Chapter 28:
Post-eradication operations: implementation of the recommendations of the Global Commission
INTRODUCTION
In order to convince the world community that smallpox had indeed been eradicated globally, WHO developed a comprehensive and sophisticated system for the certification of smallpox eradication, culminating in the final meeting in December 1979 of the Global Commission for the Certification of Smallpox Eradication (see Chapter 24). The Global Commission foresaw that the certification of eradication was not enough in itself-steps would have to be taken over the ensuing years to maintain public confidence in the reality of eradication and to ensure that advantage was rapidly taken of the benefits of the achievement, such as the ending of vaccination against smallpox. With this aim in view, the Commission’s final report (World Health Organization, 1980) listed 19 recommendations indicating the activities to be undertaken during the years following the declaration by the Thirty-third World Health Assembly on 8 May 1980 that smallpox had been eradicated throughout the world.
The 19 recommendations made by the Global Commission were unanimously endorsed by the World Health Assembly on the same date (resolution WHA33.4). They dealt with post-eradication policies under 8 headings: (1) smallpox vaccination policy; (2) reserve stocks of vaccine; (3) investigation of suspected cases of smallpox; (4) laboratories retaining variola virus stocks; (5) human monkeypox; (6) laboratory investigations; (7) documentation of the smallpox eradication programme; and (8) WHO Headquarters staff.
Concurrently with the declaration of the global eradication of smallpox the Global Commission was dissolved, and in 1981 a new 6-member committee—the Committee on Orthopoxvirus Infections—was established by the Director-General of WHO. Its membership was drawn from the Global Commission, whose chairman, Fenner, also became chairman of the Committee. Except in 1985, this Committee met annually in Geneva from 1981 to 1986, critically reviewed the progress made in the post-smallpox-eradication programme and advised WHO on the implementation of the Global Commission’s recommendations. The Committee was serviced and the implementation of post-eradication activities supervised by the Smallpox Eradication unit, which continued to exist, though with a reduced staff (consisting of 3, later 2, medical officers, an operations officer and support staff), all of whom had had extensive experience in the eradication programme. In the regional offices, all activities related to the post-eradication programme were transferred to the general programme of disease control and prevention or to the Expanded Programme on Immunization.
In this chapter, the post-eradication programme activities carried out between 1980 and 1986 are described, each of the 8 groups of recommendations being considered in turn. Each section is preceded by the relevant statement from the final report of the Global Commission (World Health Organization, 1980) together with the corresponding recommendations.
VACCINATION POLICY
“Smallpox vaccination of the general public. As smallpox has been eradicated, smallpox vaccination is no longer justified. Because vaccination may result in serious complications, which are occasionally fatal, no one except investigators at special risk should be vaccinated in any country including those where monkeypox cases have occurred.
“Recommendation (1). Smallpox vaccination should be discontinued in every country except for investigators at special risk.
“Smallpox vaccination certificates for international travelers. With the certification of global eradication of smallpox, no country should now require vaccination certificates from international travellers .
“Recommendation (2). International certificates of vaccination against smallpox should no longer be required of any travellers.”
Routine Vaccination Programmes
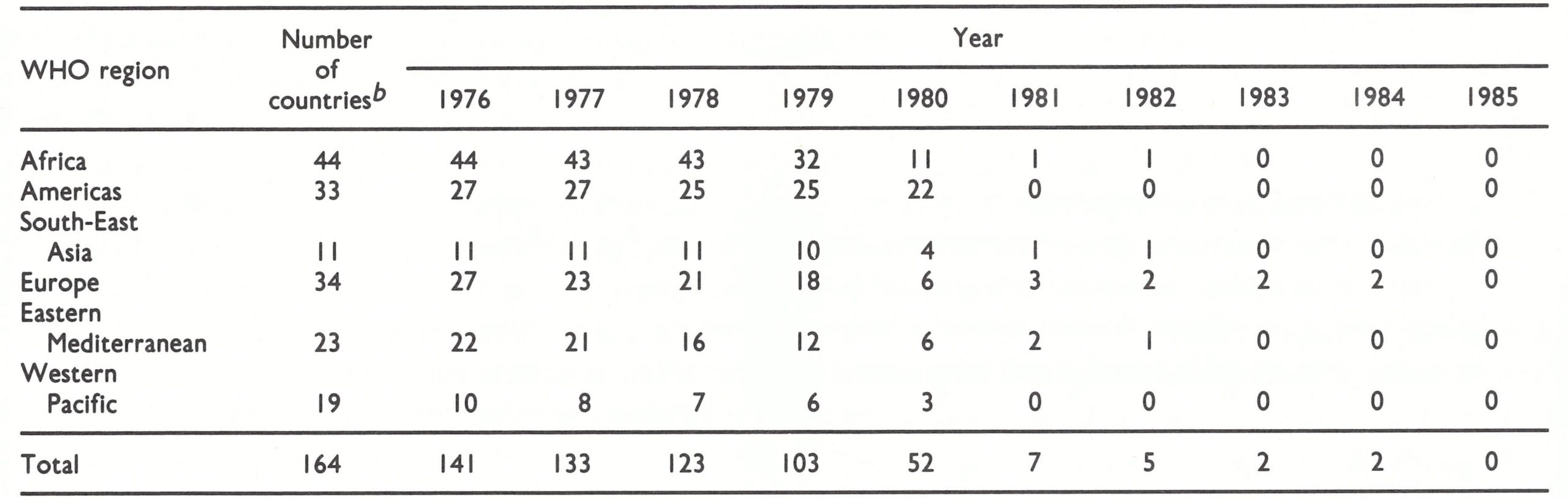
In North America and in many countries of Europe and the Western Pacific Region, routine vaccination programmes had ceased before recommendation (1) was made (Table 28.1).
The health administrations of countries in other regions, however, weighed the risks and benefits of smallpox vaccination in the light of the progress made with the global smallpox eradication programme. In previously endemic countries, international commissions for the certification of smallpox eradication had recommended that vaccination programmes should be maintained until global eradication had been certified. When this occurred, in May 1980, these programmes were soon curtailed or discontinued, particularly in countries that depended on WHO for their supplies of vaccine. Of the 156 Member States of WHO at that time, 52 were still conducting routine smallpox vaccination programmes at the end of 1980, but by the end of 1983 this number had been reduced to 2—namely, Albania, in which both primary vaccination and revaccination continued to be performed, and France, in which only revaccination was recommended. Both these countries stopped all routine vaccination in 1984.
The official discontinuation of routine vaccination programmes, however, did not always mean that vaccination had ceased altogether. In many countries, both developed and developing, appropriate instructions were not always effectively promulgated by the central health administration, and local vaccination programmes sometimes continued to be conducted. The distribution of vaccine could be readily controlled in countries in which production was carried out in national laboratories, but such control was more difficult in those in which private companies produced vaccine and sold it on request. In the USA, a few cases of severe complications (e.g .,eczema vaccinatum and progressive vaccinia) occurred when vaccinia virus was used for the treatment of herpes simplex, a form of therapy with a long history but one whose effectiveness was open to question. Eventually, agreement was reached in the USA in 1984 that, apart from making provision for the armed services, the producer would restrict the supply of smallpox vaccine to the Centers for Disease Control, for use by laboratory workers where indicated. By means of announcements in the Weekly epidemiological record and communications with governments, WHO intervened several times to advise that vaccination programmes should be stopped, stressing the risk of complications. In addition, in 1983, the Smallpox Eradication unit contacted all vaccine producers to urge that no vaccine should be made available for civilian use.
Although routine vaccination of the general public was speedily abandoned in most countries, military personnel continued to be vaccinated. As a result, incidents occurred in several countries in which a newly vaccinated recruit infected a close contact, often in the home (Laboratory Centre for Disease Control, 1981; Morbidity and mortality weekly report, 1984; Journal of the American Medical Association, 1985). Although on scientific grounds the vaccination of military personnel could have been discontinued at the same time as that of the civilian population, decisions whether or not to do so were based on political considerations, and WHO could do little to influence them. However, as time went on, an increasing number of countries discontinued the vaccination of military personnel. In order to prevent accidental vaccinial infections of civilians, the WHO Committee on Orthopoxvirus Infections recommended in 1984 that “military personnel who have been vaccinated be confined to their bases and prevented from contacting unvaccinated persons for a period of two weeks following vaccination” (WHO/SE/84.162). This recommendation was made known to all Member States . Reviewing the situation in March 1986, the Committee on Orthopoxvirus Infections recommended that “smallpox vaccination to protect military personnel against the disease be terminated” (Wkly epidem.Ree.,1986b).
Recommendation (1) exempts only “investigators at special risk” from the recomendation that vaccination should be discontinued. This group includes the following 3 categories: (1) Investigators who handle variola or monkeypox virus in a laboratory—a very small group. Everyone who enters such laboratories should undergo regular annual or triennial vaccination. (2) Investigators who work with other orthopoxviruses that are infectious for man (vaccinia and cowpox viruses). Triennial vaccination of those who handle such viruses is recommended. (3) Special surveillance teams investigating cases of human monkeypox in Zaire. At the present time, vaccination of the general population in areas in which human monkeypox virus is present is not recommended, on the grounds that the risk of monkeypox infection does not warrant either the cost of vaccination or its risks. In all, the number of persons for whom smallpox vaccination is still recommended is very small, amounting to only a few hundred persons throughout the world.
Vaccination Certificates for International Travel
At the beginning of 1980, 23 of the 155 countries for which information was available still required international travellers to hold an international certificate of vaccination against smallpox. In May 1981 the Thirty fourth World Health Assembly formally struck smallpox from the International Health Regulations and in 1983 the new edition of the International Certificates of Vaccination no longer contained any pages for smallpox vaccination (Plate 28.1).
After the declaration of the global eradication of the disease, the number of countries officially requiring international travellers to hold a certificate of vaccination against smallpox quickly decreased to only 2 (Chad and Democratic Kampuchea) in 1981 and to none in 1982. These figures are based on the report to WHO of national health administrations, but in fact during the first few years of the 1980s international travellers were sometimes asked for certificates of smallpox vaccination by health officers at ports or airports or by consulates when they applied for visas, or were informed that they would need certificates by those through whom they arranged their travel. This occurred in developed as well as developing countries, both because of poor communications between the ministries of health and other national authorities, such as ministries of foreign affairs and their representatives abroad, and because of the difficulty of ensuring that travel agencies were properly informed and gave correct advice to their clients. Following repeated interventions by WHO, the number of such incidents gradually diminished and by 1984 only a few occurred.

