Chapter 29:
Human monkeypox and other poxvirus infections of man
INTRODUCTION
Human monkeypox was first recognized in 1970; it is a severe systemic disease with a generalized pustular rash, clinically indistinguishable from smallpox. In addition to variola and monkeypox viruses, 7 other species of poxvirus, of 4 genera, can cause lesions in man (Table 29.1). Although infection with each of these viruses produces at the most mild symptoms and usually only a localized skin lesion, the diseases in question presented a potential diagnostic problem during the global eradication of smallpox, since virus particles found in lesions by electron microscopic examination could be confused with those of variola virus. Because of its importance, monkeypox is the main subject of this chapter, but a brief description is given of each of the other poxvirus infections of man.
MONKEYPOX IN CAPTIVE PRIMATES
Monkeypox virus was discovered in 1958, when it was isolated from the lesions of a generalized vesiculo-pustular disease among captive monkeys at the State Serum Institute, Copenhagen (Magnus et al ., 1959). It was apparent that if an animal reservoir of variola virus existed the eradication of smallpox would be impossible (see Chapter 10). The close resemblance between smallpox and monkeypox in captive primates focused attention on monkeypox virus as a potential threat to smallpox eradication (Arita & Henderson, 1968). WHO therefore contacted laboratories in Europe and North America which used monkeys (27 in 1968 and 51 in 1970; Arita et al., 1972), inquiring about the occurrence of monkeypox and asking specifically whether any infections had occurred among laboratory workers or animal handlers. The ensuing investigations revealed 4 other reported outbreaks and 4 hitherto unreported outbreaks in primates (Table 29.2), but there were no reports of infection in humans. Monkeypox virus was recovered in 6 of these episodes. All except episode 3 occurred in Asian monkeys, although in some outbreaks African primates (and, in episode 2, New World monkeys) were also infected.
The circumstances of these outbreaks have been summarized by Arita et al. (1972). One episode described in their paper, but omitted from Table 29.2, calls for special commentnamely, the observation made by Gispen & Kapsenberg (1966) of the National Institute of Public Health in Bilthoven, Netherlands, that monkeypox virus had been recovered from normal cynomolgus kidney cell cultures. Subsequent examination of the laboratory records led Dr J. G. Kapsenberg (personal communications, 1980, 1983) to decide that this isolation was probably due to inadvertent laboratory contamination of the culture with monkeypox virus, which had been isolated in the same laboratory at about this time from animals infected in the Blijdorp Zoo outbreak (episode 2).
Seven of the 9 outbreaks of monkeypox in captive monkey colonies between 1958 and 1968 occurred in monkeys shipped from Asia, leading to the suspicion that the reservoir of monkeypox virus was probably located in that continent. However, collaborative serological surveys organized by WHO failed to detect orthopoxvirus antibodies in over 1000 monkey sera collected in India, Indonesia, Japan and Malaysia (Arita et al.,1972). After the discovery of human monkeypox in Africa in 1970 (see later in this chapter), sera were collected from monkeys and other animals in Zaire and several countries of western Africa. Monkeypox-virus-specific antibodies were demonstrated in sera from 8 species of monkey and 2 species of squirrel, and monkeypox virus was recovered from the organs of a squirrel (see below).
Although primates from Asia, Africa and South America (and an anteater from the last mentioned area) experienced infections with monkeypox virus in captivity, there is no evidence that the virus occurs naturally anywhere except in Africa. During the period 1958-1968 large numbers of primates were being imported into Europe and North America from Asia, and smaller numbers from western Africa, mainly for the manufacture and safety testing of poliomyelitis vaccines. At that time the conditions under which monkeys were moved from their place of capture in Asia or Africa to the recipient laboratory in Europe or North America presented many opportunities for them to be infected with agents carried by other wild animals or by man while in transit (Kalter & Heberling, 1971). The cessation of outbreaks after 1968 can be ascribed to improved conditions in the shipment of primates at about that time and the much more extensive use by laboratories of monkeys bred in captivity in Europe and North America.
The clinical features of naturally occurring cases in cynomolgus monkeys have been described by Magnus et al. (1959) and Sauer et al. (1960). No signs are detected until the rash 1289 appears, usually as a single crop of discrete papules over the trunk and tail and on the face and limbs, being particularly abundant on the palms of the hands and the soles of the feet (Plate 29.1). The papules become vesicular and then pustular and are often umbilicated. Scabs develop and fall off 7-10 days after the onset of rash, leaving small scars. Circular discrete ulcers about 2 mm in diameter often occur in the oropharynx.
The severity of symptoms varied among the several different primate species infected in the outbreak of the Blijdorp Zoo in Rotterdam (episode 2, Table 29.2). All the species suffered from a generalized disease characterized by pocks on the skin, lips and mucous membranes. Orang-utans were particularly susceptible, several dying in the acute viraemic stage, before the skin lesions were fully developed.
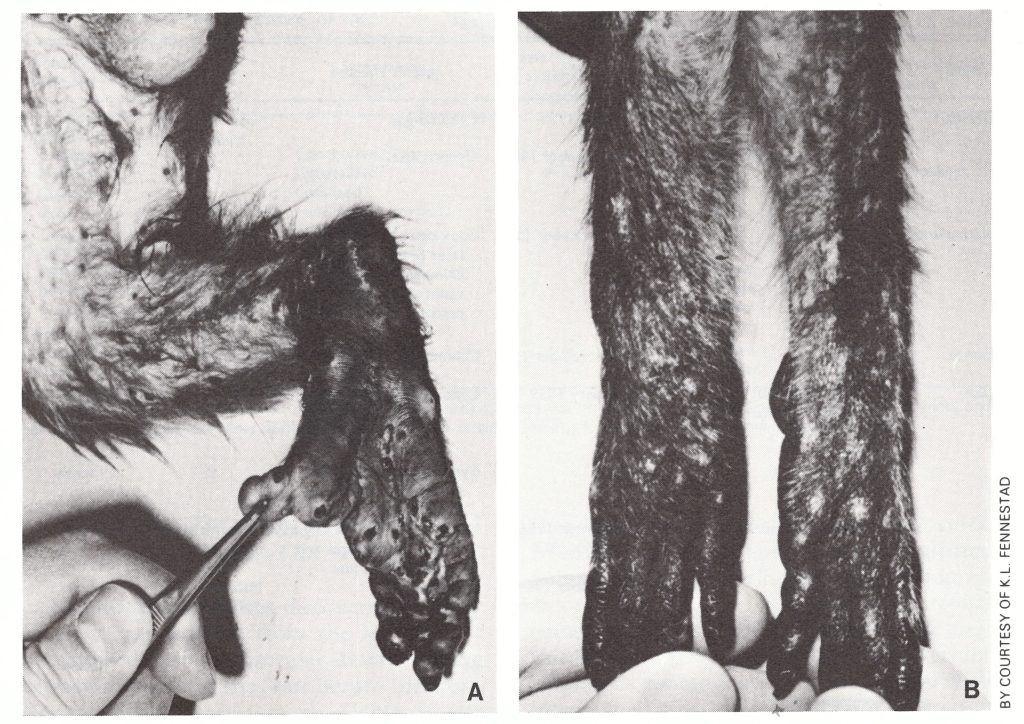
Plate 29.1. Generalized lesions of monkeypox in a cynomolgus monkey. A: Acute stage; pustules on the leg and sole of the foot. B: Convalescent stage; healing pustules and scars. (From Magnus et al., 1959.)
THE PROPERTIES OF MONKEYPOX VIRUS
In Chapter 2 the biological characteristics of monkeypox virus have been enumerated together with those of other orthopoxviruses (Table 2.3), and the restriction endonuclease map of monkeypox virus DNA has been compared with the corresponding maps of DNAs of other species of Orthopoxvirus (Fig. 2.6, 2.7 and 2.9).
Pathogenicity for Laboratory Animals
Monkeypox virus has a broad host range and infects most of the common laboratory animals, producing moderate-sized haemorrhagic pocks on the chorioallantoic membrane and a large indurated swelling with a haemorrhagic centre after intradermal inoculation into rabbits (see Chapter 2, Plates 2.5 and 2.6). It produces lytic plaques in most kinds of cultured cells, but unlike variola virus, it does not grow in pig embryo kidney cells when first cultured in them, although adaptation occurs quickly.
Comparison of DNA Maps of Strains of Monkeypox Virus
Esposito & Knight (1985) analysed the DNA of 12 strains of monkeypox virus, 4 recovered from outbreaks in laboratory primates in Europe and North America and 8 from human cases in 4 countries in central and western Africa . The physical map locations of the sites of cleavage by the restriction endonuclease HindIII for the DNA of these strains of monkeypox virus, and the DNA of 2 strains each of variola and vaccinia viruses, are compared in Fig. 29.1.
As has been shown in the other comparisons described in Chapter 2, the DNAs of all strains of monkeypox virus are clearly different from those of both variola and vaccinia viruses. However, the monkeypox virus DNAs cluster into 2 groups, according to the geographical origins of the specimens, rather than the animal of origin (man or monkey) or the year of isolation. The upper group of 4 in Fig. 29.1 are strains from human cases occurring in Zaire. The lower group consist of human isolates from 3 countries in western Africa—Nigeria, Liberia and Sierra Leone—and isolates from outbreaks in captive monkeys between 1959 and 1969. It is probable that all the outbreaks in captive monkeys (see Table 29.2) originated from western Africa rather than Zaire, since exports of monkeys from Africa in the late 1950s and during the 1960s were from western African countries.
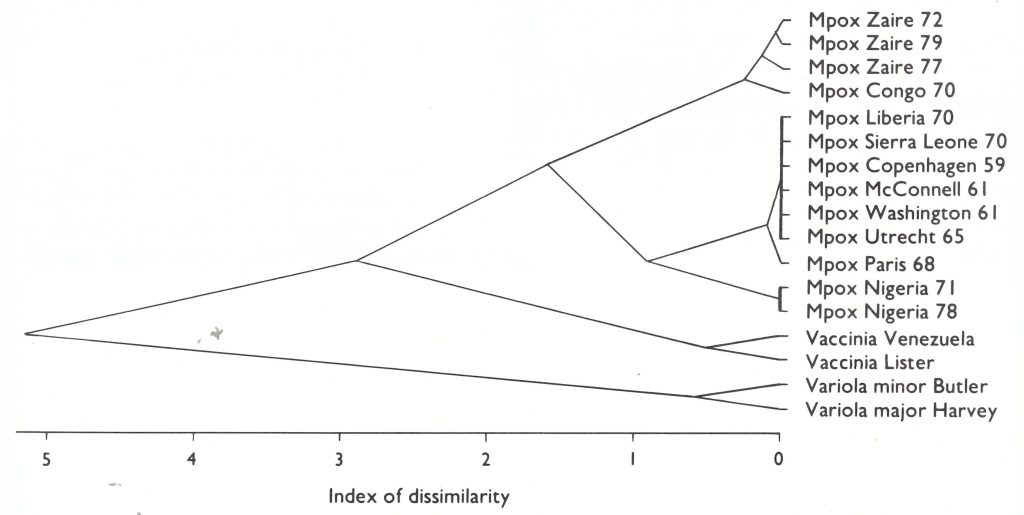
Fig. 29.1. Dendrogram illustrating the similarities and differences between the Hindlll cleavage sites of the DNAs of 12 strains of monkeypox (Mpox) virus (McConnell and Washington are different passages of the same strain), 2 strains of vaccinia virus and 2 strains of variola virus. Presence, absence or impossibility of cleavage sites were analysed as described by Gibbs & Fenner (1984) using the squared Euclidean metric (number of attributes=36). (Data from Esposito & Knight, 1985.)
Genetic Studies
Like other orthopoxviruses that produce haemorrhagic pocks on the chorioallantoic membrane, monkeypox virus produces white pock mutants. These were first observed by Bedson (1964) and first reported by Gispen & Brand-Saathof (1972). They were shown closely to resemble the parental monkeypox virus in tests for species-specific antigen (Gispen et al ., 1976) and intracellular polypeptide patterns (Harper et al ., 1979).
Subsequently it was reported that variants called “whitepox” viruses, which resembled variola virus by all biological tests, could be recovered from certain laboratory stocks of monkeypox virus, either by passage in hamsters (Marennikova & Shelukhina, 1978) or by inoculation on the chorioallantoic membrane (Marennikova et al.1979). This initially raised important questions about a possible animal reservoir of variola virus, but these were subsequently discounted. By about 1982 accumulating evidence had convinced most laboratory workers that the “whitepox” viruses were in fact strains of variola virus inadvertently introduced as laboratory contaminants (see Chapter 30).
Species Diagnosis
The biological characteristics used to identify monkeypox virus and, in material derived from human cases, to distinguish it from variola virus, are the haemorrhagic pock and high ceiling temperature on the chorioallantoic membrane, the production of a large haemorrhagic lesion after intradermal inoculation in rabbits, its wide host range and its failure to grow in pig embryo kidney cells when first inoculated into these cells. The DNA map is characteristic of the species, but can be used to distinguish western African from Zairian strains.
Serological Diagnosis of Past Monkeypox Infection
An understanding of the ecology of monkeypox virus depends either on the isolation of virus from animals captured in the field or on serological surveys for monkeypox-virus specific antibodies. The isolation of virus from animals captured in the field is likely to be a rare event in orthopoxvirus infections, in which persistent infection does not occur, and in fact only one such isolation has been made (see below).
During the 1970s methods had been developed that enabled species-specific diagnoses of recent infection with monkeypox, vaccinia and variola viruses to be made with hyperimmune or other highly potent sera, by adsorption with appropriate viral suspensions and tests for residual antibody by gel precipitation (Gispen & Brand-Saathof, 1974), immunofluorescence (Gispen et al .,1976), radioimmunoassay (Hutchinson et al.,1977), and enzyme-linked immunosorbent assay (ELISA) (Marennikova et al .,1981) (see Chapter 3). For these tests, rather large quantities of high-titre serum were required, and for the radioimmunoassay adsorption test employed by the WHO collaborating centre in the Centers for Disease Control in Atlanta, GA, USA, antibodies to the gammaglobulin of the relevant species were thought to be necessary, which were available for monkeys but not for other species of wild animals.
However, as a result of experience with sera from persons known to have human monkeypox, some of whom had been vaccinated years earlier, Dr J. H. Nakano (personal communication, 1984) developed criteria that allowed a positive or presumptive diagnosis of monkeypox to be made in most suspected cases involving human sera. In mid-1985, Dr Nakano and Mrs Donna Miller (personal communication, 1986) developed a method of carrying out radioimmunoassay adsorption tests with sera from squirrels and some other species of wild animals, using staphylococcus A protein instead of a species-specific antigammaglobulin. This made it possible to test many animal sera from the field and has helped to elucidate the ecology of monkeypox virus.
The Nomenclature of Poxvirus Diseases
For centuries it has been traditional to name poxvirus diseases after the animals in which they were first observed—for example, cowpox, horsepox, sheep-pox and fowlpox. The practice has continued in more recent times with the use of the terms monkeypox, rabbitpox, buffalopox, elephantpox virus to designate the viral agents recovered from infections of the animals concerned. Some of these designations are misleading. Thus, it now appears that "cowpox" virus is primarily a disease of rodents, which has a wide host range and occasionally infects cows, cats, zoo animals and man. Rabbitpox and buffalopox are caused by strains of vaccinia virus, propagated in series in rabbits and buffaloes respectively. And although African monkeys are infected in nature with monkeypox virus and may indeed be an important source of infection of humans, they are probably sentinel animals, only occasionally infected with this virus, rather than its principal reservoir host.
HUMAN MONKEYPOX
Discovery of Human Infections
The first case of human monkeypox was found in the Basankusu Hospital, Equateur Province, Zaire (Ladnyj et al.,1972). The Basankusu Zone covers an area of about 20 000 square kilometres and in 1970 had an estimated population of 62 000, mostly primitive farmers and hunter-gatherers living in small villages in dense tropical rain forest . The last known outbreak of smallpox in Basankusu Zone occurred in 1968 and comprised 70 cases with 18 deaths. Several suspected cases of smallpox were treated at the hospital in 1969, but none was confirmed. Two suspected cases were reported in 1970 ; one of these turned out to be chickenpox, and the other was the first case of human monkeypox to be detected. The patient, a 9-month-old boy, became ill with fever on 22 August 1970 and a rash developed 2 days later. He was admitted to hospital on 1 September, the 9th day of the rash, which had the characteristic centrifugal distribution of smallpox. Crusts were collected for laboratory examination and sent through WHO in Geneva to the WHO collaborating cenire in the Moscow Research Institute for Viral Preparations, USSR. The patient recovered and was about to be discharged, but on 23 October he developed measles (acquired while in hospital) and died 6 days later.
