessary and, if so, whether the laboratories in which such work was undertaken were microbiologically secure.
Table 30.6: Laboratories holding variola virus stocks in 1975 and the reduction in this number by July 1977, in response to requests from WHO
 a Mutants described by Dumbell & Archard (1980).
a Mutants described by Dumbell & Archard (1980).
a Although China did not respond to the WHO letter of request, stocks of variola virus were then held at the Institute for the Control of Drugs and Biological Products, Beijing.
Reduction in the Number of Laboratories Retaining Variola Virus
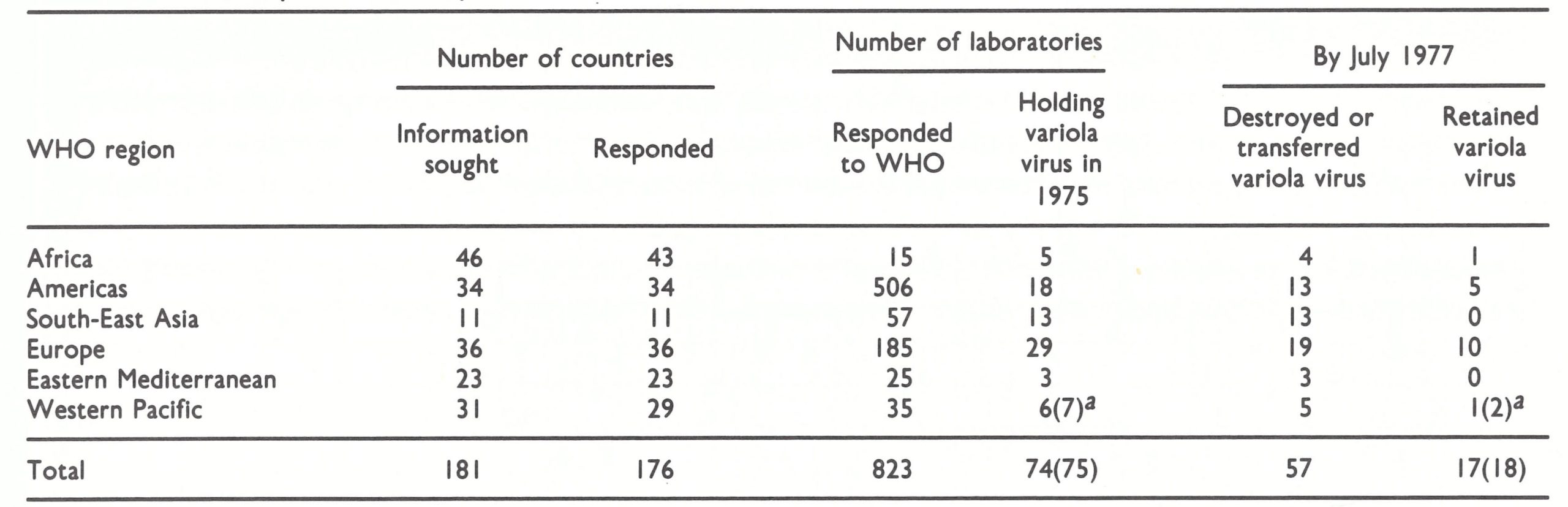
The possibility of escape of variola virus from either of the two high-security laboratories that now retain it is extremely remote. But in 1976, as global smallpox eradication appeared imminent, stocks of the virus were held by many more than these two laboratories; in fact, no one knew how many more. It was reasonable to argue that the smaller the number of laboratories holding variola virus, the lower would be the risk of the virus escaping. The Twenty-ninth World Health Assembly, in May 1976, requested “all governments and laboratories to cooperate fully in preparing an international registry of laboratories retaining stocks of variola virus” but, at the same time, urged all laboratories which did not require such stocks of variola virus to destroy them (resolution WHA29.54). This resolution followed inquiries that the WHO Smallpox Eradication unit had addressed in 1975 both to governmental authorities and to the directors of 823 laboratories included in the WHO World List of Virus Laboratories. The unit had also scanned the Index Medicus in search of references to papers on variola virus published from laboratories during the previous few decades and had written to the directors of these laboratories to find out whether they held stocks of the virus. This information having been obtained, laboratories other than the WHO collaborating centres in Atlanta and Moscow and the WHO laboratories for poxvirus research were asked to destroy their stocks or transfer them to one of the two WHO collaborating centres. Governments were asked to inform WHO of the response to this request. Prior to this, the Director of the National Smallpox Eradication Programme in India had carried out a similar survey in that country and reported that by the end of 1976 all stocks of variola virus in laboratories in India had been destroyed (Basu et al., 1979). The situation in the 6 WHO regions in 1975 and in July 1977 is shown in Table 30.6.