Plate 28.1. The 1983 version of the International Certificate of Vaccination. It no longer contains any pages for smallpox vaccination but includes a statement that it is no longer justified.
RESERVE STOCKS OF VAACINE
“Although human-to-human transmission of smallpox has been interrupted everywhere and the Global Commission believes that the likelihood of reintroduction of smallpox from laboratories or natural or animal reservoirs is negligible, it is prudent for WHO and national health authorities to be prepared for unforeseen circumstances. One measure that should be taken is to ensure that adequate reserves of potent freeze-dried vaccine are available. This vaccine should be stored at—20°C and its potency periodically checked. Seed lots of vaccinia virus for the future preparation of vaccine should be maintained, and stocks of bifurcated needles should be available.
“Recommendation (3). Sufficient freeze-dried smallpox vaccine to vaccinate 200 million people should be maintained by WHO in refrigerated depots in two countries, together with stocks of bifurcated needles.
“Recommendation (4). The stored vaccine should be periodically tested for potency.
“Recommendation (5). Seed lots of vaccinia virus suitable for the preparation of smallpox vaccine should be maintained in designated WHO collaborating centres.
“Recommendation (6). National health authorities that have vaccine stocks should be asked to inform WHO of the amount of vaccine maintained.”
WHO Reserve Stocks of Vaccine
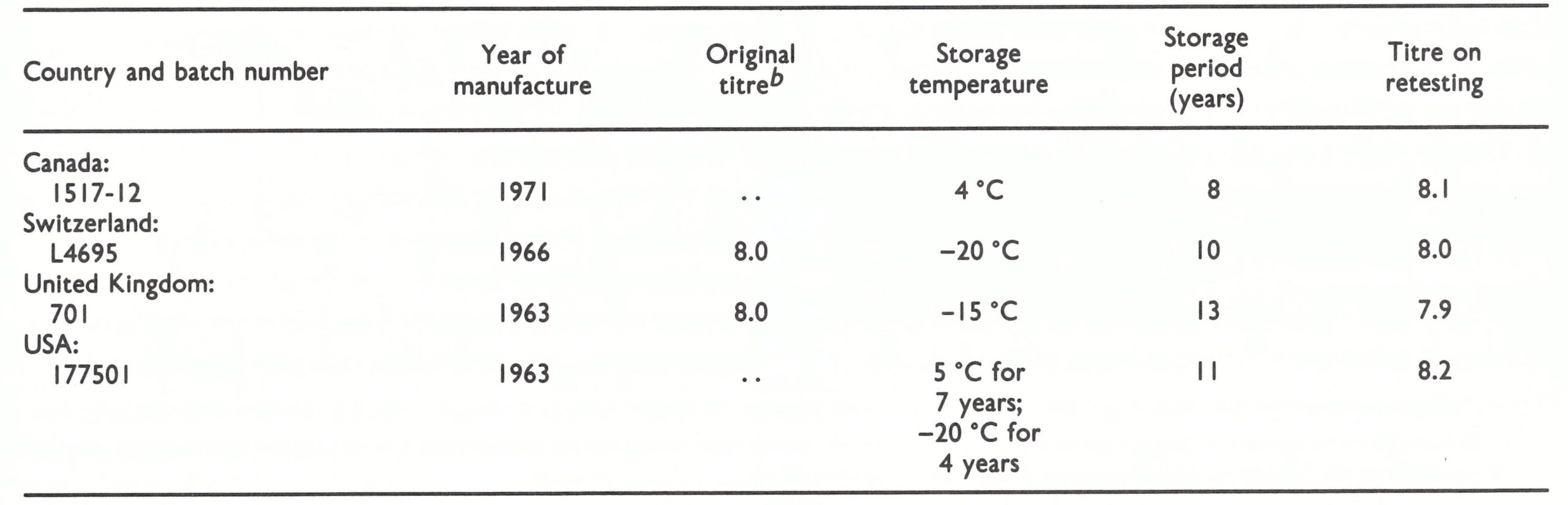
In December 1980, the Smallpox Eradication unit drew up detailed procedures for implementing recommendations (3) and (4) ; these were set out in a document entitled Management of reserve stocks of vaccine in the post smallpox eradication era (WHO/SE/80.158 Rev.1), which was approved by the WHO Committee on Orthopoxvirus Infections in March 1981. The recommended storage temperature of—20°C or lower was based on previous experience in the potency testing of batches of vaccine produced in the Connaught Laboratories, Canada ; the Lister Institute, United Kingdom; the National Institute of Public Health, Netherlands; the Swiss Serum Institute, Switzerland ; and Wyeth Laboratories, USA (Table 28.2). These batches had been stored at temperatures ranging from +4°C to -20°C over a period of 8-13 years. The potency of the great majority had remained at an adequate level, and it was assumed that storage at a temperature of -20°C would suffice to maintain a satisfactory titre for many years.
At the end of 1980, 68 million doses of vaccine were stored in Geneva and 6 million doses in New Delhi. These stocks had been donated by 9 countries—Belgium, Canada, the German Democratic Republic, India, the Islamic Republic of Iran, the Netherlands, Sweden, the USSR and the USA—India and the USSR being the major donors. Between 1981 and 1985, further donations were made by Belgium, India and the USSR, but some stocks were destroyed (see below), so that the total reserve stocks at the end of 1985 amounted to 5 034 178 vials, in 459 batches. If bifurcated needles were to be used, this amount of vaccine would be sufficient to vaccinate some 300 million persons. About 3.7 million bifurcated needles are also stored in Geneva.
These stocks can be made available for any country needing to undertake emergency containment measures, but only if a presumptive diagnosis of smallpox has been confirmed by expert clinical and laboratory investigations (see below).
Testing
Batches of vaccine will continue to be sampled periodically for as long as the WHO reserve is maintained, in accordance with the procedure laid down in document WHO/SE/80.158 Rev.1; in this way the potency of all batches can be regularly monitored so that any decline in potency can be detected without delay.
Testing is carried out by the National Institute of Public Health, Bilthoven, Netherlands, which remains the WHO International Collaborating Centre for Smallpox Vaccine. In order to ensure that the data are comparable with those obtained in previous years, the Institute has decided to continue potency testing by pock counting on the chorioallantoic membrane, rather than by the newer and somewhat more accurate technique of plaque assay.
Problems encountered
The problems encountered during the implementation of the recommendations regarding reserve stocks are described below.
Location. In accordance with recommendation (3), two depots were established in 1981, one at the Societe des Gares Frigorifiques et Ports Francs de Geneve, Switzerland, and the other in the WHO Regional Office for South East Asia, New Delhi, India. In the Geneva depot the requisite temperatures were satisfactorily maintained, but in the New Delhi depot the temperatures fluctuated from -10°C to +10°C. In 1982, the Smallpox Eradication unit arranged for a refrigeration consultant to visit the Regional Office depot in New Delhi, but it still proved impossible to keep the temperature at the proper level. In addition, the depot was flooded during heavy rains, and all the ampoules of diluent, which were stored separately from the vaccine, were submerged and had to be destroyed. In February 1984 the entire stock of vaccine in the New Delhi depot was transferred to Geneva. It was expected that, because of the high heat stability of freeze-dried smallpox vaccine, the stock would not have been affected by the temperature fluctuations, and, indeed, satisfactory results were obtained when all the batches were tested before their transfer.
The Global Commission’s recommendation that vaccine depots should be located in two countries could thus not be implemented. After the difficulties in New Delhi, attempts were made to find a second depot in Japan or the USA, but these failed because the veterinary health authorities of these countries objected to the introduction of calf skin vaccine, which might contain foot-andmouth disease virus. The Committee on Orthopoxvirus Infections therefore agreed to divide the vaccine stocks equally between the cities of Geneva and Lausanne, which are about 60 kilometres apart and connected by a motorway. Geneva has good communications with the rest of the world, and experience during the smallpox eradication programme showed that, in the event of an emergency, vaccine could be dispatched to any country in the world within 24 hours.
Potency. Although there was good evidence that vaccine could be stored indefinitely at -20°C, tests carried out in Bilthoven between 1981 and 1983 revealed that the potency of 6 batches of vaccine, from the Islamic Republic of Iran (200 000 doses of vaccine for multiple puncture), and the USA (1 .9 million doses of jet injector vaccine), had declined. No attempt was made to determine the causes of this decline ; the most probable explanation was that the ampoules were not properly sealed or that the vaccine had too high a moisture content. These batches were removed from the stock and destroyed. Otherwise all samples tested have been found to be of satisfactory potency (Table 28.3).
Table 28.3: Smallpox vaccine reserve stocks: typical results of initial and subsequent tests of batches provided by manufacturers in 7 countries, 1978-1983a
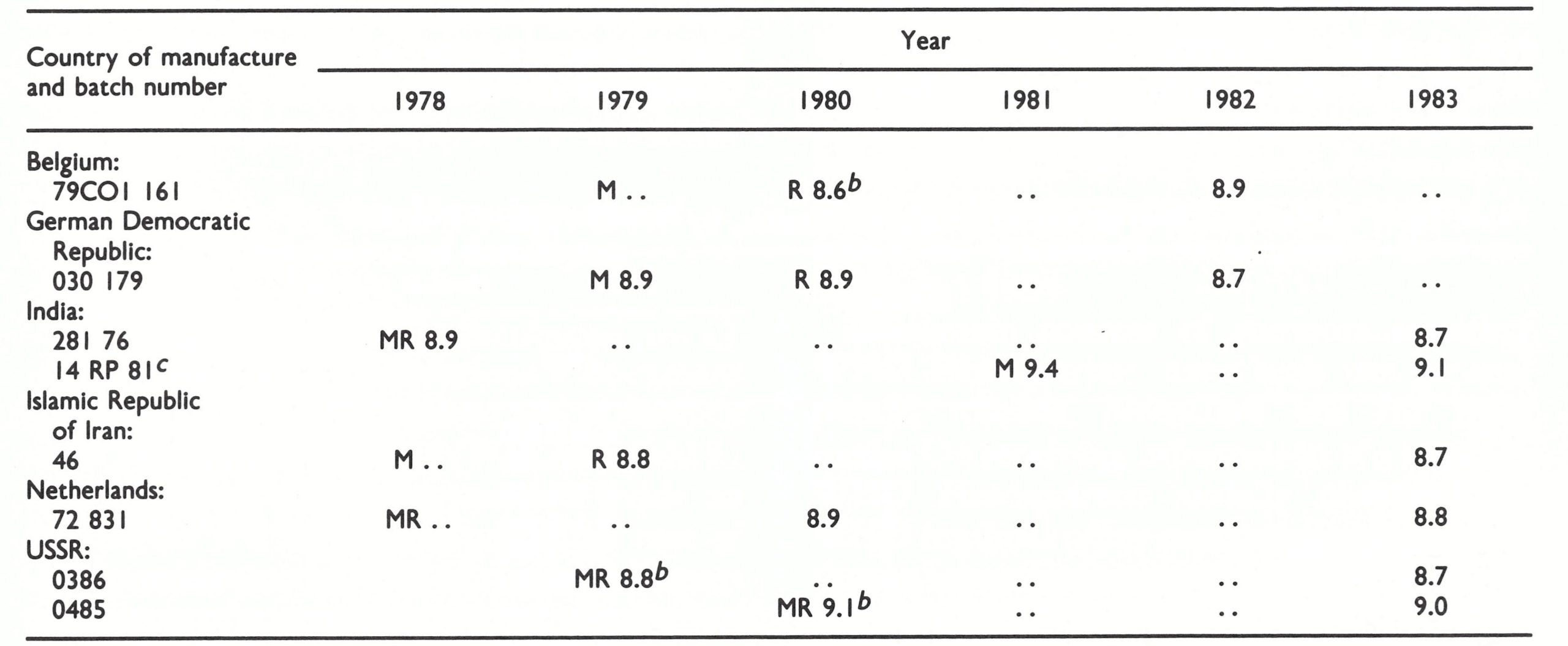 a Tested by the WHO International Collaborating Centre for Smallpox Vaccine, Bilthoven, except where indicated; titres expressed as, log, 10 pock-forming units per ml of reconstituted vaccine. M = year manufactured; R = year received; ...= data not available. Vaccine was stored at -4 °C before 1980, and at -20 °C thereafter.