During 1970 the WHO collaborating centre in Moscow had received a number of specimens from various provinces of Zaire (but not from Equateur Province) from which variola virus had been recovered. The virus from Basankusu Hospital produced pocks on the chorioallantoic membrane that were quite different from those of variola virus. More detailed studies of this isolate, including inoculation in rabbit skin, showed that it was monkeypox virus (Marennikova et al .,1972a). Investigations of the epidemiological circumstances of the patient by Ladnyiand Dr P. Ziegler in 1971 revealed that the child was the only unvaccinated member of his family, and that there had been no other cases of fever with rash recently in the village concerned or in neighbouring villages. Such an isolated case was most unlikely to be smallpox.
The discovery of human monkeypox in central Africa in September 1970 was followed by the demonstration that 4 cases of suspected smallpox in Liberia and 1 case in Sierra Leone in 1970, and 1 each in Nigeria and Cote d’Ivoire in 1971 (Foster et al.,1972) were cases of human monkeypox (Lourie et al.,1972). A series of coordinated laboratory and field studies was organized to determine the incidence of the disease, to study its clinical features and epidemiology and to search for the animal reservoir or reservoirs of the virus.

Plate 29.2. Meeting of the Informal Consultation on Monkeypox and Related Viruses, Geneva, February 1976. Left to right, front row: T. Kitamura (Japan) C. I. Sands (WHO), C. Algan (WHO), F. Fenner (Australia), I. Arita (WHO), J.H. Nakano (USA); middle row: J. G. Breman (USA), R. Netter (France), E. A. Smith (Nigeria), S. S. Kalter (USA), I. D. Ladnyi (USSR), H. S. Bedson (UK), S. S. Marennikova (USSR), A. N. Slepushkin (WHO); back row :M. V. Szczeniowski (WHO), E .S . Johnson (Sierra Leone), B . Guyer (USA), N . French (USA), I . Tagaya (Japan), W .K . Joklik (USA), D .A. Henderson (WHO), K .R . Dumbell (UK), V .N . Milushin (USSR), A .C . Hekker (Netherlands).
Organization of Laboratory Research
In order to obtain guidance on what further research might be undertaken on the problem of monkeypox and to find out whether there was any evidence of an animal reservoir of variola virus, the WHO Smallpox Eradication unit organized informal discussions on monkeypox virus studies among a group of virologists, which met first in Moscow from 26 to 31 March 1969. These experts agreed that monkeypox virus could be readily distinguished from variola and vaccinia viruses by its biological characteristics, and further studies were planned on its morphology and behaviour in experimentally infected primates, and on the use of serological tests to determine its geographical distribution.
The discovery of human monkeypox, and the subsequent reports that a variola-like virus (“whitepox” virus) had been recovered from the organs of animals captured in areas of Africa in which human monkeypox cases had occurred (see Chapter 30), clearly called for expert advice from virologists. Further meetings of the Informal Group on Monkeypox and Related Viruses were therefore arranged. In all, 5 more meetings were held, in 1971, 1973, 1976, 1978 and 1979, and were attended by a total of 32 scientists from laboratories in 10 countries, including the WHO collaborating centres in Atlanta and Moscow as well as other collaborating centres conducting poxvirus research, in Bilthoven, Birmingham, London, Paris and Tokyo. In addition, field workers from 5 countries in central and western Africa and representatives from the WHO Regional Office for Africa, in Brazzaville, Congo, attended some of the meetings. Laboratory research on monkeypox, variola and “whitepox” viruses was carried out in the laboratories of the scientists who attended the meetings, sometimes with financial assistance from WHO.
Organization of Field Research
To determine the most effective way of conducting the field research, the Smallpox Eradication unit organized a coordination meeting of representatives from central and western African countries in the WHO Regional Office for Africa, in October 1976, to assess the epidemiological situation in relation to human monkeypox, to draw up procedures for field surveys and to assess the sensitivity of epidemiological surveillance. It was clear from the data presented at this meeting that the best surveillance programme was that conducted in Zaire, the only country in the region in which smallpox surveillance had been maintained until eradication was certified in 1977 (see Chapter 25). Following a recommendation of the coordination meeting, an intensive surveillance programme was set up under the leadership of Dr Kalisa Ruti of the Ministry of Health of Zaire and Mr M. Szczeniowski, a WHO technical officer, in a geographically limited area in the northern part of Zaire (Equateur Region, Mongala Subregion), in which a high concentration of human monkeypox cases had been observed.
This activity was extended in 1979-1980; and in May 1980 the Thirty-third World Health Assembly accepted the recommendation of the Global Commission for the Certification of Smallpox Eradication that further research was needed to determine the public health importance of human monkeypox. Field activities in Zaire were strengthened (see Chapter 28), and laboratory support was provided by the WHO collaborating centres in Atlanta and Moscow. The description in the following pages of the clinical features and epidemiology of human monkeypox and the ecology of monkeypox virus is based on these studies, carried out over approximately a decade but most intensively in the years 1982-1986.
Incidence and Distribution
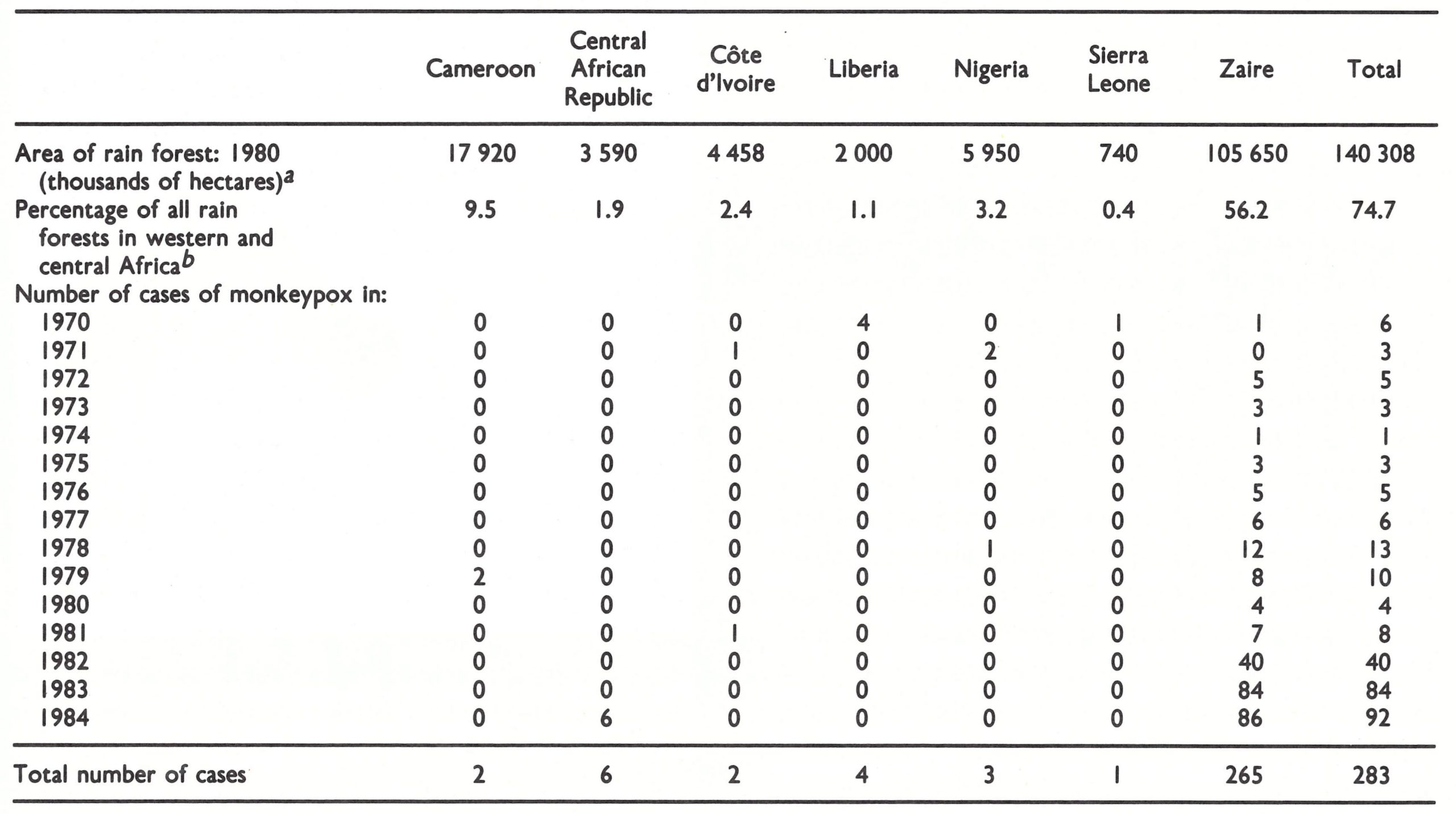
Just over 400 cases of human monkeypox were reported between 1980 and 1986, all of which occurred in tropical rain forest areas of central and western Africa. At the time of writing, detailed analyses were available of the 283 cases reported between 1970 and 1984 (Fig. 29.2; Table 29.3). Of these, 89% were in small villages (under 1000 inhabitants) and 10% in larger villages (1000-5000 inhabitants); only 3 cases were reported in towns of over 5000 inhabitants. Even the last-named population groups had ample opportunities for direct contact with animals killed in the rain forests.
Between 1972 and 1981, the cases reported from Zaire greatly outnumbered those reported from any other country (Table 29.3), probably because the number of people living in villages in tropical rain forests is much larger there. From 1982 onwards many more cases were reported from Zaire than in previous years. This was partly due to the intensive surveillance system that had been developed in enzootic foci in that country, but there appears to have been a real increase in the incidence in 1983 and 1984. The reason for this increase is still uncertain. It may have been attributable in part to the fact that there were many more unvaccinated children than in earlier years, and in part, perhaps, to fluctuations in the extent of infection in the animals from which human infections were acquired.

Plate 29.3. Mark V. Szczeniowski (b. 1944), a former United States Peace Corps volunteer, joined WHO in 1971 and worked as a leader of one of the mobile smallpox surveillance teams in Zaire. From 1980 he participated in the epidemiological surveillance of human monkeypox and viral haemorrhagic fevers in that country.
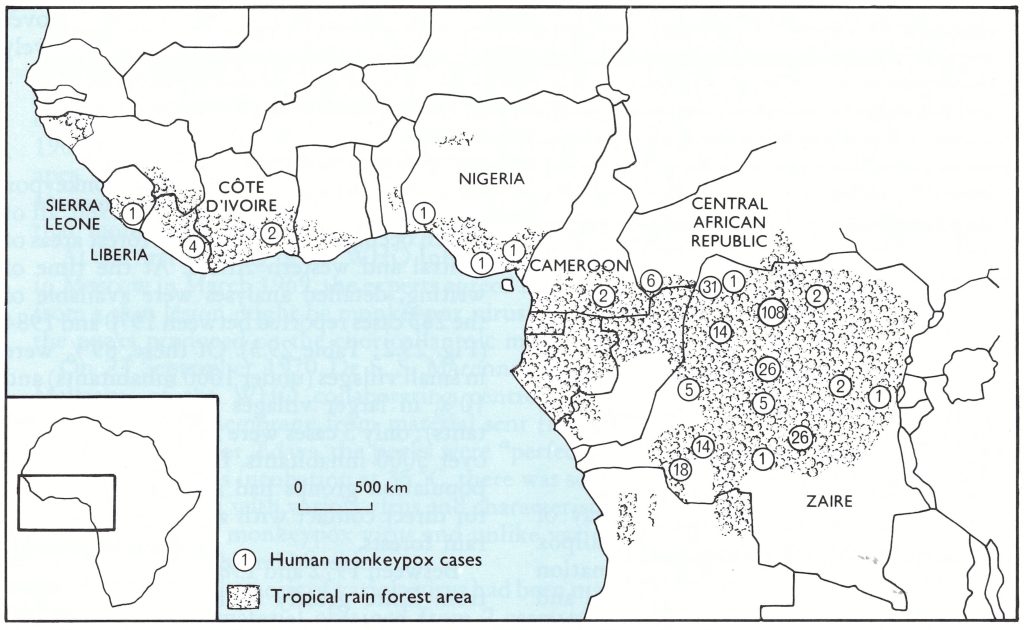
Fig. 29.2. Western and central Africa, showing the extent of tropical rain forest and the locations where casesof human monkeypox have occurred, 1970-1984.
Table 29.3: Human monkeypox: areas of tropical rain forest and annual numbers of cases reported in countries in western and central Africa, 1970-1984
 a Source: Food and Agriculture Organization of the United Nations (1981).
a Source: Food and Agriculture Organization of the United Nations (1981).
b Areas of rain forest (25 .3% of total) occur In 6 countries of western and central Africa in which human monkeypox has not been reported.
Clinical Features
A description of the clinical features of human monkeypox based on 47 cases diagnosed up to the end of 1979 (Breman et al ., 1980) needs little revision in the light of experience since then (Arita et al . 1985) . Clinically, human monkeypox closely resembles discrete ordinary-type or, occasionally, modified-type smallpox, as described in Chapter 1 . No case has yet been seen, among the cases diagnosed in the years 1970-1984, with confluent lesions on the face, nor has any case comparable to flat-type or haemorrhagictype smallpox been diagnosed. The obvious clinical feature that differentiates human monkeypox from smallpox is the pronounced lymph-node enlargement seen in most cases of monkeypox (Plates 29.4 and 29.5), sometimes only in the neck or inguinal region, but more often generalized. Lymph-node enlargement occurs early, and has often been observed at the time of onset of fever, usually 1-3 days before the rash appears. Lymph-node enlargement was observed in 90% of 98 cases in which its presence or absence was recorded and was a presenting sign, preceding the rash, in 65% of these cases.
The eruption begins after a prodromal illness lasting 1-3 days, with fever, prostration and usually lymph-node enlargement. As with smallpox, the lesions develop more or less simultaneously and evolve together at the same rate, through papules, vesicles and pustules, before umbilicating, drying and desquamating. This process usually takes about 2-3 weeks, depending on the severity of the disease. The distribution of the rash is mainly peripheral . Severe eruptions can cover the entire body (Plate 29.4), including the palms and soles. Most pustules are about 0.5 cm in diameter but some have been seen up to 1 cm in diameter. Lesions have been noted on the mucous membranes, the tongue and genitalia. One patient, who had been vaccinated several years previously, developed only 1 lesion, further emphasizing the fact that some cases can be exceedingly mild and would go unreported in the absence of active surveillance. As is described below, subclinical cases also occur, in unvaccinated as well as vaccinated subjects.
Sequelae
As in smallpox, pitting scars may develop, most frequently on the face, but they tend to diminish in prominence with time. Secondary infection of the lesions is common and this may play a role in scarring. About half of the scars from lesions seen initially on the face and body were detectable 1-4 years after the acute illness. Desquamation of crusts leaves areas of hypopigmentation (Plate 29.5 B). Hyperpigmentation follows after a few months (Plate 29.4 D) and usually diminishes with time. In some cases large shallow residual scars are seen, and in a few cases corneal lesions have caused unilateral blindness.
Laboratory confirmation
Throughout the investigations, great importance was attached to obtaining laboratory confirmation of the clinico-epidemiological diagnoses, initially because of the possible occurrence of smallpox and later because of the suspicion that “whitepox” virus (see Chapter 30) might infect humans. All laboratory diagnoses were made in the WHO collaborating centres, with the results shown in Table 29.4. The methods of laboratory diagnosis were those used for smallpox, supplemented by serology in cases in which viral isolation was not possible. This combination allowed positive diagnoses to be made in the great majority of cases. In spite of unavoidable delays in the collection and transmission of specimens, the percentage of recoveries of virus from samples taken from cases eventually diagnosed as human monkeypox was high. Virtually all the cases found positive by electron microscopy were also found positive by culture, and vice versa, but 60 (22%) of the cases were seen too late to obtain lesion material and could only be confirmed serologically. Retrospective diagnosis by serology was unequivocal in unvaccinated subjects but sometimes less clear in vaccinated persons, although with the experience gained over the past few years diagnoses are now possible in these cases also.