Some noteworthy inferences can be drawn from this table. First, the response from both national authorities and laboratory directors was remarkably comprehensive. Apart from China, the countries that failed to respond were small in size and population and were known not to have a laboratory that had ever handled variola virus. Secondly, there were no fewer than 75 laboratories, several in each WHO region, that were then holding variola virus. Indeed, the real figure was somewhat higher, for it is known that virologists in several laboratories discovered that they did still possess variola virus, after having, in good faith, declared to the contrary; in such instances, the stocks were privately destroyed or taken to the nearest WHO collaborating centre for destruction. This widespread possession of stocks of variola virus is not surprising. Smallpox was endemic in several populous countries and importations into Europe, especially, had occurred in the recent past. In order to have “controls” for the laboratory diagnosis of agents that they rarely handle, such as variola virus, virologists usually keep strains of these agents in their deep-freeze cabinets, for comparison with a suspicious isolate. Provided that their staff were regularly vaccinated and used good microbiological techniques, laboratory directors rightly regarded variola virus as less dangerous to handle than other agents sometimes encountered in diagnostic laboratories, such as various rickettsias . Further, even though smallpox might no longer be endemic in their country, it did not occur to them to destroy their laboratory stocks of variola virus. However, in response to the request from WHO, and bearing in mind the imminent world-wide eradication of smallpox, the directors of 57 of the 74 laboratories agreed to destroy or transfer their stocks of the virus.
Philosophical Considerations regarding the Destruction of Variola Virus
From time-to-time commentators in scientific journals have questioned the moral rectitude of destroying all known stocks of variola virus, on the grounds that man should not knowingly cause the extinction of any living thing. With the cloning of variola virus DNA this raises some interesting philosophical problems. Are variola virions, in an ampoule, "living things"? In fact, they are inert until their genetic potential can be expressed, and the only "natural" form of living variola virus would be that found in a human being, since man was the only known host of the virus. Presumably, the periodic occurrence of cases of smallpox is not what conservationists have in mind when they argue that variola virus stocks should never be totally destroyed.
The other problem is whether the cloned fragments of variola virus can be regarded philosophically as still constituting the essence of life of that virus . Certainly, molecular biologists are, or soon will be, able to produce all the known viral products from such cloned fragments. They could even, with considerable (albeit misguided) effort, reconstitute the intact variola virus DNA molecule and thus by laboratory manipulations, the virus itself.
When viewed against the regrettable but whole extinction of species that results from human interventions in natural ecosystems, concern about the preservation of variola virus seems to us to be misplaced. The only criterion by which to judge the necessity for the preservation of the virus, we believe, is whether it is necessary for scientific work.
Further reduction in the number of laboratories retaining variola virus, 1977-1983
With strong support from the Consultation on the Worldwide Certification of Smallpox Eradication, convened in Geneva in October 1977, and subsequently from the Global Commission, WHO continued to exert pressure to try to reduce further the number of laboratories holding variola virus stocks. In its final report in December 1979 (World Health Organization, 1980) the Global Commission recommended that: “No more than four WHO collaborating centres should be approved as suitable to hold and handle stocks of variola virus. A collaborating centre would be approved only if it had adequate containment facilities.” Biocontainment standards had been laid down for such laboratories (SME/77.2 Rev.1) and it was stipulated that each laboratory that held variola virus (whether it was a WHO collaborating centre or not) should be periodically inspected by WHO.
The Birmingham outbreak had a dramatic effect on the attitudes of the directors of several European laboratories, because of Professor Bedson’s suicide and the extensive press coverage of the event (see Chapter 23). By the end of 1979, 5 of the remaining 8 European laboratories holding variola virus had disposed of their stocks (Table 30.7). Every year, at the World Health Assembly, Member States continued to express concern about variola virus stocks in laboratories. Gradually, the remaining laboratories holding such stocks responded to the requests from WHO to destroy or transfer their holdings, since with the eradication of smallpox the rationale for retaining stocks of the virus in any laboratory except the WHO collaborating centres in Atlanta and Moscow had disappeared. Scientific developments, notably the cloning of restriction fragments representing the total genome of variola virus (see Chapter 2), removed the last objections, voiced by the Ministry of Health of South Africa (see Chapter 28, Plate 28.4), and by the end of 1983 only 2 laboratories in the world, both with newly completed biocontainment facilities, still held stocks of variola virus: the WHO collaborating centres at the Centers for Disease Control, Atlanta, and the Moscow Research Institute for Viral Preparations . At a meeting in March 1986, the Committee on Orthopoxvirus Infections recommended that all stocks of variola virus should be destroyed in October 1987, subject to approval by an ad hoc committee to be convened by WHO.