a Tested by the WHO International Collaborating Centre for Smallpox Vaccine, Bilthoven, except where indicated; titres expressed as, log, 10 pock-forming units per ml of reconstituted vaccine. M = year manufactured; R = year received; ...= data not available. Vaccine was stored at -4 °C before 1980, and at -20 °C thereafter.
b Result of test carried out by manufacturer.
c Stored initially at the WHO Regional Office for South-East Asia, New Delhi, India.

Plate 28.2. The reserve of smallpox vaccine in Geneva being checked by J. F. Wickett. At -20°C it may be expected to retain its potency for many years. Nevertheless, samples are periodically tested, in accordance with a statistically designed procedure.
Long-term maintenance
During the period 1981-1985 the existence of the WHO vaccine reserve made it much easier for Member States to decide to discontinue both smallpox vaccination and vaccine production. The annual cost of maintaining the vaccine depots amounted to US$21 000 and that of testing to US$3000. Reviewing the situation in March 1986, the Committee on Orthopoxvirus Infections concluded that smallpox vaccine reserves were no longer required and recommended that the maintenance of a global reserve by WHO was no longer indicated. However, it suggested that a decision to offer to return vaccine to donor countries should not be implemented until the situation had been considered by an ad hoc committee to be convened by WHO (Wkly epidem . rec., 1986b).
National Stocks of Vaccine
In addition to the WHO reserves, 22 countries reported in 1985 that they had their own national vaccine reserves, which amounted to over 100 million doses, though only about 84 million were properly maintained (Table 28.4). The national health administrations of the countries concerned are responsible for monitoring the potency of these stocks.
Seed Lots of Vaccinia Virus
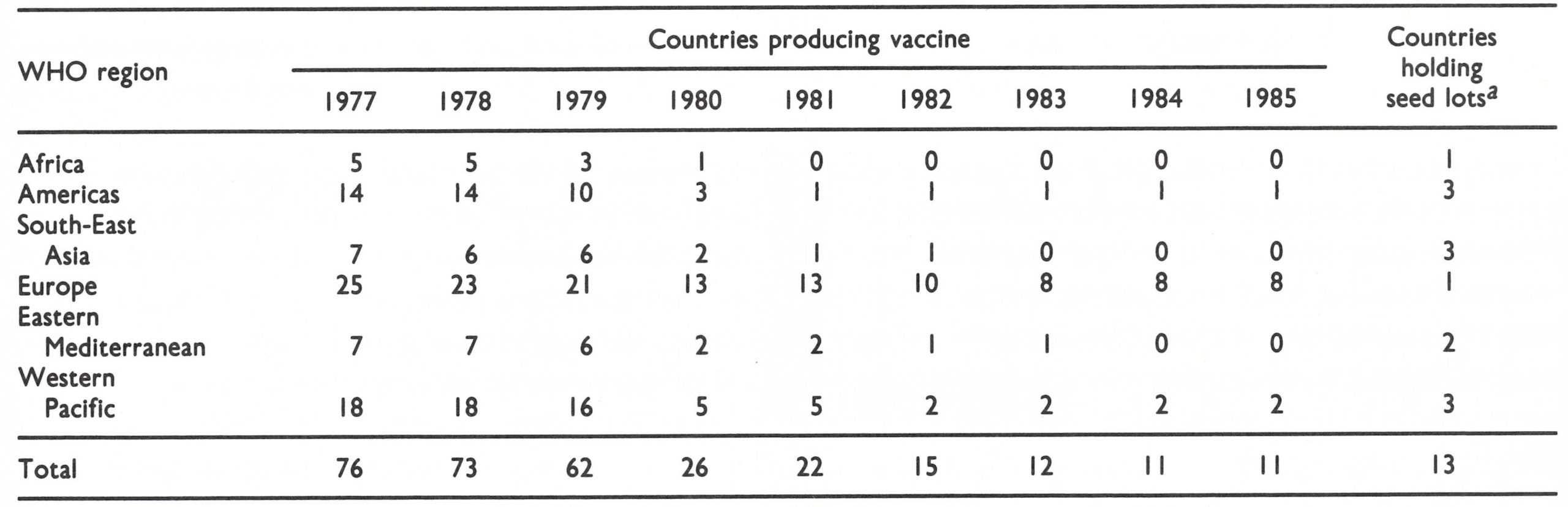
Seed lots of vaccinia virus produced and kept by the National Institute of Public Health, Bilthoven (see Chapter 11), have been distributed to 3 WHO collaborating centres-namely, the Centers for Disease Control, Atlanta, the National Health Laboratory, Paris, and the National Institute of Health, Tokyo. In addition, in 1985 13 laboratories in countries in which vaccine production had ceased held seed lot material (see Table 28.5), so that, if necessary, production could be rapidly resumed. If there were ever a national emergency, or if the WHO reserve were short of potent vaccine, the seed lots stored in the WHO collaborating centres could be made available to any country that needed to produce vaccine.
Vaccine Production Laboratories
In 1975, towards the end of the Intensified Smallpox Eradication Programme, 84 laboratories were producing freeze-dried or liquid vaccine, the latter being used for routine vaccination programmes in some countries with temperate or cold climates. A few countries discontinued production during the next few years, and, after the declaration of smallpox eradication in 1980, many more did so, often when the national health administrations discontinued routine smallpox vaccination programmes (Table 28.5).
In 1985, only 14 laboratories in 11 countries were still producing vaccine, mainly for the use of national defence forces or to stock national emergency reserves.
Table 28.4: Known national stocks of smallpox vaccine, by WHO region, 1985a
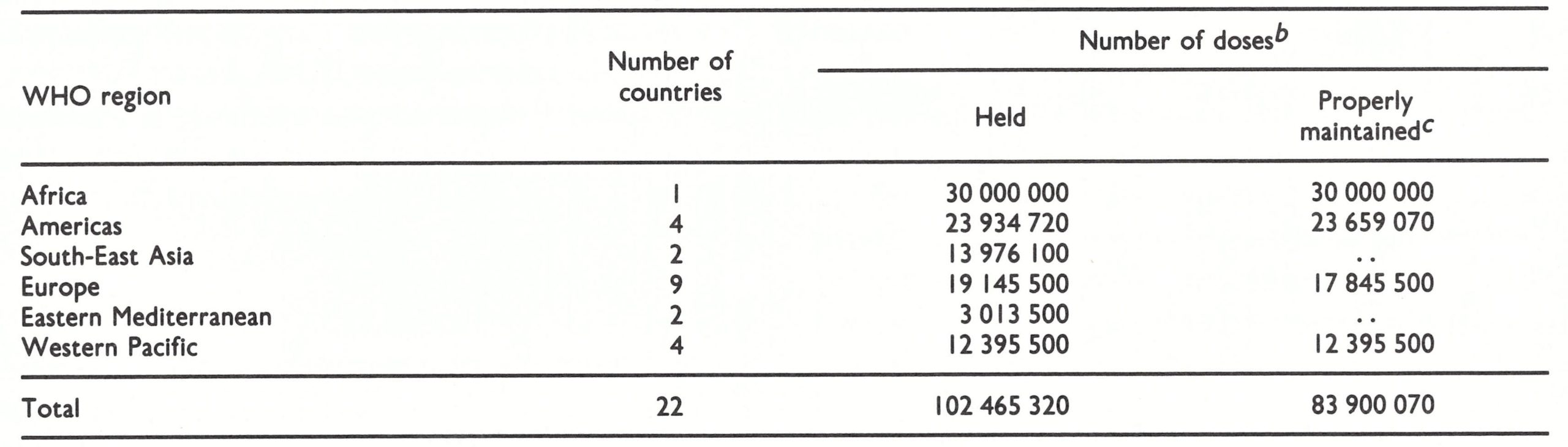 a Minimum figure; 3 of the 22 countries holding vaccine reserves did not indicate the number of doses held and are excluded from the totals shown.
a Minimum figure; 3 of the 22 countries holding vaccine reserves did not indicate the number of doses held and are excluded from the totals shown.
b Of 0. I ml, as used for multiple puncture vaccination.
c Held at a temperature of 4 °C or lower and periodically tested for potency ; . . = data not available.
INVESTIGATION OF SUSPECTED SMALLPOX CASES
“Experience in many countries indicates that reports of suspected cases of smallpox can be expected to be received from many sources for several years after the certification of global eradication. The importance of thorough investigation of these reports, if necessary with laboratory testing, is illustrated by the fact that one such report led to the recognition of human monkeypox. It is also important that public confidence in the fact of eradication should be maintained by thorough and prompt investigation of all reports and disclosure of the results to health officials throughout the world.
“Suspected smallpox cases should therefore be investigated by experienced personnel. WHO should provide an effective system to promote, coordinate, and participate in the investigation of suspected smallpox cases. The international smallpox rumour register that was established by WHO in Geneva in January 1978 should be maintained.