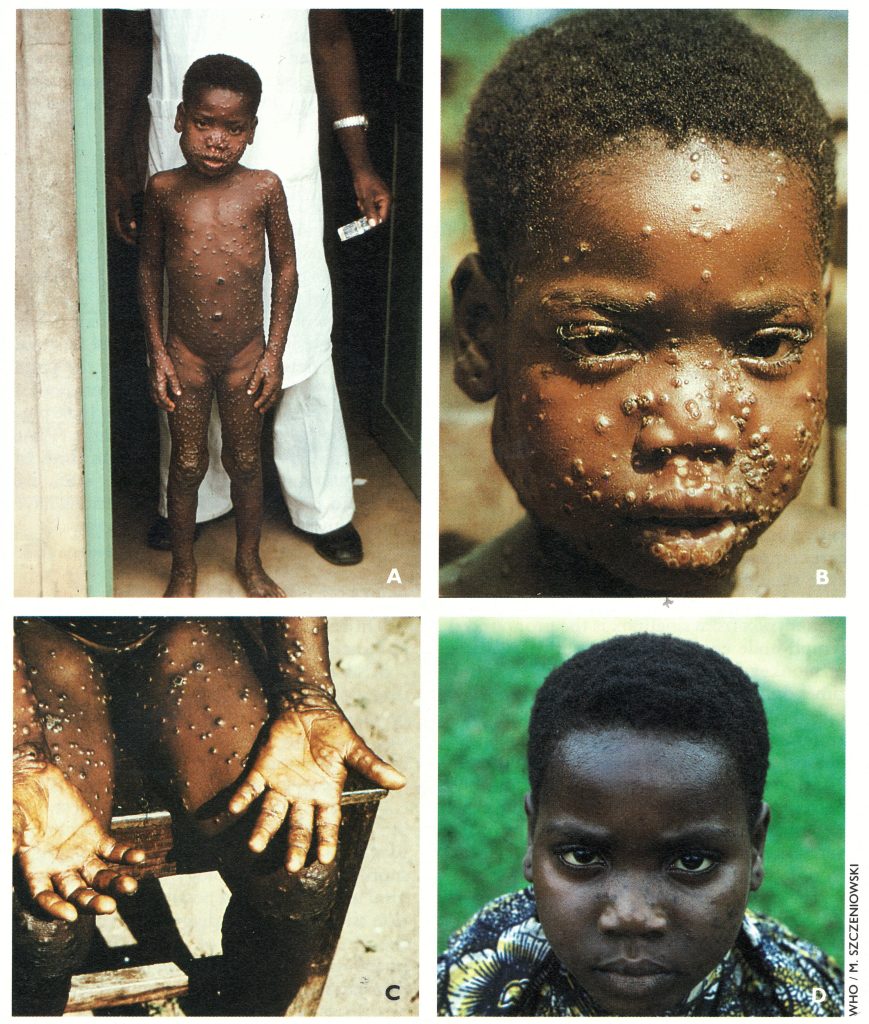
Plate 29.4. Human monkeypox in a 7-year-old Zairian girl. A, B, C: Acute stage, day 7 of rash. Note bilateral inguinal lymphadenopathy and enlarged submaxillary lymph nodes on right side. Pustular lesions on lips (B) also occur inside the mouth as ulcerated lesions: the enanthem. D: Same subject, 4 1/2 years later. There are several hyperpigmented spots and facial pockmarks; in about half the cases of monkeypox these disappear within 5 years of the attack.
Severity and case fatality rates
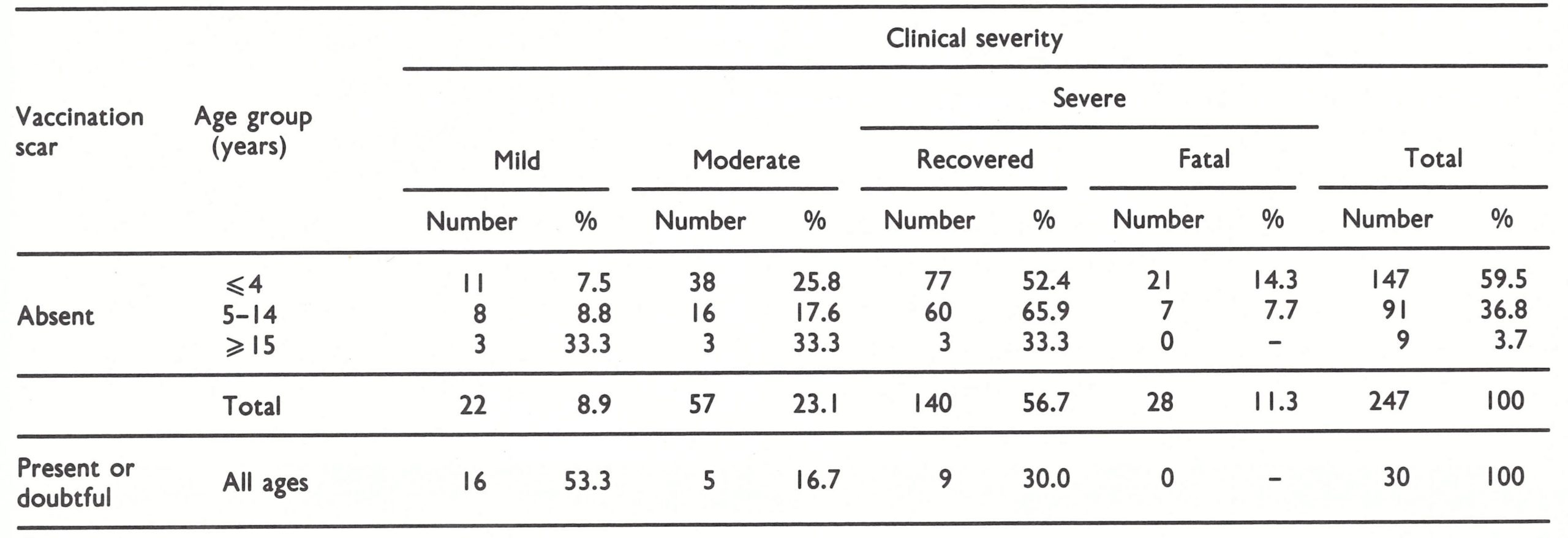
On the basis of the number of skin lesions and the severity of systemic symptoms, cases in Zaire were classified as mild, moderate or severe (Table 29.5). The majority of cases (53.1%), and the majority of severe cases among the unvaccinated (58.3%), occurred among unvaccinated children in the age group 0-4 years. The 28 deaths all occurred in children between 7 months and 7 years of age (overall case-fatality rate among unvaccinated subjects, 11.3%), the case-fatality rate for the age group 0-4 years (14.3%) being almost twice that in unvaccinated children aged 5-14 years (7.7%).
Few cases were diagnosed in vaccinated subjects, only 30 of the 277 patients (10.8%) having a visible vaccination scar. The youngest of these was a 5-year-old boy who had been vaccinated shortly after birth and developed monkeypox late in 1983—i.e., about 5 years after vaccination.
Subclinical infections
Some cases of monkeypox in vaccinated subjects were extremely mild, with very few skin lesions. By analogy with smallpox (Heiner et al.,1971a; see Chapter 1), it was to be expected that many infections in vaccinated subjects would be subclinical. A more important question was whether inapparent infections occurred in unvaccinated human subjects. Data pertaining to this problem emerged from the intensive surveillance activities in Zaire in 1982-1984 Uezek et al.,1986b). During that period 2510 contacts of 131 confirmed cases of human monkeypox were examined and questioned, often on several occasions. Sera were taken from 70% of the unvaccinated contacts and 6% of the vaccinated contacts and tested at the WHO collaborating centres in Atlanta and Moscow (Table 29.6). The laboratory tests showed that 91 (16%) of the contacts examined had been infected with monkeypox virus. Sixty of the 73 cases in unvaccinated contacts had a history or lesions compatible with human monkeypox, and 40 of them appeared to be secondary cases resulting from transmission of infection from another human case. The other 13 unvaccinated subjects (18%) gave no history and had no lesions suggestive of human monkeypox and must therefore be classed as cases of subclinical infection. The majority of such cases occurred in children aged between 2 and 10 years who had been household contacts of a severe case of human monkeypox. Only I subclinical case was recognized in a vaccinated subject, a 20-year old man, but no special effort was made to detect subclinical infections among vaccinated subjects in a way comparable to the studies of Heiner et al. (1971a) with variola major in Pakistan.
Large-scale serological surveys of unvaccinated persons in Zaire (Jezek et al.,1987a; see later in this chapter) also revealed a few cases of subclinical infection.
Table 29.5: Human monkeypox in Zaire, 1970-1984: vaccination status, age distribution of unvaccinated patients and severity of illness (including deaths)a,b
 a Number of cases and deaths by percentages of all cases in unvaccinated and vaccinated groups respectively.
a Number of cases and deaths by percentages of all cases in unvaccinated and vaccinated groups respectively.
b Mild: less than 25 skin lesions; no Incapacity and no need for special care. Moderate: 25-99 skin lesions; incapable of most physical activity but not requiring nursing care. Severe, non-fatal: 100 or more skin lesions; fully incapacitated and requiring medical care. Fatal: deaths due to monkeypox, usually occurring in "severe" cases.

Plate 29.5. A: Human monkeypox: 3-year-old Zairian boy with rash in the scabbing stage. Axillary lymph nodes are still enlarged. B: Human monkeypox: I-year-old Zairian boy, day 24 of rash. There are depigmented spots where the scabs have come off. Inguinal lymphadenopathy is still present. C: Typical tropical rain forest in a region in Zaire where cases of human monkeypox have occurred.
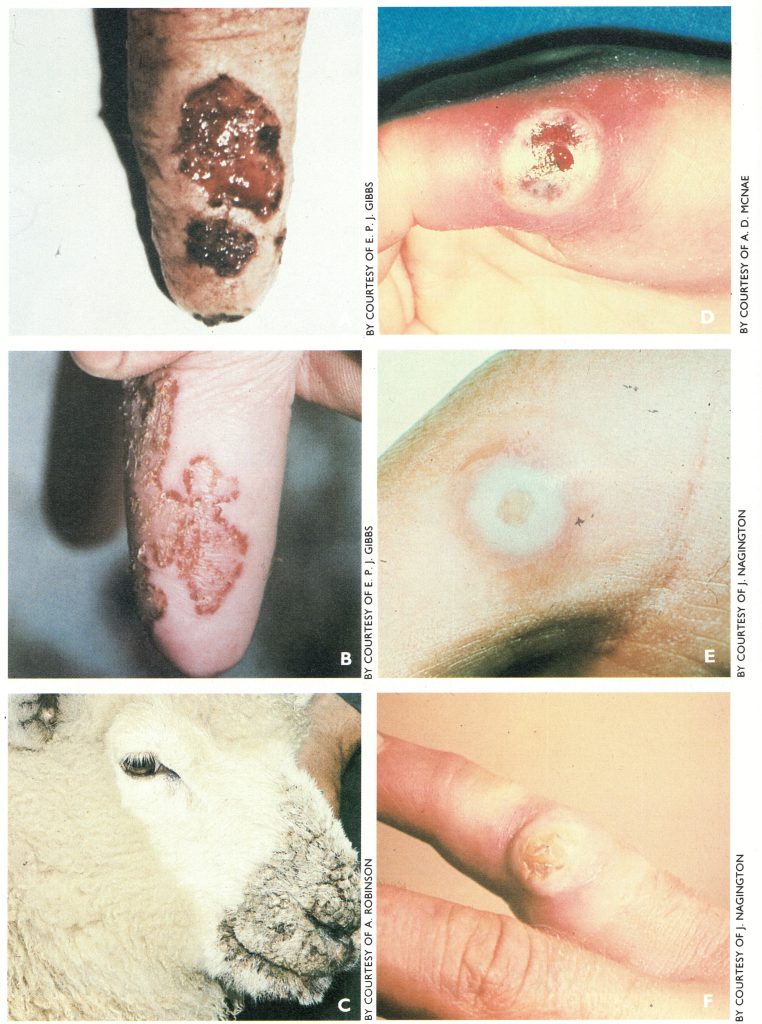
Plate 29.6. Cowpox, pseudocowpox and orf in animals and in humans. A: Cowpox ulcer on teat of a cow, 7 days after onset of symptoms. B: Pseudocowpox (milker’s nodule virus) on teat of a cow. C: Scabby mouth caused by orf virus, in a lamb. Photographs on the right show lesions caused by these viruses on the hands. D: Cowpox. E: Pseudocowpox (milker’s nodule). F: Orf.
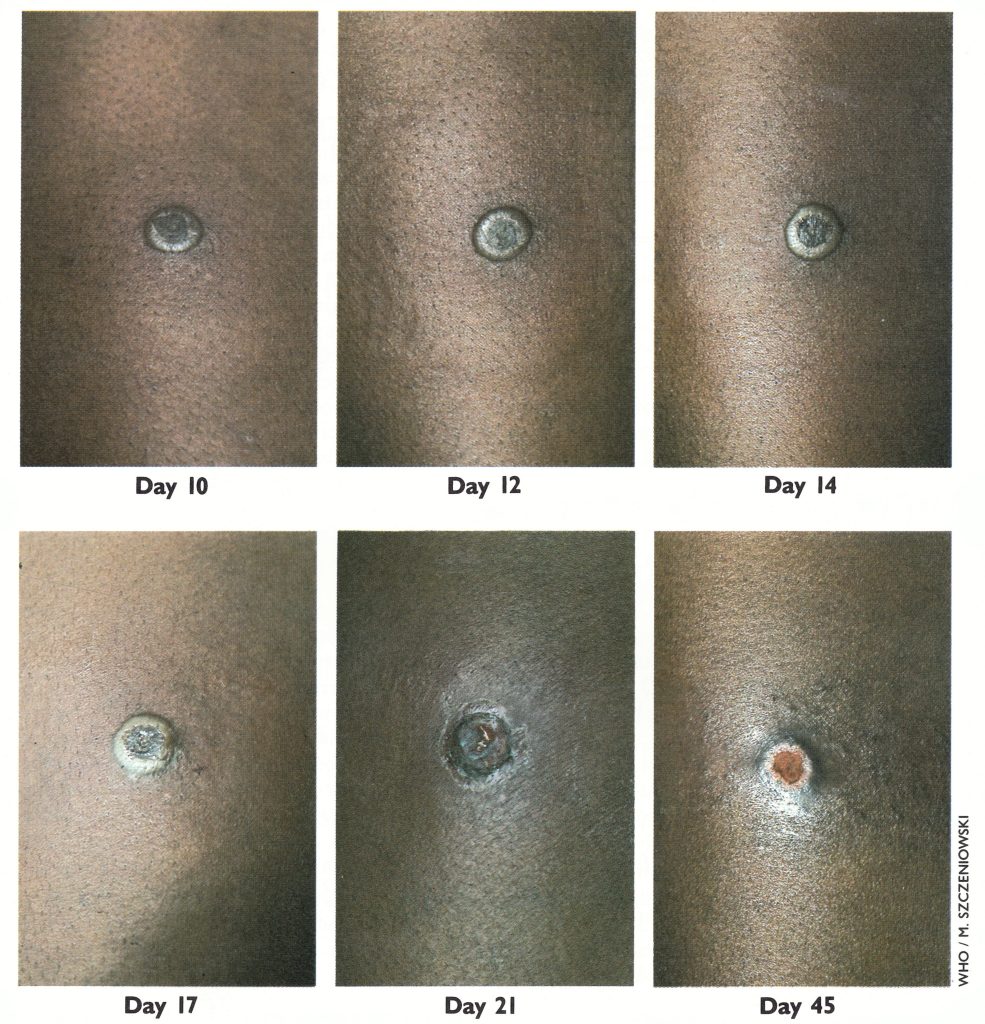
Plate 29.7. Tanapox. Lesion on the thigh of a Zairian woman aged 27 years, 10, 12, 14, 17, 21, and 45 days after its appearance. Note slow progression, pronounced surrounding oedema and erythema during the first 2 weeks, and eventual ulceration and healing.
Epidemiology
Although the clinical features of human monkeypox are very similar to those of discrete ordinary-type smallpox, the epidemiology is quite different. Human monkeypox occurs mainly as single or occasionally multiple sporadic cases, in small villages in dense tropical rain forest in a limited part of Africa, among villagers who are engaged for at least part of their time as hunters and gatherers. Human monkeypox is a zoonosis which is usually contracted from a wild animal. However, human-to-human infection does occur in a minority of cases.
Two observations in the early 1980s deserve comment. Mutombo et al. (1983) reported a bizarre case in which a 6-month-old infant in a small village in the tropical rain forest in Zaire was abducted by a chimpanzee but rescued after sustaining a superficial wound on the lower leg and a fractured femur. The infant developed typical monkeypox, fever beginning 6 days after the incident and a rash 7 days later. Monkeypox virus was isolated from crust material. Lymphadenopathy began in the left inguinal region and 1303 eventually became generalized, but the time of its appearance in relation to other symptoms could not be determined. Although not proved, it is a reasonable hypothesis that the infant acquired monkeypox from the chimpanzee.
The other observation concerns monkeypox among Pygmies who live in the tropical rain forests in the southern part of the Central African Republic, adjoining Zaire, in which Khodakevich et al. (1985) discovered a cluster of 5 cases of monkeypox, confirmed by virus isolation. The Pygmies who lived in the rain forests readily recognized the disease when shown a monkeypox recognition card, whereas the Bantus and Pygmies who lived in agricultural settlements had never seen a disease like it. Interrogation through interpreters revealed that the forest Pygmies had a special name for the disease and believed that it was acquired from animals and not from humans.