There remains the possibility that ampoules containing variola virus may still be stored in deep-freeze cabinets in laboratories, unknown to anyone in the laboratory or indeed in the country concerned. In most laboratories deep-freeze cabinets are rarely thoroughly cleaned out, though sections of them may be. As staff replacements occur and smallpox recedes into the past, all memories of working with variola virus will be lost. Two such incidents have come to the notice of the WHO Smallpox Eradication unit—the discovery of variola virus stocks in a laboratory in California in 1979 (Emmons, 1979), and in a laboratory in the United Republic of Tanzania during the visit of a WHO international commission in 1979 (J. G. Breman, personal communication, 1979). Another possible but unconfirmed example was described in London in December 1985. The dangers posed by such forgotten material are very small provided the laboratory carries out the recommended safety measures—namely, the destruction by autoclaving of any unlabelled ampoules or any ampoules labelled “variola virus” or “smallpox virus”. This risk is hypothetical and in any case beyond the control of WHO or any other organization.
DELIBERATE RELEASE
The United Nations (1970) and a WHO group of consultants (World Health Organization; 1970) both included the smallpox virus among some 20 infectious agents that could be used in biological warfare. However, it ranked low in suitability compared with several other viruses, rickettsias and bacteria. As the WHO consultants commented:
“Because of the effectiveness of the vaccine and the relative ease with which it can be produced 1341 and administered, it is unlikely that smallpox virus would be used as an agent for a large-scale biological attack against countries systematically practising periodic vaccination.
“The variola virus, however, can easily be employed for acts of sabotage . Such acts, at selected points within a country, could have serious socioeconomic effects unless efficiently dealt with by the public service.”
That was written in 1969 . At present no country is “systematically practising periodic vaccination” and as time passes the immunity of those who have been vaccinated will wane and populations will become completely susceptible.
In 1972 many countries of the world signed a convention outlawing the use of biological weapons in warfare (United Nations, 1984). Unfortunately, as international conventions are subject to infringement, this does not completely exclude the possibility that variola virus might be deliberately released as a means of warfare. However, the risk of any such act leading to the re-establishment of endemic smallpox should not be exaggerated. As has already been mentioned, smallpox spread comparatively slowly, by face-to-face contact. Unless the public health services had completely broken down, the existence of reserve stocks of vaccine (see Chapter 28) and the capacity for the production of vaccine to be rapidly reactivated would ensure the containment of any outbreak that followed a deliberate release of variola virus. With the cessation of vaccination and vaccine production, it will become increasingly difficult for any person or group contemplating the release of variola virus to assure themselves and their colleagues of protection against smallpox. A country’s resumption of vaccination against smallpox would now be interpreted as a sign that it might be considering the use of variola virus for aggressive purposes.
Deliberate release, or the threat of it, by an individual or a group, as an act of sabotage or terrorism, is remote because access to the virus is so restricted . A document made public in December 1984 (Young & Lenarcic, 1984) describes tests made in 1964 and 1965 with aerosols containing Bacillus subtilis as a marker. The aerosols were experimentally but secretly produced in a crowded airport in Washington, DC, USA, to test the possibility that variola virus might be released in this way and thus cause “inexplicable” outbreaks of smallpox a few weeks later in the diverse places to which infected passengers travelled. The existence of such a possibility emphasizes the need for “military” as well as microbiological security in laboratories holding variola virus stocks. It is melancholy but perhaps realistic to suggest that the possibilities of biological warfare or terrorism now constitute the chief reason for holding reserves of vaccine and for maintaining epidemiological and laboratory expertise for the diagnosis and control of smallpox.