“The reward of US$1000 established by the Director-General in 1978 in accordance with resolution WHA31.54 should be discontinued since global eradication has now been certified.
“Recommendation (7). In order to maintain public confidence in the fact of global eradication, it is important that rumours of suspected smallpox, which can be expected to occur in many countries, should be thoroughly investigated. Information should be provided to WHO, if requested, so that it can be made available to the world community.
“Recommendation (8). WHO should maintain an effective system to coordinate and participate in the investigation of suspected smallpox cases throughout the world. The international smallpox rumour register should be maintained.”
Guidelines for the Management of Suspected Cases of Smallpox
Early in 1981, WHO distributed to all national health administrations a document entitled Management of suspected cases of smallpox in the post-eradication period (WHO/SE/80.157 Rev.1), which provides a practical guide on how to act if a report of a suspected case of smallpox is received. One of the most important pieces of advice given is that vaccination should not be carried out unless a presumptive diagnosis of smallpox has been established, based on an examination by a physician with extensive experience in the clinical diagnosis of the disease and on a laboratory report that poxvirus particles have been demonstrated by electron microscopy. This advice was included because, when the document was being prepared, certain health officials who were not convinced that eradication had been achieved had recommended that vaccination programmes should be undertaken if a suspected case of smallpox were to be reported.
The International Rumour Register
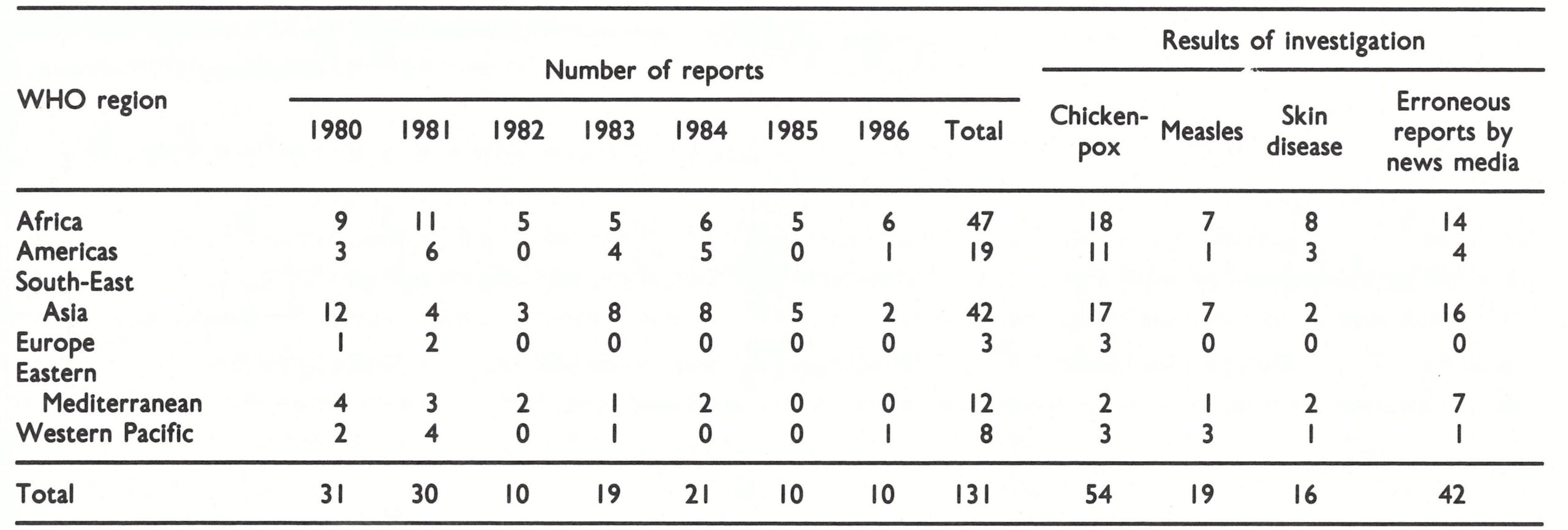
In accordance with recommendations (7) and (8), post-smallpox-eradication surveillance was initiated by the Smallpox Eradication unit in 1980 and is being continued ; 131 rumours reported to WHO between January 1980 and December 1986 were investigated with the results shown in Table 28.6. In fact, only a minority of rumours come to the attention of WHO Headquarters, the majority being dealt with at country level without being referred to WHO. In India, for example, about 2000 rumours were investigated and 79 specimens examined virologically between 1980 and 1984.
The main sources of rumours reaching WHO Headquarters have been the general public, tourists, physicians and the mass media. On receiving information of a rumour, WHO Headquarters usually contacts the WHO regional office concerned and requests that an investigation should be undertaken. If the situation is considered to be urgent, WHO Headquarters contacts the informant direct, asking for more details; at the same time it requests the government health services, as well as WHO epidemiologists in the country or at the regional office concerned, to investigate the rumour. The results of investigations of rumours deemed to be important because of the wide publicity they have received, or for other reasons, are published promptly in the Weekly epidemiological record, so that Member States are kept informed.
All these rumours have turned out to be false alarms, thus confirming that there has been no evidence of smallpox anywhere in the world since the declaration by WHO of smallpox eradication in 1980.
In checking rumours reported to WHO, use is made of the diagnostic services provided by the WHO collaborating centres at the Centers for Disease Control, Atlanta, USA, and the Moscow Research Institute for Viral Preparations, Moscow, USSR. All investigations, both by the WHO collaborating centres and by national laboratories, have failed to reveal variola virus in any sample tested.
Nature and sources of rumours reported
As was mentioned above, 131 rumours were reported to WHO Headquarters between January 1980 and December 1986 (Table 28.6). About half the total number of these reports were received between 1980 and 1982; by 1985-1986, only 10 were being reported annually. Experience in India, where most rumours were investigated locally without being referred to WHO, was comparable, the yearly totals of specimens investigated by the smallpox reference laboratory of the National Institute of Communicable Diseases being 30, 16, 8, 10 and 15, respectively, over the period 1980-1984.
Of the 131 rumours reported to Headquarters, about 70% were investigated and the results made known to WHO within 2 months, but delays of up to 6-9 months occurred in some cases. The principal diseases misdiagnosed as smallpox were chickenpox and measles, which had also been the main causes of misdiagnosis before smallpox was eradicated (see Chapter 1). A significant proportion of the rumours arose from inaccurate reporting or recording. Even cholera was sometimes reported as smallpox in the mass media. Human monkeypox was occasionally misdiagnosed as smallpox and reported as such, but these cases have been excluded from the register since they occurred in Zaire, in which the disease has been under special surveillance since 1982 (see below).
Examples of the kinds of rumours reported
Rumours arose from various sources and a wide variety of actions were taken to investigate them . A few examples are given below; others have been described by Khodakevich & Arita (1985).
Rumours circulated by the mass media. The national and international news media were a common source of rumours and occasionally reported outbreaks of smallpox in areas in which epidemics of an infectious disease had occurred and caused several deaths. Expecting to find an increased incidence of infectious diseases in refugee camps and regions affected by famine, reporters sometimes alleged the occurrence of smallpox despite the lack of any supporting evidence. For example, Newsweek, on 26 November 1984, carried an article describing the famine situation in Ethiopia, in which it stated:”. . . Starvation brought other ailments with it, including influenza, measles, tuberculosis, diarrhea, smallpox, typhus and kwashiorkor When WHO drew the attention of the editor of the magazine and of health staff in Ethiopia to the erroneous nature of this report, it transpired that there had been cases of measles, but not of smallpox.
Rumours related to variolation. On 18 November 1983, WHO was informed by the Kenyan health services that a suspected case of smallpox had occurred in Kenya in an adult male from Nandi Hill Estate in the Rift Valley Province. The patient was hospitalized on 8 November and died on 15 November, 3 days after developing a papular rash, mainly on the face and limbs. Scab specimens were taken and sent immediately from Nairobi to the WHO collaborating centre at the Centers for Disease Control, Atlanta.
It was noted that the patient, whose vaccination status was not known, had been a traditional healer, perhaps a former variolator who might have inoculated himself with old material still in his possession, but that his 5- year-old son had had chickenpox shortly before the onset of the illness . On 21 December, the collaborating centre in Atlanta informed WHO that herpesvirus particles had been seen in the specimens by electron microscopy, and this information was immediately relayed to the Kenyan health authorities. Virus cultivation on the chorioallantoic membrane gave negative results for smallpox . The prompt reporting and collection of specimens by the Kenyan health services, and the equally prompt laboratory investigation by the WHO collaborating centre, led to the correct diagnosis of this case within a few days of its occurrence.
Scabs on old corpses. Early in 1985 some student archaeologists were engaged in excavating the vaults of a church near Spitalfields, London. One coffin was found to contain the well-preserved remains of a person who had died of smallpox in about 1840 . As a precaution, the United Kingdom Health and Safety Executive closed the site and vaccinated the 3 student archaeologists concerned, and “scab” material from the corpse was sent to the WHO collaborating centre in Atlanta . Exhaustive electron microscopic examination there revealed one object that might have been a poxvirus particle but all attempts at cultivation were unsuccessful . Later, Fornaciari & Marchetti (1986) published photographs of objects that they considered to be poxvirus particles on the basis of electron microscopy of material taken from a vesiculopustular eruption on the mummy of a 2-yearold boy who died in Naples in the 16th century. Exhaustive testing in the WHO collaborating centres failed to reveal any infectious virus in samples of this material. Bodies buried in Attic permafrost, referred to in Chapter 30, might pose a greater risk, although very few smallpox victims would have been interred in such places.
Efficacy of the international rumour register
The international rumour register constituted the ultimate level of reference in a system operating primarily at the country or WHO regional office level for the investigation of rumours of smallpox, although it often received rumours direct from a source rather than through the network. This continuing surveillance failed to find any evidence of the recurrence of smallpox anywhere in the world. Had there not been such surveillance by WHO, backed up by special investigations at the WHO collaborating centres, it is highly likely that some rumours would have had significant repercussions, resulting in damage to public confidence in the WHO declaration, and even the initiation of a mass vaccination programme against smallpox. In the event, vaccination was never needed nor was it ever recommended, since no case occurred which satisfied the requirements laid down by the guide to the management of suspected cases of smallpox. The confidence thus built up undoubtedly assisted in ensuring the universal discontinuation of routine smallpox vaccination.