Age and sex distribution
The ages of patients in Zaire varied between 6 months and 53 years, but the majority were children. The two sexes were equally affected; of 283 cases reported by the end of 1984, 51.3% were in males and 49.7% in females. When analysing the epidemiology of human monkeypox it is useful to distinguish between infections acquired from an animal source (primary cases) and those due to person-to-person infection (secondary cases) (Table 29.7). The vast majority of cases in both groups occurred in children, but cases in adults tended to be more common among persons infected by contact with other human cases, usually mothers infected by sick children.
Seasonal distribution
Breman et al. (1980) reported a preponderance of cases in Zaire in the dry season, but with the institution of more intensive surveillance since 1982 the incidence of cases has been found to be much the same throughout the year. During the period 1982-1984 the monthly incidence of primary cases varied a good deal from year to year (Table 29.8) ; there was no clearly evident seasonal pattern. Secondary cases showed the same absence of a seasonal effect, although, as with primary cases, the incidence was low in October.
Sources of infection of sporadic cases
Epidemiological investigations in Zaire indicated that wild animals were the probable source of infection for some 70% of patients, and person-to-person infection was suspected in the remaining 30% (see Table 29.8). Since monkeypox virus has a wide host range and evidence of infection in African wild animals has been obtained from chimpanzees, several species of monkey and 2 species of squirrel, the disease is probably transmitted to humans by more than one species of wild animal. It was virtually impossible to determine by case control studies which animals might have been involved because the whole population in affected localities had multiple daily contacts with the same varieties of wild animals, in the settlements, agricultural areas or nearby forests. Species with which patients had multiple close contacts (within 3 weeks before the onset of rash), through hunting, skinning, playing with the animals or eating the carcasses, included various types of monkeys (65%), squirrels (12%), antelopes and gazelles (12%), terrestrial rodents (9%) and other animals (3%). Seventy-one percent of suspected monkeys associated with patients belonged to the genus Cercopithecus, 12% to Colobus, and 8 % to Cercocebus. Two-thirds of suspected rodents were squirrels and the rest were Cricetidae. The majority of animals suspected of being the source of infection were apparently healthy.
The small villages in tropical rain forests, in which cases of human monkeypox occur, are usually not closely surrounded by high forest on all sides. A common situation is that they consist of groups of houses along roads through the forests, with extensive agricultural areas around the settlement itself, consisting of gardens and secondary forest, often with many oil palms, which provide food much favoured by certain squirrels. Beyond this, perhaps 3-5 kilometres away, is the primary rain forest. Each of the 3—zones settlement, agricultural area, and forest-has a characteristic fauna. Domestic animals and commensal rodents frequent the immediate environs of the houses, terrestrial and arboreal rodents and bats are found in the agricultural areas, and larger animals, including monkeys, inhabit the rain forest itself (Khodakevich et al.,1987a).
The various age groups of the population differ in the degree to which they move in and out of these areas. Children below the age of 2 years are rarely let out of their mother’s sight; between the ages of 3 and 5 years they accompany their mothers to the agricultural area, and after the age of 5 years they go on their own to this area and hunt for small animals. Only the men and boys over 15 years of age hunt in the forest for large animals, including monkeys, antelopes and porcupines. Persons of all age groups would be exposed to infection from wild animals brought to the household for food. Those who might conceivably be exposed to an additional risk are the hunters and children old enough to capture small animals such as squirrels and rats in the agricultural areas. Very few primary cases have occurred in hunters, whereas children aged between 5 and 9 years have contracted many primary infections but a somewhat lower proportion of secondary infections (see Table 29.7). This may be related to the relatively high incidence of infection among squirrels captured in the agricultural areas (see below).
Person-to person spread
The largest proportion of cases of monkeypox (48%) have occurred as single sporadic infections . However, sometimes cases have occurred in clusters, suggesting either multiple infections from a common source-co-primary cases-(if the dates of onset lay within the presumed minimum incubation period of 7 days) or person-to-person transmission. The distribution of single sporadic cases, presumed co-primary cases and presumed secondary or subsequent person-to-person infections in Zaire in 1982-1984 is shown in Table 29.9. Intervals of 7 and 23 days between the dates of appearance of the rashes in persons in close family contact have been taken as the limits for presumed person-to-person spread. In the 3 years during which intensive surveillance was operating in Zaire, 66 out of 210 cases (31.4%) appeared to have been due to transmission from person to person . Examples of the type of pattern observed are shown in Fig. 29.3. An extreme example involving 4 probable successive person-to-person infections has been described by Jezek et al. (1986a).
If all these presumed cases of person-to person infection are accepted as such, the secondary and later generation attack rate was 15.7% among unvaccinated household contacts and 0 .6% among vaccinated household contacts. The secondary and later generation attack rate among those having casual face to-face contact with patients was 3%. These figures are much lower than those for smallpox, in which the overall first-generation secondary attack rates in household contacts were 58.4% for unvaccinated persons and 3.8% among vaccinated contacts (see Chapter 4, Table 4.12).
Using these data, which were obtained from a population in which the vaccination rate was about 70%, Jezek et al. (1987b) developed a stochastic model for person-to-person infections with monkeypox virus assuming overall vaccination rates of 50%, 25%, and zero. Although the expected numbers of generations and of cases infected by contact increased with the falling vaccination rate, the model suggested that the person-to-person infectivity of monkeypox was such that the disease always died out, after a maximum number, in the simulation, of 11 generations. This result supports an argument based on historical data—namely, that monkeypox virus has been enzootic in animals of the tropical rain forests in Zaire for centuries without ever establishing continuous person-to-person infection in a population that had been almost completely unvaccinated until about 1967.
The prevalence of monkeypox virus infection in humans
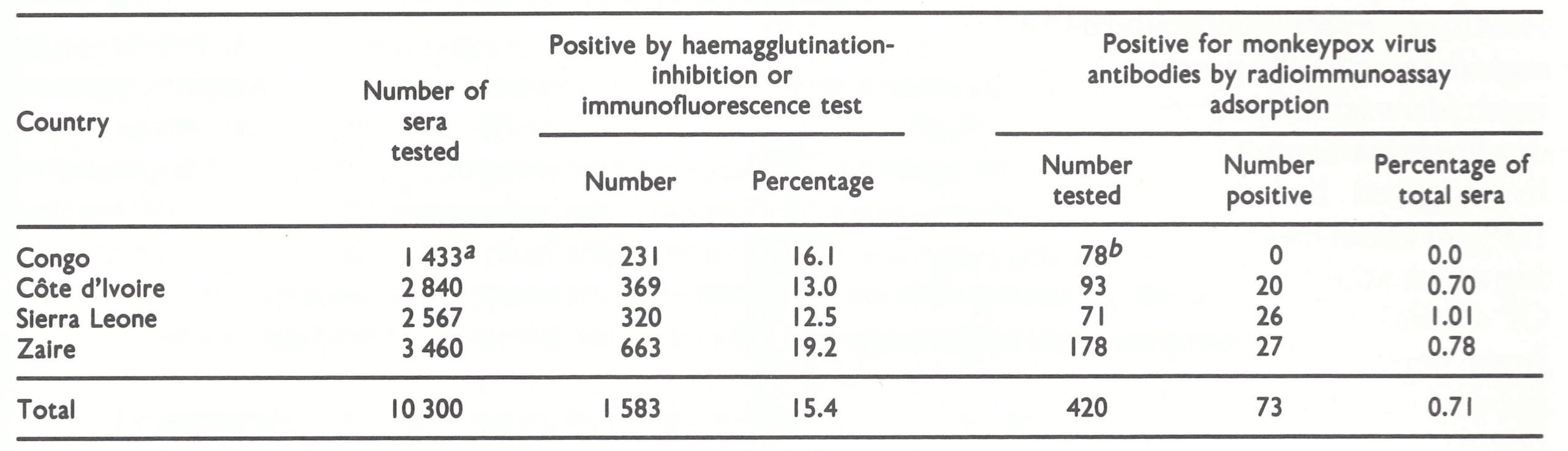
In an attempt to discover the prevalence of monkeypox virus infection of humans in tropical rain forest areas in various parts of central and western Africa, serological surveys of persons without vaccination scars were carried out in 1981 in the Congo and Zaire (central Africa) and Cote d’Ivoire and Sierra Leone (western Africa), according to a plan designed by Arita and Dr Joseph McCormick . Cases of monkeypox had been reported from all these countries except the Congo, which borders on Zaire and has a large area (over 21 million hectares) of tropical rain forest. Specimens of serum collected from allegedly unvaccinated persons were tested in the WHO collaborating centres in Atlanta and Moscow, initially for vaccinia haemagglutination-inhibiting (HI) antibody, or by immunofluorescence at the Pasteur Institute in Abidjan, Cote d’Ivoire.
The results are shown in Table 29 .10. Of 10 300 sera tested, 15.4% gave positive results by HI or immunofluorescence tests.
Supplementary examination of many of these sera by neutralization and ELISA tests showed good agreement with the results obtained with the HI or immunofluorescence test. The intention was to subject sera containing orthopoxvirus antibodies demonstrable by the screening test to further assay by either a radioimmunoassay adsorption test or an ELISA adsorption test. However, only 420 of the 1583 positive sera could be tested; of these, 73 gave results indicating that the subjects had been infected with monkeypox virus. None of the sera from the Congo gave a positive result by the ELISA adsorption test; the proportions of all sera designated as monkeypox-virus-positive varied from 0.70% for Cote d’Ivoire to 1.01% for Sierra Leone.
Follow-up visits by Dr Alexander Gromyko and Dr Jean-Paul Ryst to Cote d’Ivoire and Sierra Leone in June-July 1982 to examine those who had monkeypox virus antibody in their sera showed that some specimens had inadvertently been taken from vaccinated subjects. However, none of the 13 subjects investigated had unequivocal evidence of past vesiculo-pustular disease (by history or residual pockmarks). If any of them had been infected with monkeypox virus, as the serological results indicated, the infection was subclinical or so mild as to have been forgotten.
Table 29.10: Human monkeypox: results of serological survey among allegedly unvaccinated persons inhabiting villages in tropical rain forest areas of 4 countries of central and western Africa, 1981
 a Tested by enzyme-linked immunosorbent assay with monkeypox antigen.
a Tested by enzyme-linked immunosorbent assay with monkeypox antigen.
b Tested by enzyme-linked immunosorbent assay adsorption.

Plate 29.8. Team leaders of special investigations in Kole, outside Kole hospital, Zaire, in 1981. Left to right: K.M. Paluku, M. Mutombo, Okwo-Bele, F.M. Meier, Z. Jezek.
Because surveillance was much better in Zaire, it was possible to obtain more information about the possible frequency of subclinical infection from the survey in Kole Zone, in which Jezek was able to visit some 400 localities, involving about 10 000 households and about 50 000 persons, who were examined for vaccination scars and facial pockmarks (Jezek et al., 1987a). Only 15% of those investigated had no vaccination scar, and 1.3 % of them had facial skin changes suggesting a past attack of a vesiculo-pustular disease. Of a total of 3460 serum samples collected from persons without vaccination scars, 27 showed evidence of the presence of monkeypox-virus-specific antibodies by the radioimmunoassay adsorption test. The subsequent field investigation of 19 of these subjects, who were less than 15 years old, revealed that 12 of them had experienced vesiculo-pustular disease or fever with lymphadenopathy in the past, 1 had a possible vaccination scar, and the remaining 6 had no signs or history of a disease like human monkeypox. The prevalence rate of monkeypox-virus-specific antibodies showed significant differences in different age groups; it was 4 times higher in the children aged 5-9 years (13.1 per 1000) than in those aged 0-4 years (3.3 per 1000).
As with serological surveys among wild animals in Zaire, the lack of a serological test that is sufficiently sensitive and specific to permit the diagnosis of a previous monkeypox virus infection without resorting to serum adsorption has made it impossible to determine the prevalence of human infections with monkeypox virus from the results of these 4 surveys. The significance of the overall orthopoxvirus-positive antibody rate of 15.4% remains obscure; it may have been due to antibodies to vaccinia virus or to a “non-specific reacting material” U. H. Nakano, personal communication, 1986). However, follow-up studies in 3 countries support the view that emerged from intensive surveillance in Zaire (see Table 29.6)—namely, that some infections of unvaccinated humans with monkeypox virus are subclinical.
Ecological Studies
The epidemiology of primary cases of human monkeypox-i.e., those derived from an animal source-can be elucidated only from a knowledge of the ecology of the virus, involving the determination of which animals act as reservoir and incidental hosts and the way in which the virus is transmitted from one animal to another. Initially, studies of this problem were focused on monkeys. Serological surveys of Asian monkeys were negative, but monkeypox-virus-specific antibodies were found in several species of monkeys that occur in central and western Africa.
Because members of each species of monkey usually move in small self-contained troops, and because monkeypox virus does not cause persistent infections and is not transmitted by flying arthropods, it seems unlikely that non-human primates are the reservoir hosts of the virus. From 1979 onwards, therefore, attention was directed to a wider range of wild animals, especially terrestrial and arboreal rodents, some of which occur in populations that remain sufficiently large to support enzootic monkeypox virus infection.
Serological survey of captive African primates
Altogether 1447 sera of African primates held in various laboratories in Africa, Europe and the USA were tested for orthopoxvirus antibodies by either HI or neutralization tests; all were negative (Arita et al.,1972). With the possible exception of sera from 25 gorillas and 167 chimpanzees, all were obtained from animals captured in countries which have not reported cases of monkeypox, and the monkeys belonged to species occurring in the savanna rather than in tropical rain forests.
Serological surveys of primates from western Africa
Breman et al.(1977c) examined primate sera that had been collected in western Africa for a yellow fever survey. HI and neutralization tests were done on 206 sera obtained from 27 different sampling zones in Cote d’Ivoire, Mali and Upper Volta (now Burkina Faso), which were situated in forest and heavily wooded preforest and in the savanna. Out of 195 sera, 15 (8%) were orthopoxvirus positive by HI and 44 (23%) by neutralization tests. The testing of 3 HI-positive sera from forest-dwelling monkeys (1 Colobus badius and 2 Cercopithecus petaurista) by immunofluorescence after adsorption showed that they contained monkeypox-virus-specific antibodies (Gispen et al .,1976).
In another survey, 692 sera obtained from a variety of animals from Chad, Cote d’Ivoire, Liberia, Nigeria, Senegal, Sierra Leone, and Upper Volta between 1970 and 1972 were tested (J . H. Nakano, unpublished observations, 1973). One hundred and fifty-eight (23%) gave positive results by the HI test and 50 out of 186 (27%) were positive by neutralization. Among sera from non-human primates included in the 692 samples, 92 out of 334 (28%) gave positive HI results; 35 out of 147 sera tested by neutralization (24%) gave positive results. Positive HI titres were observed with occasional serum samples obtained from a variety of other animals, including squirrels, rodents, ungulates, and wart-hogs. Subsequently, 273 of the monkey sera were tested by radioimmunoassay adsorption tests. Seven sera contained monkeypox-virus-specific antibodies: 2 from Cercopithecus petaurista, 2 from Cercopithecus aethiops, 2 from Cercopithecus nictitans and 1 from Colobus badius (J. H. Nakano, personal communication, 1986) . The most interesting result was that obtained with C. aethiops from Cote d’Ivoire . Not only is this monkey typically an inhabitant of the savanna rather than the tropical rain forest, but it is the species that was exported from western Africa to North American and European countries on a large scale during the period when monkeypox was occurring in captive monkeys in these countries, and animals of this species may have been the source of infection of Asian monkeys during transit.
Tissues from 648 animals of 73 species obtained in Liberia and Nigeria in 1971 were tested for orthopoxviruses by 2 serial passages in primary monkey kidney cells with negative results J . H. Nakano, personal communication, 1983).
Studies on material from Zaire, 1971-1979
Since most cases of human monkeypox had occurred in Zaire, attempts to determine the reservoir host or hosts of the virus were subsequently concentrated in that country, mostly in places in which human monkeypox cases had occurred.
Investigations by the WHO collaborating centre, Moscow. Between 1971 and 1975 serological and virological investigations concerning a wild-animal reservoir of monkeypox virus were carried out at the WHO collaborating centre in Moscow. Some 200 sera from areas distant from what is now recognized as the monkeypox enzootic area (see Fig. 29.2) were virtually all negative, whereas monkey sera from Zaire collected in 1971 and 1973 showed 14 out of 81 positive by the HI test and 11 out of 65 by the neutralization test (Marennikova et al .,1975). Subsequently another collection of sera from Zaire yielded 24 HI-positive monkey sera out of 117 tested and 26 HI-positive rodent sera out of 245 tested.
Attempts were made to isolate virus on the chorioallantoic membrane from the kidneys of primates, rats, and squirrels collected in Zaire. None yielded monkeypox virus, but “whitepox” virus was said to have been obtained from 4 specimens and vaccinia virus from 1 specimen (see Chapter 30, Table 30 .2).