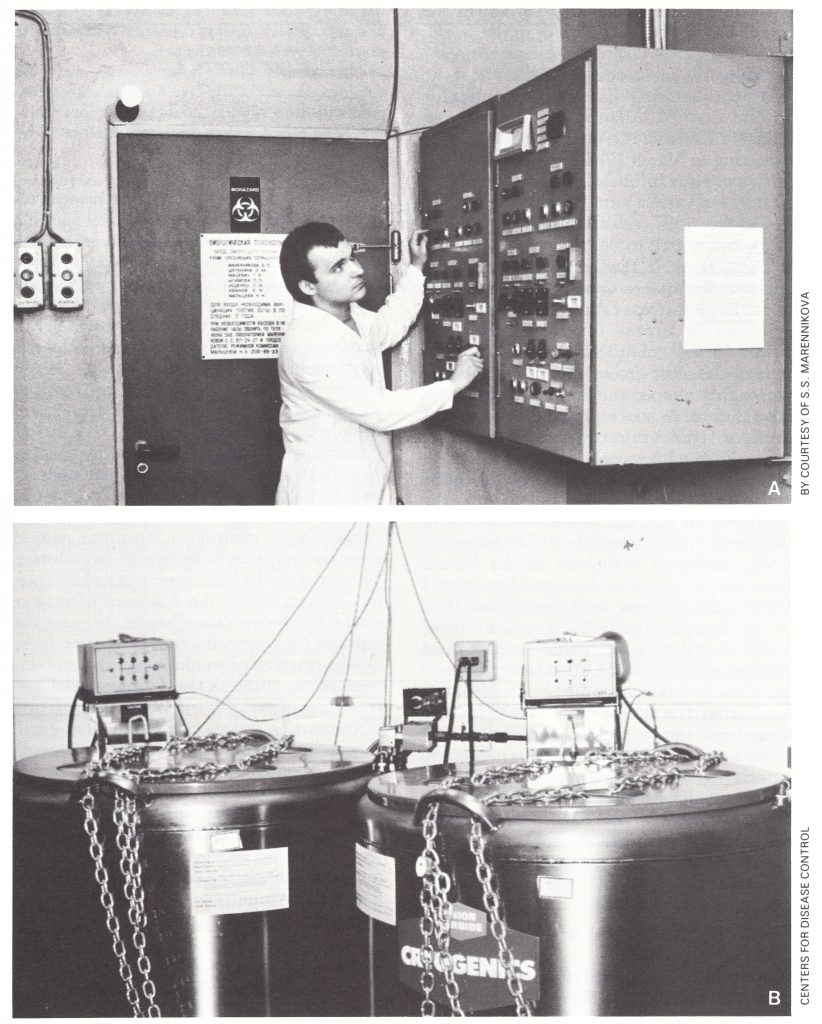
Plate 30.4. Only 2 laboratories in the world retain variola virus. A: Entrance to the maximum containment laboratory at the Moscow Research Institute for Viral Preparations, USSR. The control panel for biowaste disinfection and disposal is on the right and on the door is a list of persons authorized to enter. The door is kept locked and the key retained by the director. B: Variola virus is stored in two nitrogen vapour phase freezers in a maximum containment laboratory at the Centers for Disease Control, Atlanta, GA, USA.
REACTIVATION AND EXCRETION OF VIRUS IN HUMANS
Viruses of many viral families persist for long periods in their hosts and viruses in some families are persistently (Arenaviridae) or intermittently (Herpesviridae) released into the environment. In addition, there is now a vast experience of the reactivation of latent herpesvirus infections (especially cytomegalovirus) in immunosuppressed persons, both those suffering from lymphoreticular diseases and those in whom chemical suppression of cell-mediated immunity is employed to prevent transplant rejection. In addition, steroid therapy has immunosuppressive effects and is associated with the reactivation of latent infections. Such therapies have now been practised for many years, in many countries, but no evidence has ever come to light of the reactivation of variola or vaccinia virus in immunosuppressed persons or those on steroid therapy. This supports the view elaborated in Chapter 3 that the poxviruses in general and variola and vaccinia viruses in particular do not produce persistent infections . This hypothetical possibility can be dismissed.
VIRAL PERSISTENCE IN THE ENVIRONMENT
Although it does not cause persistent infection in man, variola virus in scabs is, for a virus, very resistant to inactivation, especially at moderate temperatures and out of sunlight. Clothing and bedding from smallpox cases, which were often heavily contaminated with virus from salivary excretions and skin lesions, were an occasional source of outbreaks of smallpox among laundry workers (England and Wales, Ministry of Health, 1928b), and virus on imported raw cotton was suspected (without good reason, according to Dixon, 1962) of causing some outbreaks in England. However, the periods for which the virus survived to cause such infections were measured in days or weeks—not years.
Interred corpses have also been suggested as a possible source of infectious virus (e.g.,Razzell, 1976), but the only evidence is anecdotal and comes from situations in which the possibility of infection by direct contact with cases of smallpox could not be excluded. The chance of viable variola virus surviving in corpses or coffins for the length of time that has elapsed since there were numerous smallpox deaths in temperate climates seems to us to be extremely remote, and the topic does not lend itself to scientific investigation.