LABORATORIES RETAINING VARIOLA VIRUS STOCKS
“A committee of experts meeting in February 1979 advised the Global Commission that it was necessary for scientific reasons to preserve stocks of variola virus in a few laboratories, but that the position should be reviewed in 1982. In view of the potential danger of reintroduction of smallpox from variola virus stocks held in laboratories, no more than four WHO collaborating centres should be approved as suitable for the storage of and work with variola virus in accordance with WHO safety standards. These WHO collaborating centres should report annually to WHO and their containment facilities should be periodically inspected to ensure that storage is secure and that safe operating conditions are maintained. All other laboratories should be asked to destroy any stocks of variola virus that they hold, or to transfer them to an approved WHO collaborating centre.
“Recommendation (9). No more than four WHO collaborating centres should be approved as suitable to hold and handle stocks of variola virus. A collaborating centre would be approved only if it had adequate containment facilities. Each such centre should report relevant information on its safety measures annually to WHO and be inspected periodically by WHO.
“Recommendation (10). Other laboratories should be asked to destroy any stocks of variola virus that they hold, or transfer them to an approved WHO collaborating centre.”
Reduction in Number
As early as 1976 the Smallpox Eradication unit began trying to reduce the number of laboratories holding stocks of variola virus (see Chapter 30). This was successful in that the number of such laboratories fell from 75 in 1975 to 7 at the time of the final meeting of the Global Commission in December 1979 (see Chapter 30, Tables 30.6 and 30.7).
A laboratory-associated outbreak of smallpox in Birmingham, England, in 1978 and the declaration of smallpox eradication by the Thirty-third World Health Assembly in 1980 provided a strong incentive for laboratories not actively working with variola virus to destroy their stocks or transfer them to one of the WHO collaborating centres that was equipped with a high-security containment laboratory. By the end of 1981, only 4 laboratories still retained variola virus stocks : one each in South Africa, the United Kingdom, the USSR and the USA. Except for the South African laboratory, all were WHO collaborating centres for the diagnosis of, or research on, orthopoxvirus infections.
Between 1979 and 1982 Dr Keith Dumbell, a member of the Committee on Orthopoxvirus Infections, was engaged in research on variola virus, including the cloning of its DNA, in the biocontainment laboratory at the Centre for Applied Microbiology and Research, Porton Down, England. With the completion of this work in September 1982, all the variola virus stocks held at Porton Down were transferred to the Centers for Disease Control, Atlanta.

Plate 28.3. The first stage in the transfer of stocks of variola virus from the National Institute of Public Health, Bilthoven, Netherlands, to the Centers for Disease Control, Atlanta, GA, USA, on 2 December 1981, with appropriate safeguards.
In South Africa, the National Institute for Virology in Sandringham retained locally isolated strains of variola virus in a highsecurity biocontainment laboratory, although they had not been used for many years . Because of South Africa’s political isolation, the health authorities were afraid that, if the virus were destroyed, they would have difficulties in diagnosing variola virus should any unexpected circumstances occur . However, when cloned preparations of fragments of variola virus DNA (which cannot replicate and are completely safe to work with) were made available to the National Institute for Virology by Dr Dumbell, their anxieties were relieved and the South African government destroyed all virus stocks in December 1983, in the presence of Dr Dumbell (Plate 28.4).
Since the beginning of 1984, stocks of variola virus have been retained only in the WHO collaborating centres in the Centers for Disease Control, Atlanta, USA, and the Moscow Research Institute for Viral Preparations, USSR, each of which possesses a highsecurity biocontainment laboratory. During 1983, the remodelling of these laboratories was completed, and both were approved by WHO inspection teams for work with variola virus. The retention of these facilities and their associated personnel has been costly, but the governments of the USSR and the USA have continued to provide the necessary support because of their value in examining material from cases of human monkeypox and from suspected cases of smallpox referred to them through the international rumour register.
The stocks held at the Centers for Disease Control include those transferred from the National Institute of Health, Japan; the National Institute of Public Health, Netherlands; the Centre for Applied Microbiology and Research, United Kingdom ; the American Type Culture Collection, New York; and the United States Army Medical Research Institute for Infectious Diseases, Frederick, Maryland, USA. In addition to variola virus stocks, the Moscow laboratory holds “whitepox” viruses isolated from animal specimens and from monkeypox virus stocks (see Chapter 30). These virus stocks are regarded as just as dangerous as variola virus and are kept in the containment laboratory under the same security conditions as that virus. The variola virus stocks in Atlanta and Moscow contain strains of variola major and variola minor viruses from most areas in which smallpox has occurred since 1967.
In March 1986 the Committee on Orthopoxvirus Infections reviewed its policy on the retention of stocks of variola virus in the light of the existence of DNA from 5 strains of variola virus cloned in Escherichia coli and the current situation in relation to the use of the virus in research and diagnosis. The meeting was attended by senior public health officials from the two countries in which variola virus stocks are currently held-namely, the USSR and the USA. The Committee advised WHO that the cloned DNA would be satisfactory both for assisting diagnosis in any situation in which that might prove necessary and for archival purposes . Since the Committee considered that research work requiring the use of viable variola virus was no longer justified, it recommended that remaining stocks of the virus should be destroyed, and that this recommendation should be widely publicized but not implemented until approval had been given by an ad hoc committee of experts, which it suggested should be convened by WHO (Wkly epidem . rec., 1986b).
Inspection
Since 1978, WHO inspection teams consisting of epidemiologists, virologists and biosafety experts have periodically visited all the laboratories retaining variola virus, in order to determine whether each of them satisfied the relevant WHO requirements . Inspections have been made annually or every second or third year (Table 28.7), a special check-list being used to cover all safety aspects. These inspections have revealed that problems sometimes occur in the efficient operation of biocontainment laboratories, even though the personnel concerned are highly trained.

Plate 28.4. Destruction of variola virus stocks at the National Institute for Virology, Sandringham, South Africa, on 9 December 1983. The operation was carried out by the Minister of Health and Welfare of South Africa and witnessed by Dr K. R. Dumbell (left), a member of the Committee on Orthopoxvirus Infections and Dr Bob Swanepoel (right), Senior Virologist in charge of the P4 Laboratory.
Table 28.7: Inspections of laboratories holding stocks of variola virus, 1978-1986
 a Stocks destroyed in 1983.
a Stocks destroyed in 1983.
b Stocks transferred to Centers for Disease Control in December 1981.
c Stocks destroyed in 1983.
d Stocks transferred to Centre for Applied Microbiology and Research in September 1978.
e Stocks transferred to Centers for Disease Control in September 1982.
f Stocks destroyed in September 1978.
g Stocks transferred to Centers for Disease Control in April 1980.
HUMAN MONKEYPOX
“Human monkeypox is a rare zoonosis that was not recognized until smallpox was eliminated from the area where it occurs. Clinically it resembles smallpox. Human cases can be expected to appear where the ecological conditions are appropriate and perhaps to show some increase as smallpox vaccination ceases and immunity wanes. Because it is caused by a poxvirus distinct from variola virus and has a limited capacity to spread between humans, monkeypox virus does not constitute a threat to the permanence of smallpox eradication. However, it is important that close surveillance of human cases should continue and that further investigation should be made into the natural history of the disease.
“Recommendation (11). In collaboration with country health services, WHO should organize and assist a special surveillance programme on human monkeypox, its epidemiology, and its ecology in areas where it is known to have occurred. The programme should continue until 1985, when a further assessment of the situation should be made”.
The demonstration that monkeypox virus caused sporadic infections in man was an important scientific discovery directly attributable to the smallpox eradication programme. Consideration of the ecology of the disease indicated that it was not a new, but a newly discovered, one. The implementation of recommendation (11) of the Global Commission constituted the greater part of the post-smallpox-eradication programme. The organization of surveillance is described below and the results of these studies are reported in Chapter 29. Activities relating to human monkeypox were planned by Arita and Jezek, and implemented by the joint efforts of WHO, national staff in western Africa and Zaire, and the WHO collaborating centres.
African Viral Haemorrhagic Fevers
In addition to smallpox and yellow fever, which were for centuries the commonest viral diseases with haemorrhagic manifestations, several other viral haemorrhagic fevers have been recognized in recent decades (McCormick & Johnson, 1984). All are presumed to be contracted by man from an animal source. Three of them, Ebola virus disease, Lassa fever and Crimean-Congo haemorrhagic fever, occur in central and western Africa. The natural history of Ebola virus, which caused a major outbreak in a hospital in Zaire in 1976, is unknown, and there are many gaps in our knowledge of the ecology of the other two diseases. In the studies planned in Zaire, surveillance for haemorrhagic fever was added to monkeypox surveillance, since the geographical areas in which these diseases have been identified overlap and the methods of investigation are similar. In this way the best use could be made of the sparse resources available in central and western Africa.
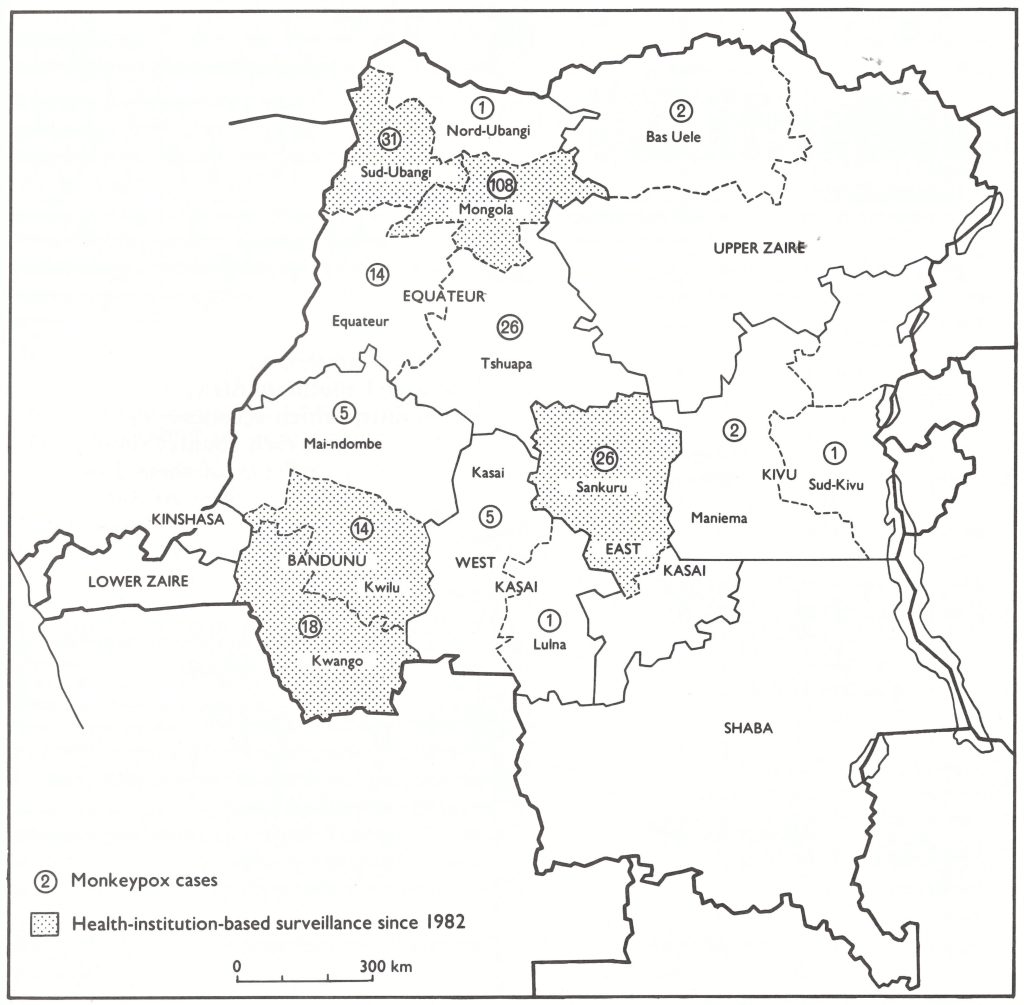
Fig. 28.1. Zaire: subregions where intensive health-institution-based surveillance has been carried out since 1982, and numbers of cases of human monkeypox by subregion, 1970-1984.