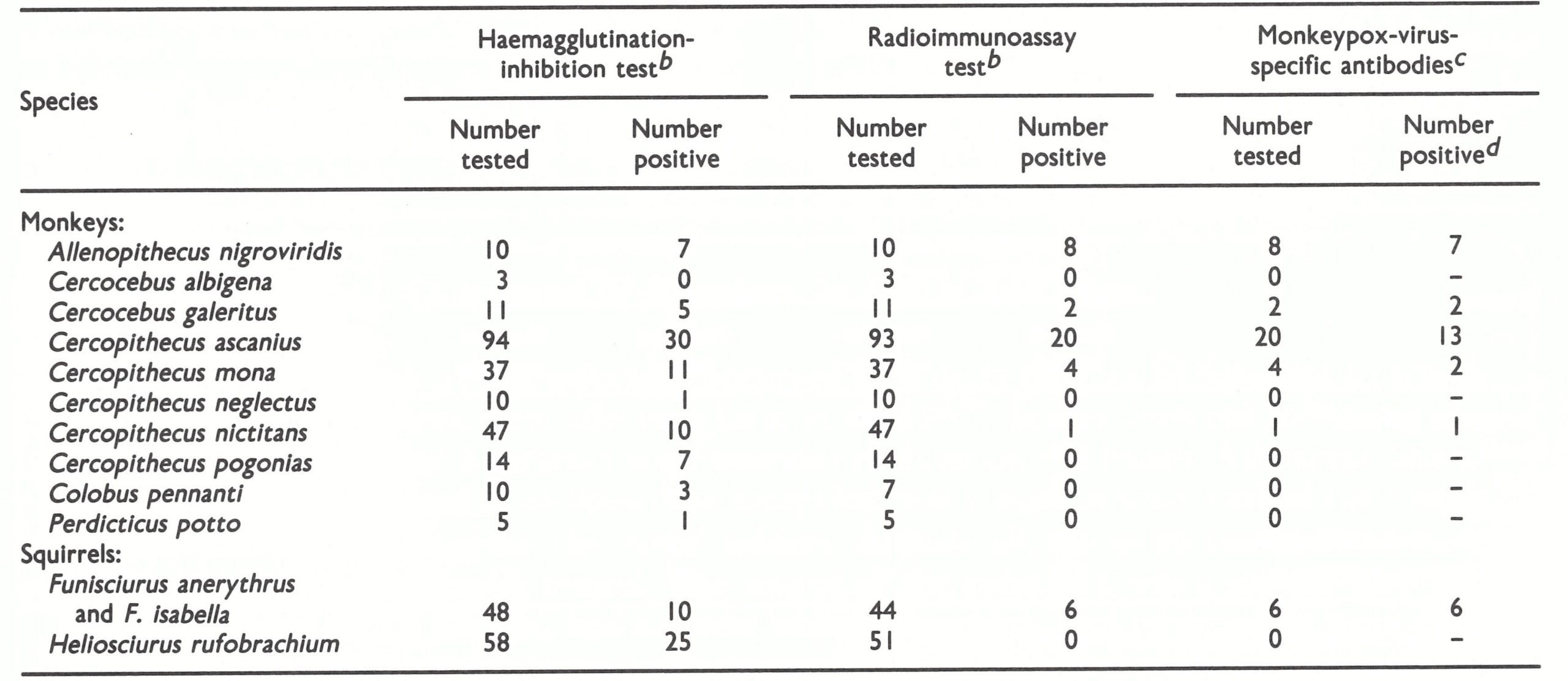
Investigations by the WHO collaborating centre, Atlanta. In July 1979 a large-scale ecological survey in Zaire was organized by Dr Joel Breman, of the WHO Smallpox Eradication unit. Sera and organs were obtained from a wide variety of wild animals. The animal species were identified by expert zoologists and the sera and organs were tested at the WHO collaborating centre in Atlanta. In all, 1331 sera from 45 species of wild animals were tested by the HI test as a screening test for orthopoxvirus antibodies; 227 sera (17%), from a wide range of animals, gave positive results (J. H. Nakano, personal communications, 1983, 1986). All 50 sera from Rattus spp. were negative.
The subsequent testing of certain sera by radioimmunoassay adsorption tests cast doubt on the significance of the positive results obtained by the HI test, since none of the 25 HI-positive sera of the squirrel Heliosciurus rufobrachium gave positive results by radioimmunoassay (Table 29.11). On the other hand, additional radioimmunoassay adsorption tests on monkey and squirrel sera from this collection revealed positive results in 5 species of monkey and in squirrels of the genus Funisciurus (J. H . Nakano, personal communication, 1986).
Kidneys and spleens from 930 of the animals from the 1979 Zaire study, including all the monkeys, were passaged in Vero cells, and the monkey material was also tested on the chorioallantoic membrane, with negative results (J. H. Nakano, personal communication, 1983).
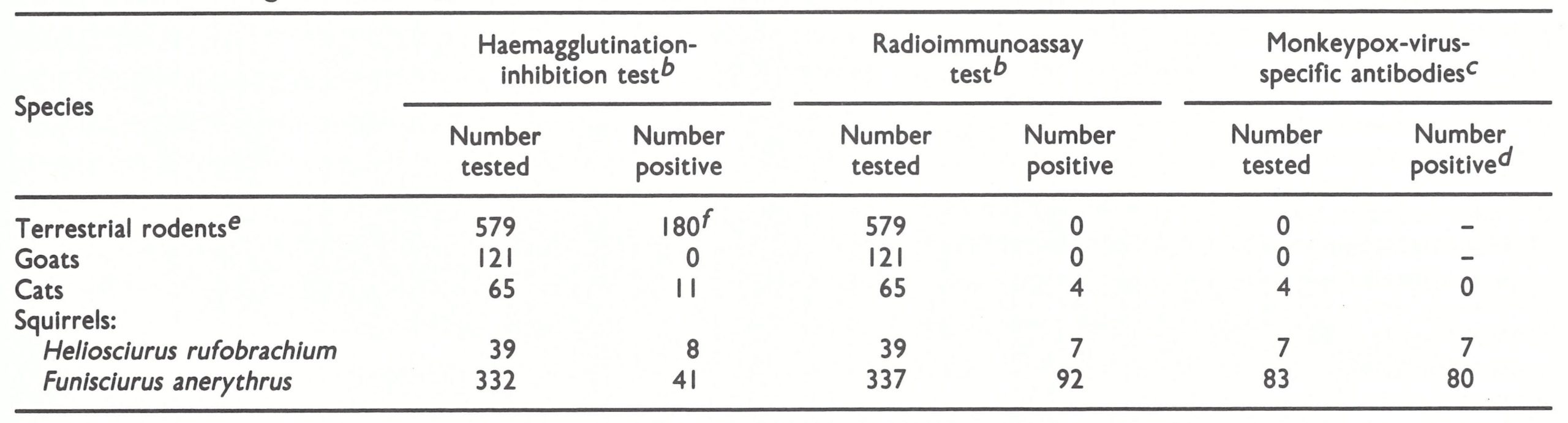
Studies in Zaire, 1985-1986. Ecological investigations in Zaire were renewed in 1985, under the direction of Dr L. Khodakevich. Attention was concentrated on animals found around the houses and in the adjacent agricultural area near villages in which cases of human monkeypox had recently occurred. An early and exciting result was the recovery of monkeypox virus from a diseased squirrel (Funisciurus anerythrus) (Khodakevich et al., 1986). This species of squirrel is quite common in the agricultural areas adjoining villages, where it feeds on oil palm seeds.
Subsequent studies on sera from terrestrial rodents and goats found near houses and squirrels found in the agricultural area revealed many monkeypox-virus-specific sera in 2 species of squirrel (Funisciurus anerythrus and Heliosciurus rufobrachium), but none in the other animals (Table 29.12; Khodakevich et al., 987b). Investigations into the significance of the squirrel, Funisciurus anerythrus, as a possible reservoir host of monkeypox virus are proceeding as this book goes to press.
Table 29.11: Results of haemagglutination-inhibition, radioimmunoassay and radioimmunoassay adsorptiontests on monkey and squirrel sera collected in Zaire in July 1979a
 a Based on unpublished observations by J. H. Nakano.
a Based on unpublished observations by J. H. Nakano.
b Using vaccinia virus antigens.
c By radioimmunoassay adsorption tests.
d Discrepancies between number tested and number positive due to non-specific reacting material.
Table 29.12: Results of haemagglutination-inhibition, radioimmunoassay and radioimmunoassay adsorption tests on sera from animals living in the settlements and agricultural areas adjacent to selected villages in Zaire, 1985-1986a
 a Based on unpublished observations by J.H. Nakano.
a Based on unpublished observations by J.H. Nakano.
b Using vaccinia virus antigens.
c By radioimmunoassay adsorption tests.
d Discrepancies between number tested and number positive due to non-specific reacting material.
e Various species found near houses.
f Non-specific.
MONKEYPOX: THE OVERALL PICTURE
Laboratory studies show that monkeypox virus is a distinct species of Orthopoxvirus. First reported as the cause of epizootics among captive monkeys in laboratory colonies in Europe and the USA and in an epizootic in a zoological garden in the Netherlands, it was found in 1970 to be the causative agent of a generalized human infection that clinically resembled smallpox.
Unlike smallpox, however, human monkeypox occurs only in persons living in small villages in tropical rain forests in central and western Africa, where hunting is an important method of obtaining food. The vast majority of reported cases have been found in Zaire, during an intensive surveillance campaign based on health institutions that has been in operation there since late in 1981. The majority of cases can be attributed to infection from an animal source, but personto-person infection sometimes occurs, mainly between unvaccinated children. The longest chain of transmission observed so far is an incident in which there appeared to be 4 serial person-to-person infections (Jezek et al.1986a). It seems likely that any of several animal species (chimpanzee, several species of monkey, 2 species of squirrel, and perhaps other animals) may serve as the source of human infections.
Even in the parts of Zaire in which it appears to be the most common and is best reported, monkeypox is a rare disease (331 known cases in a population of about 5 million during the 5 years 1982-1986). However, serological studies suggest that occasionally subclinical infections occur among unvaccinated as well as vaccinated persons. There is no reason to believe that it is a new disease or that its frequency is increasing. Indeed, it appears to be disappearing from countries in western Africa, probably because of ecological changes associated with development.
OTHER ORTHOPOXVIRUS INFECTIONS OF MAN
Outbreaks of Vaccinia in Cattle and Man
In 1964 an outbreak of pox infection occurred on a dairy farm in El Salvador in which 22 persons and 450 cows were affected (Lum et al., 1967). It was detected following the admission to hospital of 2 patients with pustular nodules on the hands. All except one of the human cases occurred in milkers; the exception was a woman who washed the towels used by the milkers to clean the cows' udders. Almost all the cows in the herd were infected before the epizootic ceased. The source of the infection was a milker who had been vaccinated on 18 August, had a severe primary reaction, and returned to work on 2 September. The first primary human case occurred 9 days later, presumably via lesions on a cow. Vaccinia virus was recovered from 5 human patients and 1 cow.
As well as being the natural host of variola virus and an occasional, incidental host of monkeypox virus, man is susceptible to 2 other species of Orthopoxvirus, each of which has a broad host range: vaccinia and cowpox. Deliberate vaccination and accidental person-to-person infection with vaccinia virus have been described in Chapter 7. The present chapter is here concerned with human infections with vaccinia and cowpox viruses acquired from animals and with camelpox.
Vaccinia
General
Since vaccination was formerly practised on such a large scale and since vaccinia virus has a broad host range, it is not surprising that domestic animals were sometimes accidentally infected with the virus (Topciu et al .,1976). Human beings could, in turn, be infected from the lesions on domestic animals. Dekking (1964) found that in 36 virologically confirmed outbreaks of “cowpox” in cattle in the Netherlands, 28 were caused by cowpox virus and 8 by vaccinia virus. In the USSR, Maltseva et al.(1966) showed that each of 5 outbreaks of a pox disease affecting cattle and human beings between 1959 and 1963 was caused by vaccinia virus.
Buffalopox
Buffalopox, due to infection of buffaloes with vaccinia virus, was a relatively common disease in India and in other countries in which buffaloes are used for milk production (Lal & Singh, 1977). Usually lesions were confined to the teats of milking buffaloes, but sometimes generalized lesions occurred and calves got lesions on the face and mouth which interfered with their ability to suck. Human infections, usually comprising small lesions on the hands or forearms of milkers, occurred in most outbreaks and acted as the principal mode of transfer of the virus from one buffalo cow to another. The vaccination of milkers was positively incriminated as the source of one outbreak in the USSR (Ganiev & Farzaliev, 1964) and all other outbreaks have occurred in situations in which the infection of the buffaloes could have originated from vaccinated human beings. The causative agent was identified as vaccinia virus in most outbreaks. Baxby & Hill (1971) categorized 1 isolate as a separate species—“buffalopox virus”—on the basis of its biological characteristics, notably a ceiling temperature of 38.5C compared with 41°C for vaccinia virus and the production of smaller pocks on the chorioallantoic membrane and smaller plaques in RK 13 cells. However, analysis of the DNA of this isolate indicates that it is also a strain of vaccinia virus (K. R. Dumbell, personal communication, 1982).
It was assumed that buffalopox would cease to occur after the cessation of routine vaccination in 1979-1980, but outbreaks continue to be reported in Maharashtra State and other parts of India. Analysis of the DNA of viruses recovered from lesions in buffaloes shows that they are strains of vaccinia virus (K. R. Dumbell, personal communication, 1986). These outbreaks do not appear to have been associated with human vaccination; studies on their epidemiology are in progress as this book goes to press.
Cowpox
History and geographical distribution
The history of cowpox in relation to the origins of Jennerian vaccination has been described in Chapters 2 and 6. It was not until 1939 that Downie (1939a,b) clearly differentiated cowpox virus from vaccinia virus. Classical cowpox has not been described outside of Europe, but strains of cowpox virus have been recovered from rodents in Turkmenia (USSR).
Epidemiology
The traditional mode of infection of human beings with cowpox virus was by “inoculation” of the hands of milkers by contact with ulcers on the teats of cattle caused by cowpox virus (see Plate 29.6 A and 29.6 D). This was undoubtedly the usual mode of infection and over the years many such episodes have been reported. Human cowpox was regarded as a rare zoonosis, contracted by the direct contact of milkers with lesions on the teats of cows and resulting in an ulcer or ulcers that remained localized at the inoculation site (Downie, 1951, 1965a ; Dekking, 1964).

Plate 29.9. Derrick Baxby (b. 1940), British authority on orthopoxviruses, with a particular interest in cowpox and the history of vaccination.
Bovine cowpox is not a common disease (Gibbs et al ., 1973) and apparently never was, even in Jenner’s time, and the occurrence of lesions of “spurious cowpox” on cows’ teats (see below) gave rise to much confusion when such lesions were used as a source of vaccine. Ceely (1842), who provided one of the best and most detailed descriptions of cowpox in bovines, noted that: “The disease is occasionally epizootic . . . more commonly sporadic or nearly solitary. It may be seen sometimes at several contiguous farms, at other times one or two farms entirely escape its visitation. Many years may elapse before it recurs at a given farm or vicinity, although all the animals may have been changed in the meantime.”
Baxby (1977a; personal communication, 1983) has pointed out that cows were directly implicated as a source of cowpox virus in only 3 out of 16 virologically confirmed cases in human beings in England between 1969 and 1981 (Table 29.13). No source of infection could be discovered for the other 13 cases. Only 4 of the cases occurred in farm workers. Other studies (review: Baxby et al.,1979) have shown that cowpox virus (defining the species according to the biological characteristics described in Chapter 2, Table 2.3) has caused sporadic infections in domestic cats, large felines, elephants, okapis and a rhinoceros (Table 29.14), none of these infections having originated from contact with cases of bovine or human cowpox. The validity of the species diagnosis is supported by analyses of the DNA obtained from several of these isolates (Fig. 29.4). All could be clearly differentiated from vaccinia and variola DNA. The dissimilarities between different strains of cowpox DNA relate in a general way to their geographical origins, and strains from unusual hosts (elephants and large felines) clearly have cowpox virus DNA.

Fig. 29.4. Dendrogram illustrating the similarities and differences between HindIII, Xhoi and Smal cleavage sites on the DNAs of 8 strains of cowpox virus,2 strains of variola virus and 2 strains of vaccinia virus, Analysis as fig. 29.1. Number of attributes= 70. (Data from Mackett, 1981.)

Plate 29.10. Reservoir hosts of cowpox virus in Turkmenia. A: Yellow suslik (Citellus fulvus). B: Great gerbil (Rhombomys opimus).