A more bizarre possibility has been raised by Ewart (1983). Noting that bacteria can be preserved in permafrost for years (Boyd & Boyd, 1964), he suggested that active variola virus might still be preserved in interred bodies at York Factory on the shores of Hudson Bay in northern Canada. Six Indians were reported to have died of smallpox there in 1782, and it was the practice to inter the bodies of the dead in wooden coffins, which were placed in graves hacked out of the rockhard earth of the permafrost. Most of this graveyard has now been eroded by the nearby Hayes river, but Ewart’s contention is that variola virus could be preserved for long periods in some such way, somewhere in the Arctic. Whether viable variola virus could be released from a thawed corpse in such a way as to infect a susceptible person is a matter of conjecture. There is no scientific evidence on which to base a firm reply to speculations of this kind; one can merely say that the circumstances that would give rise to infection from such a source are exceedingly remote and impossible to study.
In whatever form the virus may persist in the environment, it is gradually inactivated, probably even at subzero temperatures (see Chapter 2). As the intervals since the last cases of smallpox in various parts of the world have extended from months to years and now to decades, this danger has lessened until it must now (1987) be considered to be very small indeed.
"TRANSFORMATION" AS A SOURCE OF VARIOLA VIRUS
The term “transformation” belongs to an era predating the development of microbial genetics. It now has a specific meaning : the alteration of the genome of a bacterium or eukaryotic cell by the incorporation of DNA from another source. As it used to be applied to the orthopoxviruses, the term was most frequently used to describe what was regarded as the conversion of variola virus into vaccinia virus, usually by passage through cows. It is impossible to achieve any such change when experiments are carried out under conditions that rigidly exclude the possibility of contamination (Herrlich et al ., 1963).
The “white clones” of monkeypox virus, described above, were viewed by Marennikova and her colleagues as examples of the “transformation” of monkeypox into variola virus, but the evidence presented earlier argues strongly that these isolates arose because of the contamination of monkeypox virus stocks with variola virus. Likewise, there is no likelihood that either vaccinia or cowpox virus, of which suitable strains yield a great variety of white pock mutants, could give rise to variola virus by one or a few mutational steps. The clear-cut distinctions in the DNA maps of different species of Orthopoxvirus (see Chapter 2) preclude such a possibility.

Plate 30.5. Albert Herrlich (1902-1970), a leading German virologist and public health expert who was Director of the Institute for Comparative Tropical Medicine at the University of Munich. He was principal author of major books on the poxviruses and on vaccination and carried out a definitive study which showed that variola virus could not be “transformed” into vaccinia virus by passage in laboratory animals or cattle.
GENERAL CONCLUSIONS
At this time, 10 years after the last known case of endemic smallpox, there is only one credible source from which another case of smallpox might arise—namely, the infection of a susceptible person with variola virus held in storage, either in a known location (stocks held in the WHO collaborating centres in Atlanta or Moscow) or in an unknown place. The latter could be either a stock maintained for possible use in microbiological warfare or a forgotten and possibly unlabelled specimen lying in a deep-freeze cabinet in a laboratory in which smallpox diagnosis or research was once performed. The likelihood of variola virus still surviving on scabs or associated with corpses or coffins in a form that might give rise to new cases of smallpox is now extremely remote.
Unless there were a complete breakdown of health services, so that countermeasures could not be mounted, the occurrence of an accidental case of smallpox could be readily contained, as was apparent in the Birmingham outbreak. Even microbiological warfare with variola virus should not present a significant hazard, in terms of the reestablishment of smallpox as an endemic disease. Its nature would be quickly recognized and countermeasures could be taken, calling if necessary on the personnel and materials provided by WHO as part of the “insurance policy” (see Chapter 28).





RobertTeels
August 27, 2025Goods delivery from China is reliable and fast. Our company delivers custom solutions for businesses of any capacity. We manage all logistics processes to make your operations smooth. air express shipping from china With direct shipments, we secure timely delivery of your consignments. Clients value our skilled team and reasonable rates. Choosing us means assurance in every order.
EdgarGlink
September 1, 2025В мессенджере Telegram появилась функция звёзд. Теперь люди могут помечать важные сообщения. Это помогает быстро открывать нужную информацию. купить звезды тг Функция комфортна для работы. Благодаря этому легко сохранить ключевые фразы. Такой инструмент экономит время и делает общение быстрее.
EdgarGlink
September 3, 2025В мессенджере Telegram появилась функция звезд. Теперь люди могут помечать важные чаты. Это дает возможность быстро находить нужную заметку. где купить звезды в тг Функция комфортна для работы. С её помощью легко оставить ключевые заметки. Такой инструмент бережёт ресурсы и делает общение проще.