Epidemiological Surveillance in Zaire
Work in Zaire from 1980 onwards built on the surveillance system established there in the late 1970s (see Chapter 29).
Seminar in Brazzaville, May 1980
A seminar on the surveillance of monkeypox and viral haemorrhagic fevers was held in the WHO Regional Office for Africa, Brazzaville, Congo, in May 1980. Its objective was to devise ways of further strengthening the surveillance of monkeypox and viral haemorrhagic fevers in areas of central and western Africa in which these zoonoses occurred. WHO staff from Geneva and the regional office and delegates from 10 countries in central and western Africa and 2 countries in eastern and southern Africa participated in the seminar, which recommended that the health services of each country should develop special surveillance of these diseases. A document entitled Procedures for the surveillance and management of monkeypox and viral haemorrhagicfevers (CDS/80.1) was promptly prepared by WHO and widely distributed, but only 3 cases of monkeypox were reported in 1980 and 8 in 1981.
Health-institution-based surveillance in Zaire
In the summer of 1980, Arita visited Kinshasa, Zaire, to discuss with the health authorities and the staff of the monkeypox programme how to strengthen surveillance activities. A review of the previous 3 years of surveillance in Zaire revealed 2 important findings: (1) most of the cases reported were patients who had visited hospitals or dispensaries seeking medical treatment; and (2) an earlier recommendation that mobile surveillance teams should visit all health stations every 3 months had never been implemented because of limited resources. Furthermore, the mobile teams were frequently used in the control of cholera outbreaks. In the circumstances, health-institution-based surveillance, which was based on the collaboration of hospital and dispensary staff, appeared to be the most promising method of obtaining more information about human monkeypox. The system was concentrated in 4 subregions of 3 regions in northern Zaire, which had extensive areas of tropical rain forest and in which a relatively high incidence of the disease had been observed in 1978 and 1979.
Health-Institution-Based Surveillance
The idea of using health-institution-based surveillance for human monkeypox in Zaire stemmed from experience in western Africa with Lassa fever. This is a haemorrhagic fever caused by an arenavirus, which was first recognized in a hospital outbreak in Lassa,Nigeria, in January 1969 . Outbreaks subsequently occurred in hospitals in several other parts of western Africa, and in the early 1970s it was thought to be a rare zoonosis, which was associated with a case-fatality rate of about 50% when outbreaks did occur. In 1977 a study was commenced in the Kenema District, Sierra Leone, under the direction of Dr Joseph B. McCormick, of the United States Center for Disease Control, to assess the prevalence and importance of Lassa fever. A surveillance system based on the hospital reporting of febrile disease was established, together with a small diagnostic laboratory. Local personnel were trained to take specimens from patients and to investigate any suspected cases of Lassa fever. The system was successful and showed that the disease was a common cause of hospital admission for both adults and children, with several hundred cases occurring annually in the area. The severity of cases varied greatly, with a case-fatality rate of less than 5% among subjects sick enough to report to hospital. The overall casefatality rate was even lower, since other studies revealed that 6 % of a population surveyed in Kenema District had complement-fixing antibodies against Lassa virus while only 0.2% had recognized disease (Fraser et al., 1974).
This experience suggested that health-institution-based surveillance might be a practical method for monitoring the prevalence of a disease in areas in which the infrastructure for public health and preventive medicine was rudimentary.
Between December 1980 and November 1981, 4 seminars on monkeypox and viral haemorrhagic fever surveillance were organized by Jezek, in collaboration with Dr Kalisa Ruti, of the Ministry of Health of Zaire, in which medical staff from 154 health institutions participated. The seminars, which brought together all senior staff of health units working within a radius of 250-500 kilometres, were held in Lisala for Mongola subregion (December 1980), in Gemena for Sud-Ubangi subregion (February 1981), in Lodja for Sankuru subregion (June 1981) and in Kikwit for Kwango and Kwilu subregions (November 1981) (see Fig. 28.1). It was the first time in central and western Africa that seminars had been devoted specifically to these newly discovered diseases, with the participation of senior government officials and technical experts. Monkeypox recognition cards, sample collection kits and the document Instructions for health-institution-based surveillance of monkeypox and viral haemorrhagic fevers (CDS/80.2) were distributed to participants. It was announced that a reward of 500 zaires (US$90) was to be offered to any person, including health staff, who reported a case of human monkeypox.
Following each seminar, health-institution-based surveillance for human monkeypox and viral haemorrhagic fevers, supported by WHO funds, was developed in each subregion (Fig. 28.1), and by the end of 1981 was functioning effectively in all of them. The selected areas together represented about 15 % of the land area of Zaire and contained about 5 million inhabitants—18% of the country’s population-among whom 89% of the human monkeypox cases recorded in Zaire up to that time had been detected. Sporadic cases of Ebola haemorrhagic fever were also known to occur in some of the selected areas. A total of 154 health establishments participated in surveillance activities, including 55 government and missionary hospitals, 62 dispensaries and 37 other health units (health centres and maternal and child health clinics). They were responsible for the discovery, identification and notification of suspected cases of monkeypox or viral haemorrhagic fever as well as the proper collection and dispatch of specimens for laboratory investigation . Their activities were supported by 4 mobile surveillance teams, which provided technical advice and material support. This well-designed and well-supervised surveillance system based on peripheral health units and mobile surveillance teams was supplemented by posting special surveillance officers in 4 areas of epidemiological priority to carry out village-based surveillance and assist in ecological surveys.
The striking increase in the number of monkeypox cases reported in Zaire in 1982 (39 cases) and 1983 (84 cases), as compared with previous years (see Chapter 29, Table 29.3), was due principally to the strengthened health-institution-based surveillance and intensified field activities, since 90% of the cases reported in Zaire in 1982-1983 were detected in areas of strengthened surveillance; and health-institution-based surveillance itself accounted for about 60% of the reported cases. The description of the clinical features and epidemiology of human monkeypox given in Chapter 29 is based on the results obtained by this intensive surveillance in Zaire, supported by laboratory studies in the WHO collaborating centres in Atlanta and Moscow.
Serological survey of unvaccinated subjects
In order to estimate the prevalence of human monkeypox in central and western Africa, serological surveys were conducted on unvaccinated subjects living in tropical rain forest areas of the Congo, Cote d’Ivoire, Sierra Leone and Zaire (see Chapter 29, Table 29.10). These surveys revealed that at least 0.7% of the persons concerned, who lived in areas in which monkeypox could be expected to occur, had specific antibody to it. Follow-up examinations of persons with positive serological findings indicated that over half of them had no history of a vesiculopustular disease or any facial pockmarks. It was concluded that at least some of them had suffered from a subclinical infection with monkeypox virus (see Chapter 29).
Studies on the ecology of monkeypox virus In 1984-1986, Dr Lev Khodakevich and his collaborators carried out surveys in the Central African Republic and Zaire in order to try to determine the reservoir host of monkeypox virus. These were successful in incriminating squirrels as a significant wild animal host of monkeypox virus in some areas (see Chapter 29).
Review of Monkeypox, March 1986
In its review of the situation in March 1986, the Committee on Orthopoxvirus Infections noted that excellent progress had been made in understanding the epidemiology of human monkeypox and the ecology of monkeypox virus since its third meeting, in 1984. The results of these studies, described in detail in Chapter 29, indicated that human monkeypox was a rare zoonosis in which human-to-human infection sometimes occurred. The Committee concluded that it was now clear that the disease was not a serious public health problem and recommended that WHO involvement in monkeypox surveillance and research should be phased out (Wkly epidem . rec .,1986b).
LABORATORY INVESTIGATIONS
“There are still some important unsolved virological problems that are relevant to smallpox eradication, especially in relation to the “whitepox” viruses. The solution of these problems and preparedness for unexpected problems that might arise in relation to smallpox or other poxvirus diseases of man call for the maintenance of suitable virological expertise.
“Besides encouraging scientists in various nations to continue research on orthopoxviruses, WHO has responsibility for the regular testing of the potency of the WHO vaccine reserves and for the provision of laboratory diagnostic facilities for suspected smallpox cases. It can best discharge this responsibility by continuing the system of WHO collaborating centres. If competent research workers from laboratories not approved by WHO for work with variola and whitepox viruses wish to conduct experiments with these viruses, facilities should, if possible, be provided by a suitable WHO collaborating centre. These experiments must be approved by the appropriate WHO committee.
“Recommendation (12). WHO should continue to encourage and coordinate research on orthopoxviruses.
“Recommendation (13). WHO should maintain the system of WHO collaborating centres for carrying out diagnostic work and research on orthopoxviruses.
“Recommendation (14). Research workers who do not work in a WHO collaborating centre and who wish to carry out experiments with variola or whitepox virus that are approved by the appropriate WHO committee should be offered the use of the special facilities in a WHO collaborating centre.
“Recommendation (15). Research on poxviruses other than variola or whitepox viruses should not be performed under circumstances where there is any possibility of cross-contamination with these two agents.”