Man, cows and the other animals listed in Table 29.14 are probably all incidental hosts of cowpox virus, of no importance as far as its perpetutation in nature is concerned. The recovery of cowpox virus from wild rodents in Turkmenia (Ladnyj et al .,1975; Marennikova et al.,1978b; Plate 29.10) and the demonstration that a substantial number of them were serologically positive (Table 29.15) suggests that susliks and gerbils might be natural reservoir hosts of cowpox virus in Turkmenia. These animals do not occur in the United Kingdom, but Kaplan et al.(1980), in a study of virus infections in small British field rodents, demonstrated orthopoxvirus antibodies in wild voles; these could be due to cowpox virus. It is not unreasonable to suppose that cowpox virus, which has a wide host range, produces enzootic infections in a variety of rodents, from which it is occasionally transferred to other animals cows, cats, zoo animals (possibly via domestic rats used as feed, as in the Moscow Zoo outbreak ; Marennikova & Shelukhina, 1976) and sometimes man (Baxby, 1977a, 1982b). In turn, cows, cats and sometimes zoo animals (Marennikova et al.,1977) could serve as the source of infection for humans.
Differential diagnosis of lesions on cows’ teats
Jenner recognized that not all ulcers on the teats of dairy cows were caused by “variolae vaccinae”, so that material taken from such lesions sometimes lacked the capacity to protect humans against smallpox; he termed such lesions “spurious cowpox” (Jenner, 1799). In addition to cowpox and vaccinia virus, 2 other viruses can cause ulcers on the teats of cows (Gibbs et al., 1970; Baxby, 1981). These are bovine herpes mammillitis virus and pseudocowpox virus (Plate 29.6 B). Both are enzootic diseases of bovines, and are much more common than cowpox virus infections in dairy herds. Pseudocowpox virus is transmissible to man, to produce milker’s nodules (see below).
Clinical features of cowpox in man
Downie (1965a) has described the lesions found in humans infected with cowpox virus (Plate 29.6D) as follows. One or more lesions usually appear on the hands—the thumbs, the first interdigital cleft and the forefinger being especially liable to attack. Scratches or abrasions of the skin may determine the localization of the lesions elsewhere on the hands, forearms or face. The lesions resemble those of primary vaccination, passing through the stages of vesicle and pustule before a scab forms. Local oedema is usually more pronounced than in vaccination and there is lymphangitis, lymphadenitis and often fever for a few days. Baxby (1977a) noted that cowpox in children was occasionally rather severe. However, although multiple primary lesions sometimes occur, a generalized rash has not been reported, but one case of post-cowpox encephalitis has been described (Verlinde,1951).
Camelpox
Camelpox is a common disease of dromedary camels. The original report identifying camelpox virus as an orthopoxvirus (Baxby, 1972) caused some concern to those involved in the global smallpox eradication campaign since it was entitled “Smallpox-like viruses from camels in Iran”. However, subsequent investigations (see Chapter 2) showed that it was caused by a distinct species of Orthopoxvirus that has a narrow host range. Among camels, skin lesions occur mainly on the head, neck and forelegs, or all over the body. Young animals, in particular, may suffer a severe disease which is sometimes fatal (Plate 29.11). Camelpox is enzootic in Somalia (Je2ek et al .,1983) and in most other areas in which camels are common (Egypt, India, the Islamic Republic of Iran, Iraq, Kenya and the USSR) but not among feral camels in Australia.
Although there were occasional reports in the older literature that camel drivers could contract local lesions on the hands and arms from contact with affected animals, the experience during the global smallpox eradication programme, especially in Somalia, suggested that human camelpox rarely if .ever occurred. Kf1 (1982) described a possible case in a 40-year-old unvaccinated Somali man who was a member of a nomadic group among whose camels there was a severe epizootic of camelpox. There were 3 lesions on the left arm and 1 on the right, which went through vesicular and pustular stages before scabbing. It was not possible to obtain lesion material for laboratory confirmation, but the serum from this patient gave a positive orthopoxvirus HI test. A survey among 286 camel herdsmen in the area, only one-third of whom had been vaccinated, revealed only 2 other cases of skin lesions, both diagnosed as tropical ulcers. A subsequent survey of another 179 herdsmen handling affected camels, 12% of whom had been vaccinated, revealed few skin eruptions, none of which yielded a poxvirus (Jezek et al .,1983). Out of a total of 335 specimens taken from the skin lesions of persons who might have come into contact with diseased camels, none was positive for poxvirus particles, and inquiries among some 20 000 persons at risk yielded only 1 possible case of human camelpox, that reported by Kfi2 (1982).
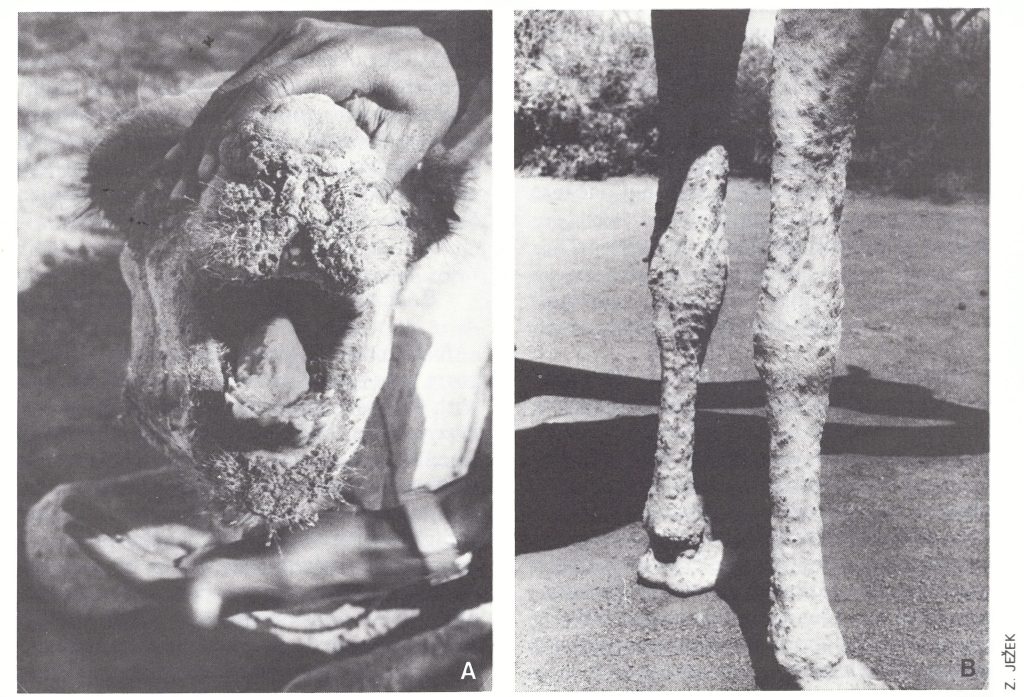
Plate 29.11. Camelpox in camels in Somalia. A: Thick brown crusts around the mouth and lesions on the tongue. B: Generalized lesions.
PARAPOXVIRUS INFECTIONS
A number of domestic—animals, goats, cattle, and camels—sustain infections with different strains or species of the genus Parapoxvirus. The lesions in each species of animal usually take the form either of scattered papules and nodules in the skin or of a papular stomatitis, with lesions on the lips and gums (“scabby mouth” of sheep; bovine papular stomatitis). One strain of parapoxvirus is spread among cows and produces ulcerative lesions on the teats (Plate 29.6B), which are called pseudocowpox and constitute one of the forms of the “spurious cowpox” of Jenner (Gibbs & Osborne, 1974). Calves sucking from dams with pseudocowpox usually get lesions on the mouth and lips . Bovine papular stomatitis is caused by a different species of parapoxvirus and is found more commonly in beef than in dairy cattle (Tripathy et al .,1981). Five out of 57 cases of “camelpox” in Somalia that were investigated virologically in 1978-1979 were caused by a parapoxvirus (J. H. Nakano, personal communication, 1986).
Humans can be infected accidentally with these parapoxviruses through abrasions of the skin. The disease acquired from sheep or goats is termed orf (review: Johannessen et al .,1975) ; that acquired by milkers from the ulcers on the teats of cattle is called milker’s nodules. The lesions in cattle and sheep often ulcerate ; milker’s nodules in humans are usually small indolent papules (Plate 29.6E). Human orf is associated with umbilicated proliferative lesions that often ulcerate before healing (Plate 29.6 F).
All parapoxviruses have an identical morphology, which is quite distinctive (Plate 29.12), the virions being smaller than those of the orthopoxviruses and having a regular surface structure.
Parapoxvirus infections are of some importance in the consideration of smallpox for two rather trivial reasons; the lesions on cows’ teats constituted an early source of confusion with genuine cowpox, and the particles found in scrapings of human lesions reported by electron microscopists simply as “poxvirus particles” might unnecessarily alarm public health authorities.
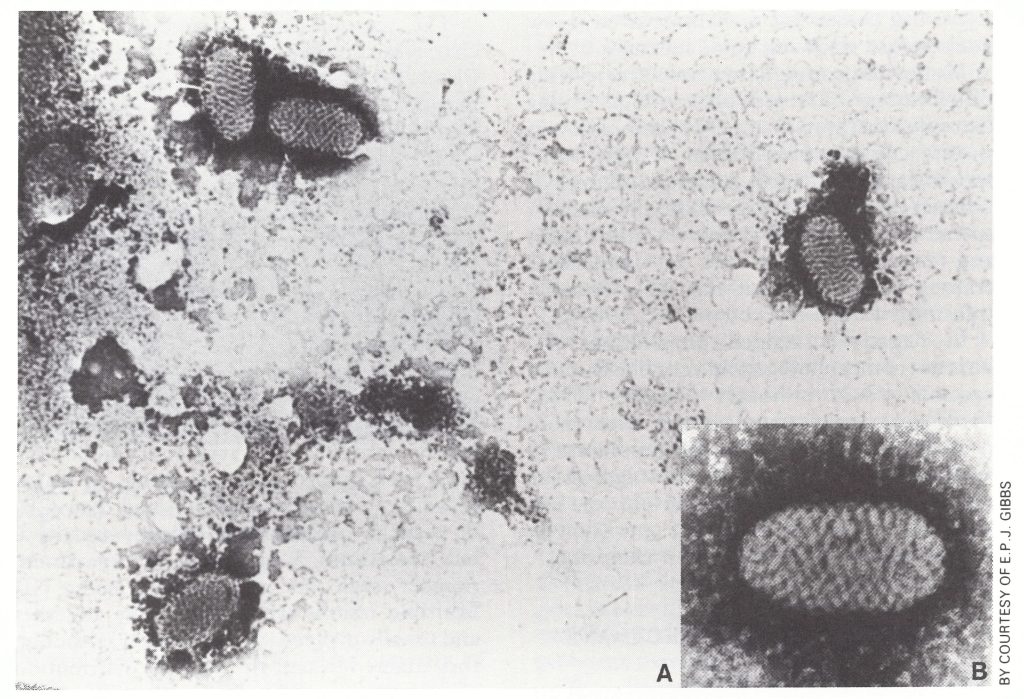
Plate 29.12. A: Virions of the parapoxvirus of pseudocowpox, which produces lesions of milker’s nodules in humans. B: Virion at higher magnification, showing the regular spiral structure of the tubule of the outer coat, which is characteristic of the genus Parapoxvirus.
MOLLUSCUM CONTAGIOSUM
Molluscum contagiosum is a specifically human skin disease caused by a poxvirus which has not yet been cultivated or transmitted to laboratory animals (review: Postlethwaite, 1970). The lesions are pearly, flesh coloured, raised, firm, umbilicated skin nodules, 2-5 mm in diameter, which may appear anywhere on the body except the palms and soles. The nodules are painless and at the top of each there is often an opening through which a small white core can be seen . There are no constitutional disturbances. The lesions may persist for months or even a few years before resolving spontaneously.
Molluscum contagiosum has a world-wide distribution. In some places—e.g ., Papua New Guinea (Sturt et al.,1971) and Zaire—it is very common in children. It may occur sporadically or in small epidemics. Direct or indirect contagion appears to be the mode of spread and in western countries public baths and swimming-pools may be implicated. Among young adults it may be a sexually transmitted disease (Brown et al ., 1981).
In negatively stained preparations the virions are morphologically very like those of vaccinia virus, although Nakano (1985) noted that the surface tubules were more prominent. However, since the lesions are so distinctive, and since the virus cannot be cultivated, molluscum contagiosum was not regarded as a serious source of confusion in the global smallpox eradication campaign.
TANAPOX VIRUS INFECTIONS
Tanapox was first observed as an acute febrile illness, associated with localized nodular skin lesions and caused by a poxvirus, which occurred in epidemics in 1957 and 1962 among people living in the flood plain of the Tana river in Kenya (Downie et al .,1971). Serological studies (Manson-Bahr & Downie, 1973) showed that it was endemic in this area, and subsequently many cases were seen during surveillance for monkeypox in Zaire in 1977-1984 (Jezek et al ., 1985). The same virus (Downie & Espana, 1972) gave rise to epizootic infection in rhesus monkeys in 3 primate centres in the USA in 1966; in each of these outbreaks some of the animal handlers were infected, apparently through skin abrasions (Nakano, 1978).
Jezek et al. (1985) have published a detailed analysis of the clinical and epidemiological features of tanapox as seen in 264 cases in Zaire in which the diagnosis was confirmed by electron microscopy. The incubation period in natural human cases is unknown, but in a person infected by the intradermal inoculation of about 104 infectious particles (as assayed in tissue culture), erythema and central thickening appeared by the 4th day (Downie et al.,1971). Most patients have a mild pre-eruptive fever, sometimes accompanied by severe headache and backache and often with itching at the site of the eventual skin lesion.
The appearance and evolution of the characteristic skin lesions are illustrated in Plate 29.7. Initially there is a small nodule, without the central abrasion that is often seen with an insect bite. The nodule soon becomes papular and gradually enlarges to reach a maximum diameter of about 15 mm by the end of the second week. It is surrounded by an oedematous zone and a large erythematous areola. The draining lymph nodes are enlarged and tender from about the 5th day after the appearance of the skin lesion, which may remain nodular but usually ulcerates during the third week and then gradually heals within 5-6 weeks, leaving a scar. In Kenya, Downie et al. (1971) noted that the lesions were almost always solitary and on the face, neck, upper arm and trunk. In Zaire, however, Jezek et al. (1985) noted that 22% of patients had multiple lesions-usually 2 but sometimes 3 or more, the maximum number seen on one patient being 10. Multiple lesions were often close together and usually evolved simultaneously, although they differed in size. In Zaire the distribution of lesions was different from that seen in Kenya, 72% being on the lower limbs, 17% on the upper limbs, 7% on the trunk and 5 on the head.
Especially if there were multiple lesions, a case of tanapox could initially be mistaken for human monkeypox (or in former times smallpox), perhaps modified because of vaccination, but the slow evolution and lack of pustulation clearly differentiate tanapox from any of the orthopoxvirus infections. The clinical diagnosis can be confirmed by the demonstration with the electron microscope of poxviruses which have a characteristic envelope (Plate 29.13) and fail to grow on the chorioallantoic membrane (Nakano, 1985).
Some strains of tanapox virus grow in cultures of monkey or human cells, producing focal lesions characterized by intense granularity followed by rounding up of the cells. Monkeys, but no other laboratory animals, are susceptible to experimental infection.
Human tanapox has been recognized in Kenya and Zaire, but probably occurs much more widely throughout tropical Africa. In Kenya, Downie et al. (1971) noted that epidemics in 1957 and 1963 were associated with periods of extensive flooding. In Zaire, cases occurred throughout the year but mainly in the period between November and March (Jezek et al.,1985). The majority of cases seen in Zaire were found in the township of Lisala, among persons living within 300 metres of the Zaire river. Both sexes and all age groups were affected, and cases occurred much more frequently among persons who worked or played close to the river than among those engaged in hunting or working as plantation farmers. Although clusters of cases occurred both temporally and spatially, there was no indication that person-to-person spread occurred. Tanapox appears to be a zoonosis, but neither the reservoir host nor the mode of transmission from wild animals to man is known. Manson-Bahr & Downie (1973) suggested that tanapox virus may be transferred from monkeys or some other reservoir host to man by biting arthropods, possibly acting as mechanical vectors. Infection by mechanical transmission has been described among animal attendants (McNulty et al ., 1968).