Laboratory Diagnosis of Suspected Smallpox and Monkeypox
Since 1980 the two WHO collaborating centres in Atlanta (Dr James Nakano, director) and Moscow (Dr Svetlana Marennikova, director) have continued the diagnostic testing of specimens collected from suspected cases of smallpox throughout the world, employing for this purpose laboratory workers with extensive experience obtained during the Intensified Smallpox Eradication Programme. In addition to examining material from some of the cases reported to the international rumour register, the centres examined specimens collected from suspected cases of human monkeypox and from other poxvirus infections (tanapox and molluscum contagiosum) in western and central Africa. Between 1980 and 1984, 16 650 specimens were tested (Table 28.8), the majority having been collected during serological surveys for human monkeypox. Requests for viral isolation came mainly from countries in which human monkeypox cases had occurred or were suspected, mostly as part of the special surveillance for human monkeypox described earlier in this chapter and in greater detail in Chapter 29. In addition, 193 requests were received from 28 other countries in connection with suspected cases of smallpox. In no case was variola virus found.
Table 28.8: Numbers of specimens (lesion material and sera) examined for evidence of orthopoxvirus infection by the WHO collaborating centers in Atlanta and Moscow, by WHO region, 1980-1984
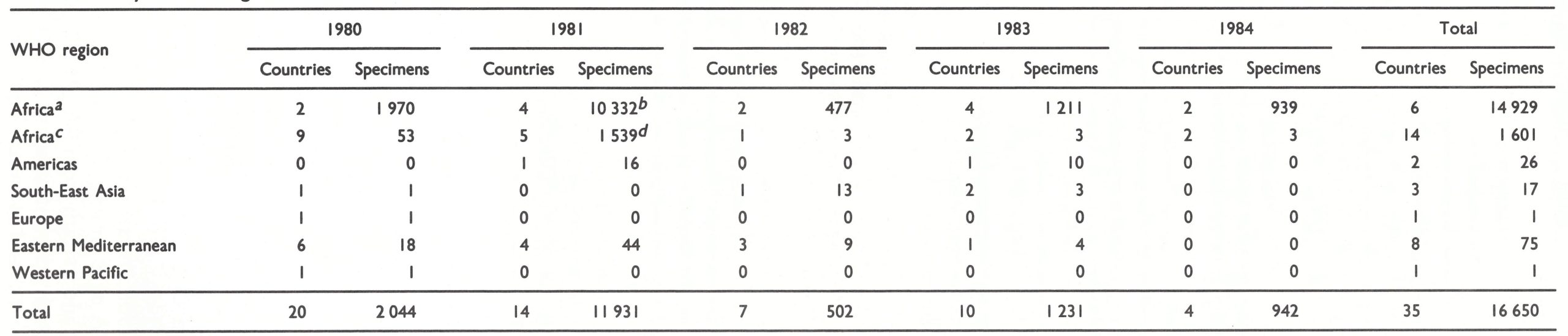 a Countries in which monkeypox has been found (Cameroon, Central African Republic, Cote d’lvoire, Nigeria, Sierra leone, Zaire).
a Countries in which monkeypox has been found (Cameroon, Central African Republic, Cote d’lvoire, Nigeria, Sierra leone, Zaire).
b Includes 8867 specimens of serum from Cote d’lvoire, Sierra Leone and Zaire collected in serological surveys (see Chapter 29, Table 29.10).
c Other countries (i.e., those not mentioned in footnotea).
c Includes 1433 specimens of serum from the Congo collected in serological surbeys (see Chapter 29, Table 29.10).
Continuing Research on Orthopoxviruses
Research problems of importance to WHO in the post-smallpox-eradication era fell into 3 categories: (1) analysis of the DNA of variola virus and other orthopoxviruses; (2) development of sensitive tests to detect antibody specific to monkeypox virus in human and animal sera; and (3) ecological and epidemiological research on the natural history of monkeypox.
Analysis of orthopoxvirus DNA
In June 1979, in view of the fact that the certification of global eradication was imminent, WHO organized a meeting on orthopoxvirus research, which was co-sponsored by the Centers for Disease Control and held in Atlanta (SME/79.9). The objective was to define fields of research relevant to the post smallpox-eradication era. At that time there was still considerable controversy as to the significance of the “whitepox” viruses that had been isolated in laboratories in the Netherlands (Bilthoven), and the USSR (Moscow). Following discussions at the meeting, collaborative work with personnel from the Bilthoven and Moscow laboratories was carried out by Dr Dumbell at the Centre for Applied Microbiology and Research, Porton Down, and by Dr Joseph Esposito at the Centers for Disease Control. These studies showed that all strains of “whitepox” virus were identical with variola virus and probably originated as laboratory contaminants (see Chapter 30).
Dr Dumbell, Dr Esposito and their colleagues continued to carry out studies on the DNA of various strains of variola and monkeypox viruses, thereby considerably increasing our understanding of these viruses; the results of these studies are summarized in Chapters 2, 29 and 30. Dr Esposito also produced, for the first time, restriction endonuclease maps of the DNA of camelpox, taterapox and raccoonpox viruses, all of which are species of Orthopoxvirus (Esposito & Knight, 1985).
Both Dr Dumbell and Dr Esposito cloned variola virus DNA in Escherichia coli, rendering it safe to handle in any laboratory. By March 1986, libraries of cloned fragments of the DNA of 2 strains of variola major virus (Harvey and a strain from Bangladesh) and of 3 strains of variola minor virus (2 strains of alastrim virus and a strain from Somalia) were available. At that time, the Committee on Orthopoxvirus Infections suggested that a western African strain should also be cloned, so as to provide a reasonable range of strains of variola virus DNA for archival purposes and for scientific research. It was because this material was or would be available that the Committee recommended the destruction of the remaining stocks of viable variola virus (see below).
Development of tests for antibody specific to monkeypox virus
The genus Orthopoxvirus includes 9 known species, of which 3 (vaccinia, monkeypox and taterapox viruses) or possibly 4 (with Uasin Gishu disease virus, an orthopoxvirus that affects horses in Kenya) occur in western and central Africa. All species of Orthopoxvirus show extensive serological cross-reactivity. Methods existed for making presumptive species-specific diagnoses of infections with monkeypox, variola and vaccinia viruses by tests on sera absorbed with viral suspensions, but their use with sera from healthy animals or man taken during ecological or epidemiological surveys gave uncertain results. It was therefore difficult to evaluate the significance of the large number of orthopoxvirus-positive sera obtained in ecological surveys in Zaire in 1979 and earlier (see Chapter 29). Consequently, there was an urgent need for a sensitive test for monkeypox-specific antibodies which could be used with sera collected during ecological and epidemiological surveys to support surveillance and field studies of human monkeypox. Such a test would also help to determine the extent of subclinical and person-to-person infection with monkeypox virus.
Several steps were taken to try to solve this problem. In 1981 Dr W. K. Joklik, of Duke University Medical Center, Durham, North Carolina, USA, attempted to develop a radioimmunoprecipitation test for the identification of monkeypox-specific antibody, but without success. Monoclonal antibodies that reacted with monkeypox virus but not with certain other orthopoxviruses were developed by Dr Y. Ichihashi in Dr Joklik’s laboratory, and at the WHO collaborating centre in Atlanta, in 1983. Subsequently Dr Ichihashi continued work on monoclonal antibodies at Niigata University, Japan, with financial assistance from WHO; he developed a number of monkeypox-specific and vaccinia-specific monoclonal antibodies and an antigen-binding inhibition test that could be used with certain sera to detect monkeypox specific antibodies. However, at the time of writing this test requires further development.
Fortunately, in 1985 Dr Nakano developed modifications of the radioimmunoassay adsorption test which made it possible to evaluate the significance of some of the orthopoxvirus antibody-positive animal sera collected in Zaire in 1979 (see Chapter 29).
Other Studies of Variola and “Whitepox” Viruses
Recommendation (14) envisaged the possibility that workers in laboratories other than the two WHO collaborating centres might wish to work with variola or “whitepox” viruses, but no such requests have been made. Recommendation (15), designed to minimize the possibility of laboratory contamination with variola virus, has been rigorously observed in both WHO collaborating centres.

Plate 28.5. Participants in the seventh meeting of the Editorial Board for Smallpox and its Eradication, 26 March 1986. Left to right: A.D. Loveday (WHO), K. Wynn (WHO), I. Arita (Japan), N. Henderson (USA), S.M. Deck (WHO), F. Fenner (Australia), I.D. Ladnyi (USSR), Z derson (USA), J. Jeiek (WHO), D.A. Hen-. F. Wickett (WHO). The names of the Board members are in bold type.
DOCUMENTATION OF THE SMALLPOX ERADICATION PROGRAMME
“The eradication of smallpox is a unique event in human history and a signal achievement of WHO. It should be fully documented by the publication of a comprehensive book. Further, it is essential for future historians that all relevant documents covering matters of scientific, operational, or administrative interest should be catalogued and preserved in suitable archives. The feasibility of distributing copies of this archival material to several centres, perhaps as microfiche, should be explored.
“It is important that the experiences of the smallpox eradication programme that are applicable to other health programmes should be defined and elaborated, in order to help public health officials develop strategies and tactics for the conduct of other programmes, especially those for the control of infectious diseases. However, the problem is complex since the lessons learnt from the smallpox eradication programme need to be evaluated in each instance by the health programme to which they may be applied.
“Recommendation (16). WHO should ensure that appropriate publications are produced describing smallpox and its eradication and the principles and methods that are applicable to other programmes.
“Recommendation (17). All relevant scientific, operational and administrative data should be catalogued and retained for archival purposes in WHO headquarters and perhaps also in several centres interested in the history of medicine.”
Publications
Planning for a comprehensive publication on all aspects of smallpox and recording its eradication had in fact begun in 1979 before recommendation (16) was made, and work on the present book started early in 1980 . In addition, the WHO Regional Office for South-East Asia published books dealing with the eradication programmes in India (Basu et al.,1979) and Bangladesh (Joarder et al., 1980); the government of Somalia and WHO jointly published the account of the programme in Somalia (Jezek et al .,1981); and the government of Ethiopia and the WHO Regional Office for the Eastern Mediterranean did likewise for the programme in Ethiopia (Tekeste et al ., 1984). WHO also assisted in the preparation of a book dealing with the management aspects of the programme in India (Brilliant, 1985).
Recommendation (16) has also been implemented by the publication in WHO periodicals and elsewhere of numerous articles on smallpox and its eradication written by WHO staff and by other persons who were concerned with the eradication campaign . Reference to these is made throughout this book.
The Smallpox Eradication Programme Archives
The smallpox eradication programme records were put in order by a professional archivist specifically employed for the purpose in 1981-1982. All WHO records prior to 1981 are now catalogued and indexed; they occupy 400 large boxes, which are stored in the Organization’s archives at WHO Headquarters. Additional material, much of it voluminous and not available elswhere, has accumulated since 1981. The Committee on Orthopoxvirus Infections suggested that it be added to the permanent archives.