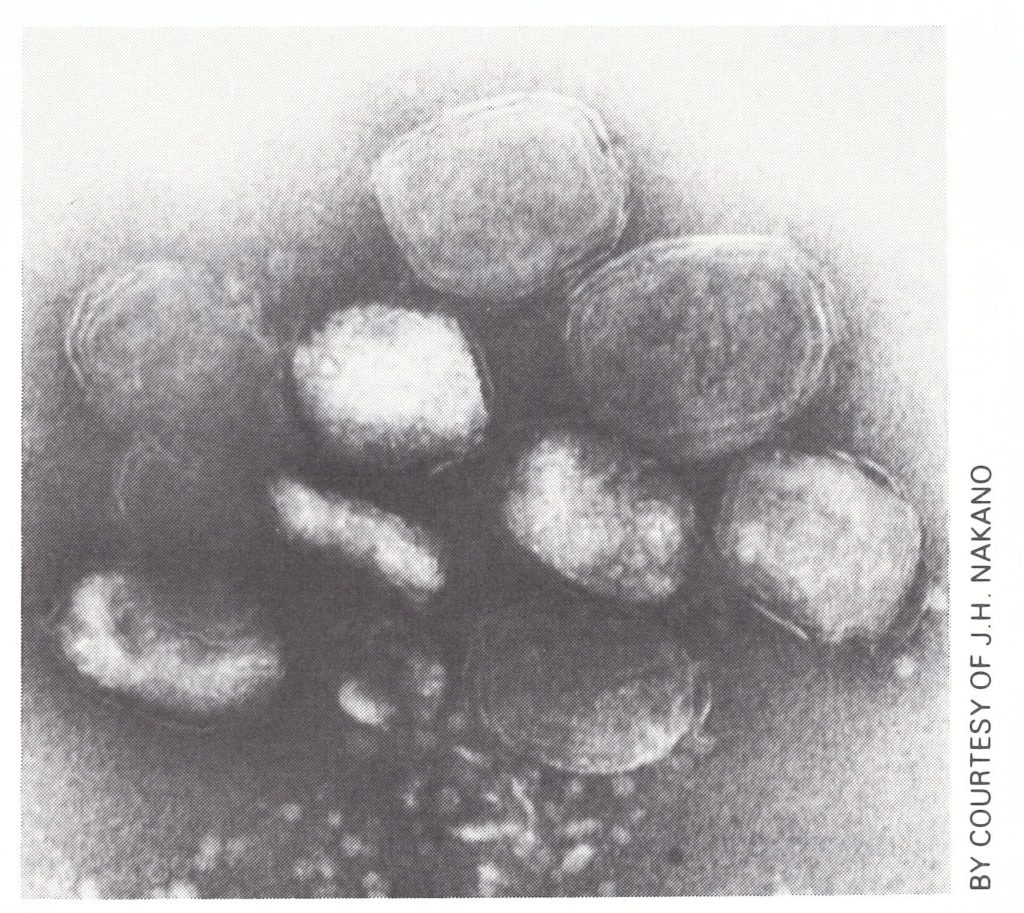
Plate 29.13. Virions of tanapox virus, as seen in negatively stained scrapings from a lesion. Most virions appear to have an envelope.
GENERAL COMMENT
Man is susceptible to a range of poxvirus infections, but only two of these, smallpox and human monkeypox, regularly produce an acute systemic infection with a generalized rash. Human monkeypox can be distinguished from smallpox only by the cultivation of the virus or the performance of a virus-specific serological test with convalescent serum, but the epidemiology of the two infections is quite different. Monkeypox was the only poxvirus infection other than smallpox seen during the eradication programme that gave rise to serious concern. However, the studies in Zaire described in this chapter provide good evidence that it is a rare zoonosis which cannot be sustained indefinitely by serial transmission in man.
Because vaccination can greatly modify the response of humans to either variola or monkeypox virus, so that if skin lesions do occur, they are very few or perhaps only a solitary one develops, other poxvirus infections sometimes cause problems in the differential diagnosis of smallpox or monkeypox, especially because electron microscopic examination of lesion material might reveal poxvirus particles.
The diagnosis of cowpox and vaccinia infections depends on the cultivation of the virus on the chorioallantoic membrane; that of tanapox on the clinical picture, the appearance of the virion in the electron microscope and its failure to grow on the chorioallantoic membrane. The lack of systemic symptoms and the characteristic chronic nodular skin lesions distinguish molluscum contagiosum from all other poxvirus diseases. The viruses of orf and milker’s nodules can readily be distinguished by the characteristic morphology of the virion, as well as by serological tests.
When they were first studied in the laboratory, camelpox virus (Baxby, 1972) and taterapox virus (Lourie et al .,1975) were regarded with considerable suspicion because the pocks they produced on the chorioallantoic membrane very closely resembled those produced by variola virus. It is possible that similar causes of concern may arise, perhaps with orthopoxviruses of wild animals that have yet to be discovered. Comparison of their DNA with that of variola and monkeypox viruses should allow the proper categorization of any such isolates.


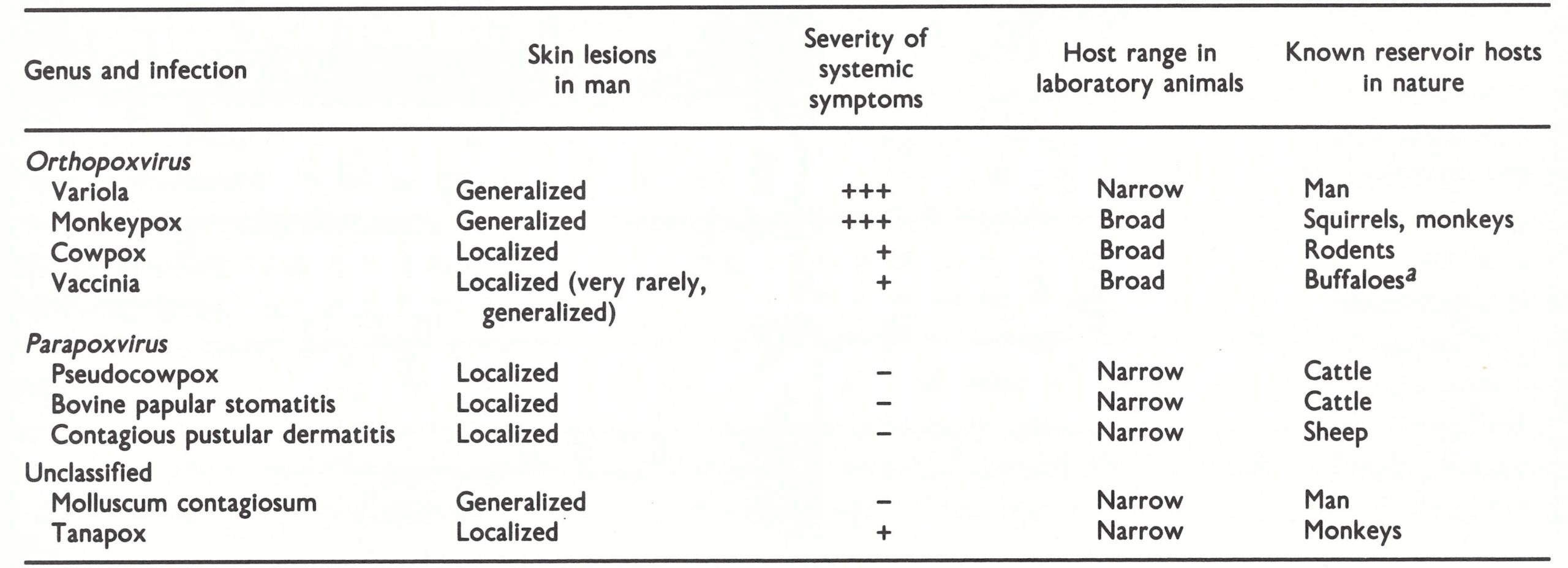
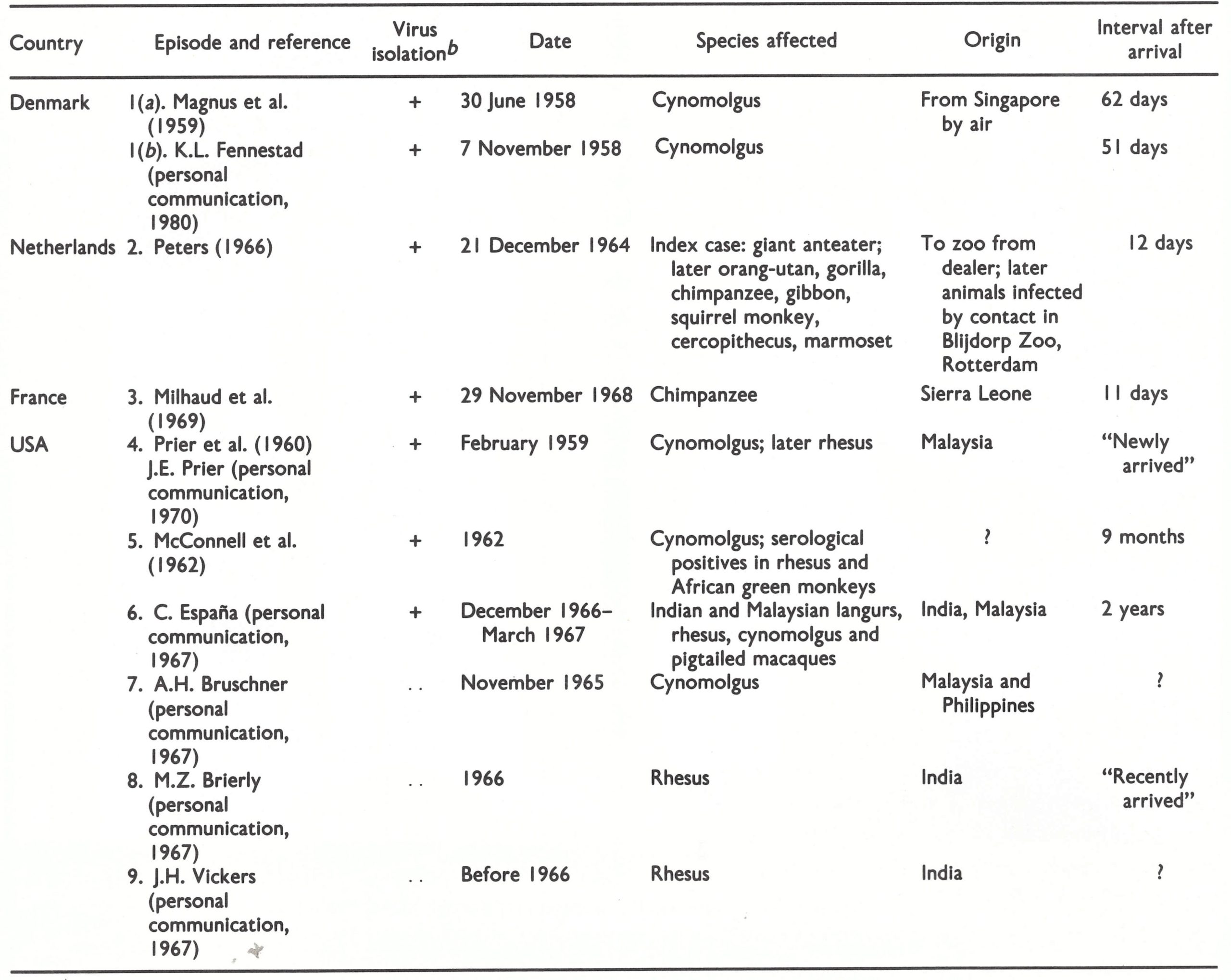
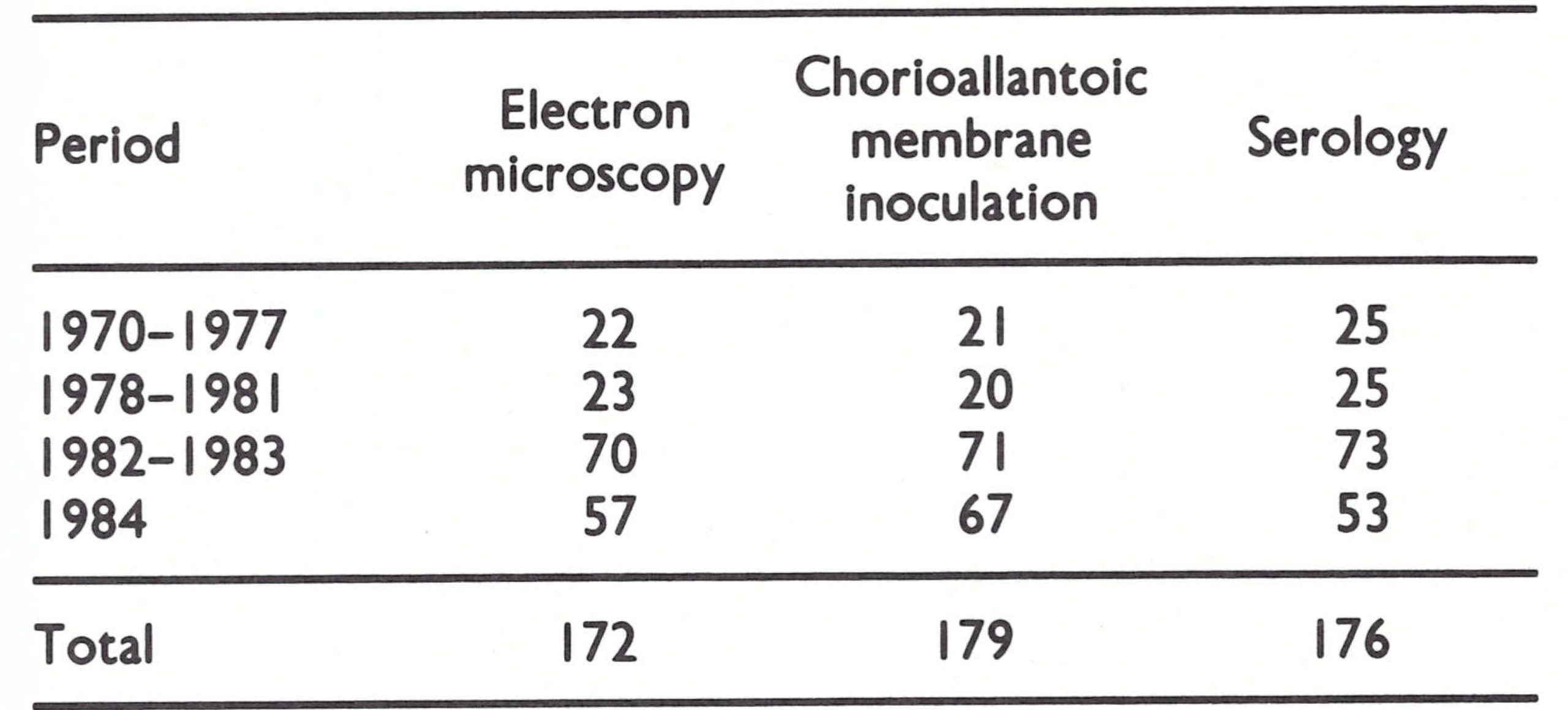
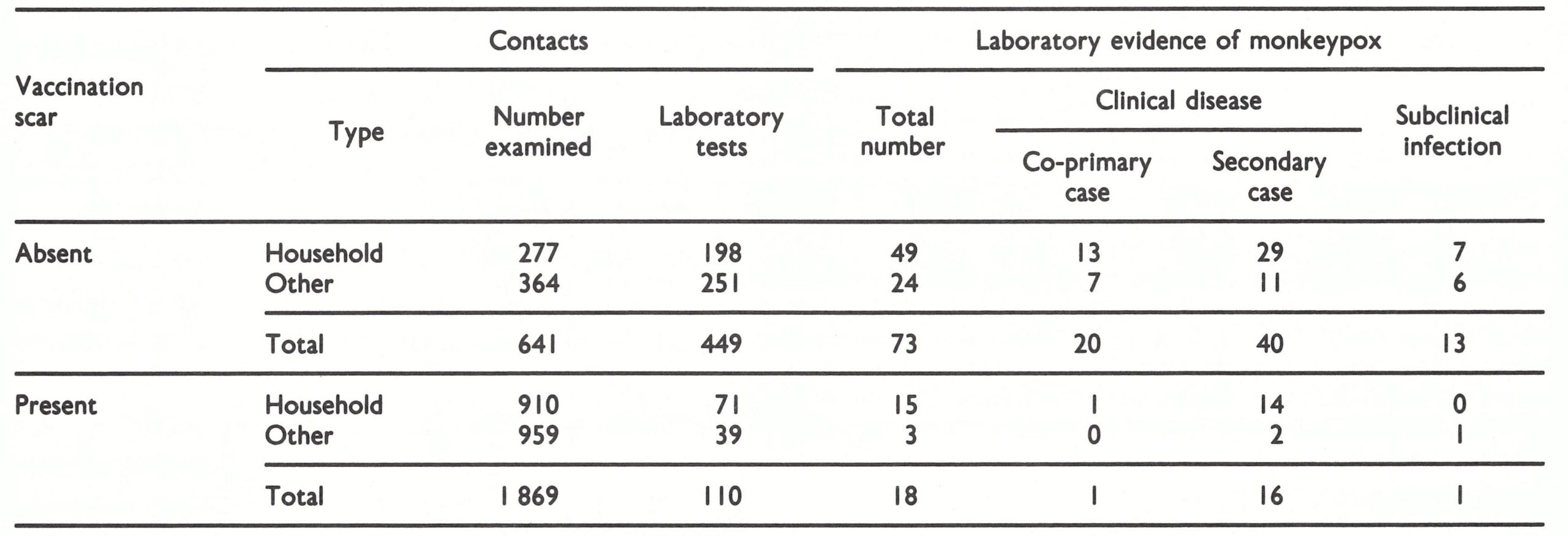
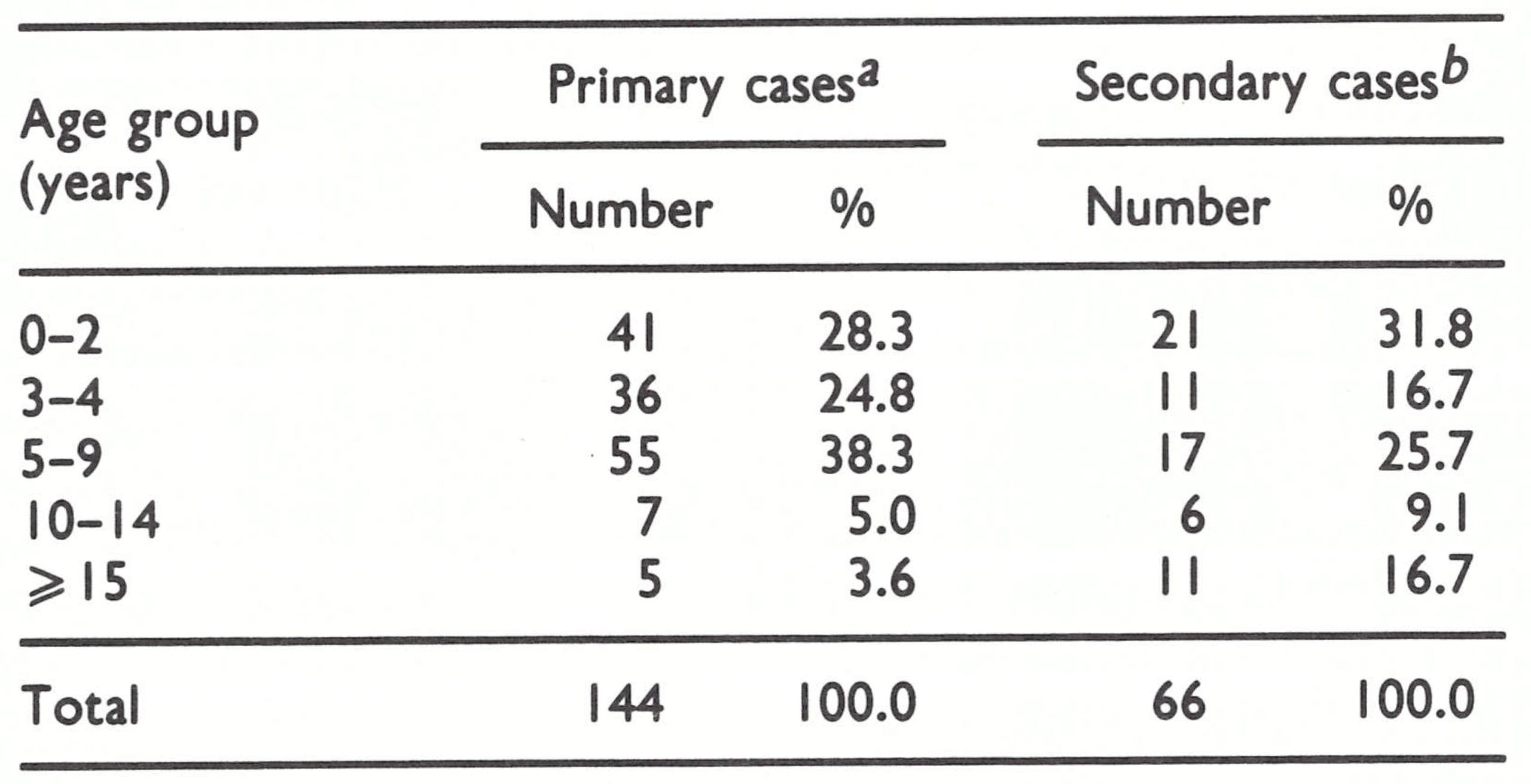
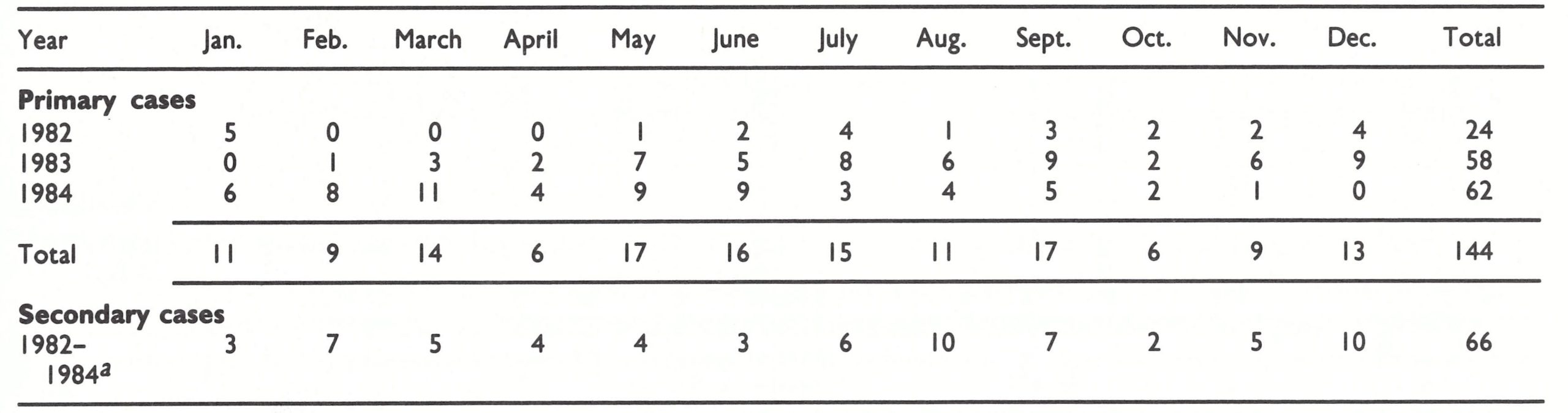
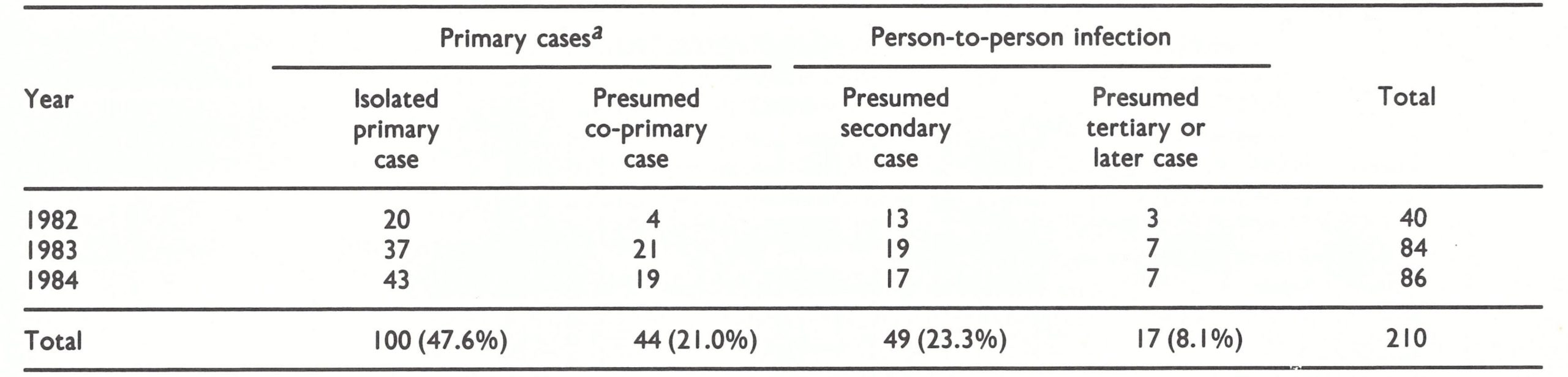

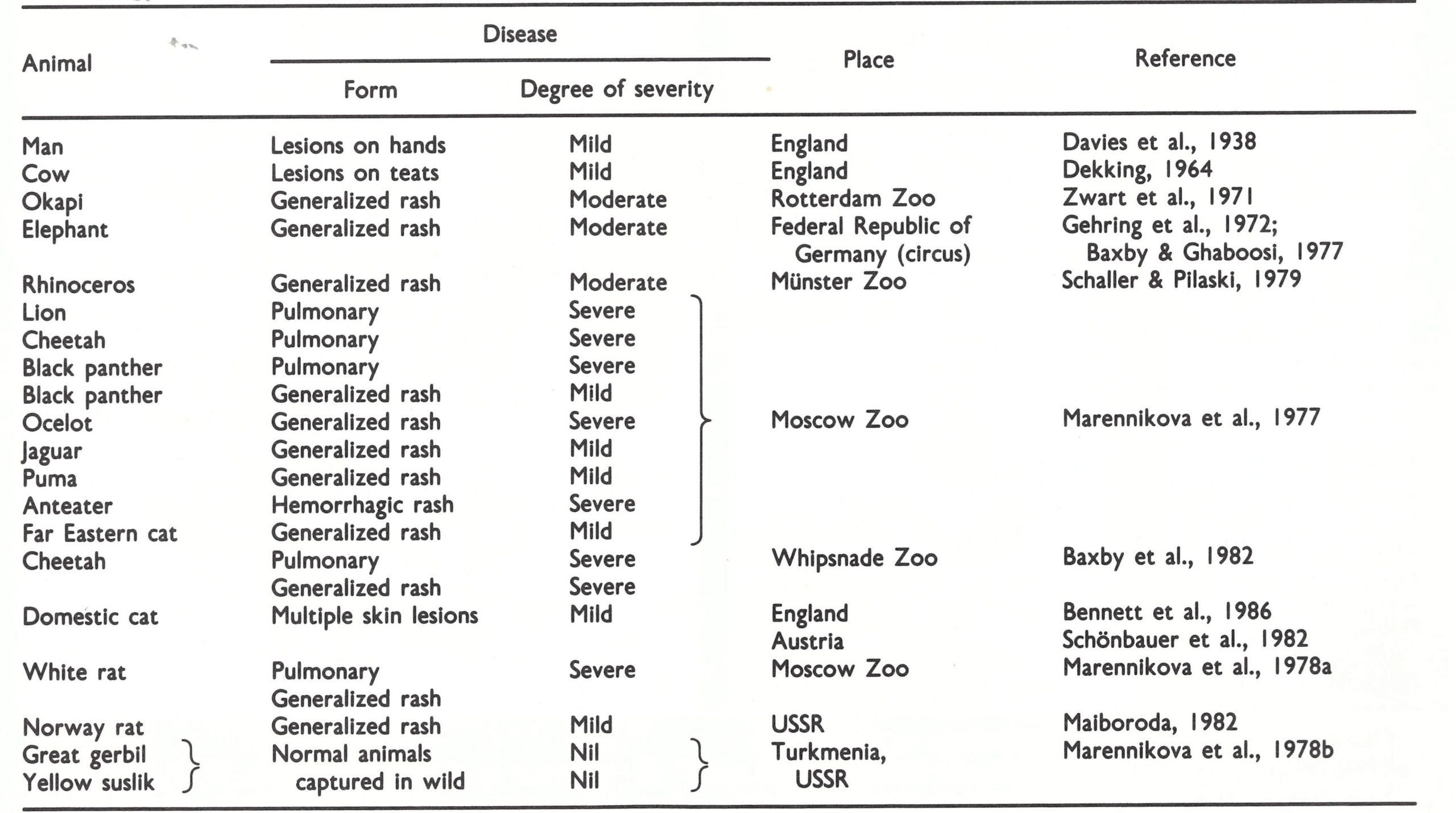

Robertbor
July 3, 2025Хотите собрать данные о пользователе? Этот бот предоставит полный профиль в режиме реального времени . Воспользуйтесь уникальные алгоритмы для анализа публичных записей в соцсетях . Выясните контактные данные или интересы через систему мониторинга с гарантией точности . глаз бога поиск по телеграм Бот работает в рамках закона , обрабатывая общедоступную информацию. Получите детализированную выжимку с геолокационными метками и графиками активности . Попробуйте надежному помощнику для digital-расследований — результаты вас удивят !
KennethLobre
July 5, 2025Выгребная яма — это водонепроницаемый резервуар, предназначенная для сбора и частичной переработки отходов. Принцип действия заключается в том, что жидкость из дома поступает в бак , где твердые частицы оседают , а жиры и масла собираются в верхнем слое. Основные элементы: входная труба, бетонный резервуар, соединительный канал и почвенный фильтр для дочистки воды . http://midoma.ru/node/kupit-septik-v-moskve-nedorogo Преимущества: экономичность, минимальное обслуживание и безопасность для окружающей среды при соблюдении норм. Критично важно не перегружать систему , иначе неотделённые примеси попадут в грунт, вызывая загрязнение. Типы конструкций: бетонные блоки, полиэтиленовые резервуары и композитные баки для разных условий монтажа .