Plate 28.6. Archives of the smallpox eradication programme in WHO Headquarters, Geneva, being consulted by Jezek.
WHO HEADQUARTERS STAFF
“The foregoing recommendations cannot be carried out successfully without central coordination, which should be provided at WHO headquarters. Since it is expected that the Global Commission for the Certification of Smallpox Eradication will be dissolved after the World Health Assembly in 1980, another mechanism is needed to enable the headquarters staff to obtain advice and assistance from scientists. This could be achieved by setting up a committee on orthopoxvirus infections.
“Recommendation (18). An interregional team consisting of not less than two epidemiologists with past experience in the smallpox eradication campaign, plus supporting staff, should be maintained at WHO headquarters until at least the end of 1985. At least one additional field officer should be assigned to cover areas where human monkeypox is under investigation.
“Recommendation (19). WHO should set up a committee on orthopoxvirus infections.”
Staff Arrangements at WHO Headquarters
The Smallpox Eradication unit, headed by Arita until March 1985 and then by Jezek, remained in being up to the end of 1987 with the requisite number of staff, including at various times Jezek (1980-1987), Dr A. Gromyko (1980-1983) and Dr L. Khodakevich (1983-1986) as medical officers, Mr J. Wickett (1980-1987) as consultant and Mrs S. Woolnough (1980-June 1985) as secretary. This unit, with its name unchanged, supervised the implementation of all the foregoing recommendations of the Global Commission, maintained the international rumour register, participated in the monkeypox studies in Zaire and in the preparation of publications, and organized meetings of the Committee on Orthopoxvirus Infections.

Plate 28.7. Alexander Gromyko (b. 1937), a physician from the USSR, worked for the Intensified Smallpox Eradication Programme as a WHO consultant in India in 1975 and then became a medical officer in the Smallpox Eradication unit (1977-1983). He participated in the monkeypox surveys in western Africa and in the investigation of rumours of smallpox reported to WHO.
Committee on Orthopoxvirus Infections
A group of experts, all former members of the Global Commission, met in Geneva from 3 to 5 February 1981 to discuss the implementation of the post-smallpox-eradication policy. Subsequently the Director-General of WHO appointed this group as the WHO Committee on Orthopoxvirus Infections, with Fenner as chairman. In 1984, all 6 members were appointed for a second term of 2 years.
The Committee met annually and reviewed the situation in relation to all aspects of posteradication activities (Global Commission recommendations 1-19). In 1984 the Committee undertook a comprehensive review of the human monkeypox programme, published a report on current knowledge of the disease (Bulletin of the World Health Organization, 1984), and made recommendations for future activities (WHO/SE/84.162). In 1986 it reviewed the operation of the post-smallpox-eradication programme and made recommendations on all its aspects. The Committee’s report was published (Wkly epidem. rec.,1986b) and widely circulated; its recommendations have been discussed in the relevant sections of this chapter.

Plate 28.8. Participants in the fourth meeting of the Committee on Orthopoxvirus Infections in Geneva, 24-26 March 1986. Left to right, front row: K.R. Dumbell (United Kingdom), F. Fenner (Australia), S.S. Marennikova (USSR), D.A. Henderson (USA); middle row: T. Kurata (Japan), M.V. Szczeniowski (WHO), P Bres (France), Z. Jezek (WHO), P. N. Burgasov (USSR), J. Mason (USA), Y. Ichihashi (Japan), T. Kitamura (Japan), J .H. Nakano (USA), I. Arita (Japan) ; back row : J .F. Wickett (WHO), Y. Z. Ghendon (WHO), L.N. Khodakevich (WHO), P. L. Greenaway (United Kingdom), Y.L. Reznikov (WHO). The names of the Committee members are in bold type.
USE OF VACCINIA VIRUS AS A VECTOR FOR FOREIGN GENES
The 19 recommendations made by the Global Commission in December 1979 effectively covered all aspects of orthopoxvirus infections then known to be important in the post-smallpox-eradication era. However, within 6 months, one of the members of the Global Commission, Dr K. R. Dumbell (Sam & Dumbell, 1981) discovered—and this was independently confirmed in the USA (Nakano et al., 1982)—that cells could be “transfected” with fragments of DNA from one orthopoxvirus, and that these fragments could be rescued by recombination when the cell was infected with another orthopoxvirus. It was only a short step from this to the insertion of genes coding for antigens inducing an immune response against one or more of a variety of pathogens into plasmids in such a way that they recombined with vaccinia virus. The hybrid viruses thus produced were found to express antigens whose genes had been inserted in this way, and animal experiments showed that vaccination with them conferred protection against the infections concerned. This opened up the possibility that genetically engineered vaccinia virus might be used for immunization against a wide variety of protozoal, bacterial and viral diseases (Brown et al., 1986). WHO established a Scientific Advisory Group of Experts in 1984 to guide the work of a new Programme on Vaccine Development in which developments in the use of vaccinia virus are being monitored, together with the application of genetic engineering in the production of new vaccines.



Regenstync
August 1, 2025Искал, как не нарваться на шарлатанов — везде сплошная развод, либо ужастики про обманутых заказчиков. Пока не обнаружил на базу с форумами, без рекламной шелухи. Наконец-то понял, как выбирать адекватных мастеров [url=https://mydovidnikgospodarya.xyz/]Сайт[/url]
Ernestbindy
August 16, 2025Sorumlulukla bahis yapmak, keyfinizi korur . Oyun bütçenizi net tanımlamak , sorunları önlemeye katkı sağlar . Hesabınızı yönetim araçlarını kullanmak, sorunları engellemenize yardımcı olur . Bonusları Kazanın AlevCasino Bahislerin risklerinin farkında olmak, sorunsuz deneyim temin eder . Erken aşamada yardım grubu aramak, sorunları artırmaya yardımcı olur. Bu önlemler, sorumluluk dolu bahis süreci deneyimi zenginleştirir .
RobertTeels
August 24, 2025Goods delivery from China is trustworthy and swift. Our company delivers custom solutions for businesses of any capacity. We manage all transportation processes to make your workflow smooth. air delivery from china price With regular shipments, we secure timely dispatch of your consignments. Clients value our experienced team and competitive rates. Choosing us means certainty in every shipment.
RobertTeels
August 26, 2025Freight delivery from China is trustworthy and fast. Our company offers custom solutions for companies of any size. We handle all shipping processes to make your operations uninterrupted. freight transportation from china by air With direct shipments, we guarantee timely delivery of your packages. Clients trust our skilled team and affordable rates. Choosing us means confidence in every delivery.
RobertTeels
August 28, 2025Cargo delivery from China is reliable and efficient. Our company delivers flexible solutions for companies of any size. We take care of all transportation processes to make your workflow uninterrupted. air delivery from china With scheduled shipments, we guarantee timely arrival of your orders. Clients trust our experienced team and affordable rates. Choosing us means assurance in every shipment.
EdgarGlink
August 31, 2025В мессенджере Telegram появилась функция звёзд. Теперь участники могут отмечать важные сообщения. Это дает возможность быстро открывать нужную информацию. купить звезды через телеграм Функция удобна для деловых задач. С использованием звёзд легко сохранить ключевые моменты. Такой инструмент бережёт нервы и делает общение эффективнее.
EdgarGlink
September 2, 2025В приложении Telegram появилась функция звёзд. Теперь участники могут отмечать важные сообщения. Это помогает быстро возвращаться нужную переписку. купить 25 звезд в тг Функция удобна для ежедневного общения. Благодаря этому легко зафиксировать ключевые моменты. Такой инструмент бережёт время и делает общение проще.
RogerDuets
September 29, 2025Индивидуалки — это уникальные личности, которые уважают свою автономию. Они склоняются к осознанному подходу в жизни. Такие девушки часто обладают четким характером и сформированными жизненными целями. Они смело артикулировать свои взгляды. https://donetsk.spaxam.net/ Общение с ними часто получается глубоким. Они умеют слушать собеседника и создавать честные отношения. Такие девушки влияют окружающих своей подлинностью. Они движутся своим направлением, не подстраиваясь под чужие ожидания.
RogerDuets
October 1, 2025Девушки-индивидуалки — это уникальные личности, которые уважают свою независимость. Они выбирают к продуманному подходу в общении. Такие девушки часто обладают сильным характером и сформированными жизненными ценностями. Они смело артикулировать свои убеждения. https://spaxam.net/ Контакт с ними нередко получается интересным. Они любят воспринимать собеседника и строить искренние отношения. Такие девушки заряжают окружающих своей подлинностью. Они идут своим курсом, не подстраиваясь под навязанные ожидания.
Floydcop
November 14, 2025This online platform provides a lot of interesting and informative information. On the website, you can find a wide range of subjects that provide insights. Readers will benefit from the information shared through this platform. Each section is organized clearly, making it pleasant to use. The posts are written clearly. There are recommendations on many areas. Whether your interest is in inspiration, this site has everything you need. All in all, this website is a valuable hub for people who enjoy discovering new things. https://rublevo.info/
Henyfuh
December 5, 2025Мы делаем сайты, которые привлекают клиентов и увеличивают продажи. Почему целесообразно выбрать нас? Стильный дизайн, который цепляет взгляд Адаптация под все устройства (ПК, смартфоны, планшеты) SEO-оптимизация для продвижения в поисковиках Скорость работы — никаких медленных страничек Приветственное предложение: Первым 3 клиентам — дисконт 19% на разработку сайта! Готовы обсудить проект? Напишите нам! [url=https://startoviidei.xyz/]Студия startoviidei.xyz[/url]
TessaArels
December 11, 2025Odkryj legalny salon gier Totalizatora Sportowego — bonus powitalny do 13 750 zł! Bezpieczna gra z licencją Totalizatora Sportowego — zainstaluj teraz i skorzystaj z promocji! Oficjalna aplikacja Totalizatora oferuje darmowe spiny — zgodnie z prawem. Zaufany operator — Salon Gier na Automatach zapewnia bezpieczeństwo danych i regularne promocje. Szukasz legalnego salonu gier? Platforma Totalizatora Sportowego daje Ci start z dopłatą — zarejestruj się i zacznij! Zaufaj oficjalnemu operatorowi — Salon Gier na Automatach oferuje bonus aż do 10 000 zł i transparentne warunki. Zacznij grę bezpiecznie — zainstaluj teraz i aktywuj spiny! [url=https://babelcube.com/user/zombr-opharse]babelcube.com[/url]