rolex-submariner-shop.ru
July 19, 2025Rolex Submariner, представленная в 1953 году стала первой дайверской моделью, выдерживающими глубину до 100 метров . Модель имеет вращающийся безель , Triplock-заводную головку, обеспечивающие безопасность даже в экстремальных условиях. Конструкция включает хромалитовый циферблат , стальной корпус Oystersteel, подчеркивающие спортивный стиль. rolex-submariner-shop.ru Автоподзавод до 3 суток сочетается с перманентной работой, что делает их надежным спутником для активного образа жизни. За десятилетия Submariner стал символом часового искусства, оцениваемым как эксперты.
DavidHam
August 2, 2025Этот сайт предлагает актуальные новости в одном месте. Здесь вы легко найдёте факты и мнения, науке и разных направлениях. Материалы выходят в режиме реального времени, что позволяет держать руку на пульсе. Минималистичный дизайн делает использование комфортным. https://sochidaily.ru Каждое сообщение проходят проверку. Редакция придерживается объективности. Следите за обновлениями, чтобы быть в курсе самых главных событий.
RobertTeels
August 24, 2025Goods delivery from China is trustworthy and fast. Our company provides flexible solutions for enterprises of any size. We handle all logistics processes to make your supply chain smooth. delivery of goods from china by air With regular shipments, we guarantee timely arrival of your consignments. Clients trust our experienced team and competitive rates. Choosing us means assurance in every shipment.
RobertTeels
August 27, 2025Freight delivery from China is trustworthy and swift. Our company delivers flexible solutions for companies of any size. We manage all logistics processes to make your workflow uninterrupted. delivery of cargo from china by air With regular shipments, we ensure timely dispatch of your packages. Clients value our skilled team and reasonable rates. Choosing us means assurance in every order.
EdgarGlink
September 1, 2025В мессенджере Telegram появилась функция звёзд. Теперь пользователи могут выделять важные месседжи. Это помогает быстро открывать нужную переписку. купить звезды телеграм бот Функция комфортна для деловых задач. Благодаря этому легко сохранить ключевые моменты. Такой инструмент экономит время и делает общение эффективнее.
EdgarGlink
September 3, 2025В Telegram появилась функция звезд. Теперь люди могут выделять важные месседжи. Это позволяет быстро открывать нужную переписку. купить 25 звезд Функция удобна для ежедневного общения. С её помощью легко сохранить ключевые моменты. Такой инструмент экономит ресурсы и делает общение проще.
AlvinKah
September 28, 2025Подбор аппаратуры для косметологии — серьёзный процесс в развитии красоты. Для начала стоит уточнить цели и услуги, которые собираетесь проводить. Полезно изучить разрешения и надёжность выбранного оборудования. Комментарии прошлых специалистов помогут сделать осознанный решение. Бьюти Инструмент Также необходимо обратить внимание на набор функций и удобство эксплуатации. Начальная проверка оборудования позволяет понять эффективность работы. Важно оценить тарифы и гарантийные условия. Взвешенный выбор косметологического оборудования способен улучшить качество процедур.
RogerDuets
September 30, 2025Самостоятельные девушки — это яркие личности, которые берегут свою индивидуальность. Они предпочитают к вдумчивому подходу в взаимодействии. Такие девушки обычно обладают четким характером и определенными жизненными установками. Они не стесняются артикулировать свои мысли. https://spaxam.net/ Общение с ними всегда получается глубоким. Они любят слушать собеседника и выстраивать настоящие отношения. Такие девушки влияют окружающих своей естественностью. Они движутся своим маршрутом, не подстраиваясь под сторонние ожидания.
MichaelSpisa
October 3, 2025Современное медицинское оборудование играет важную роль в диагностике и работе пациентов. Клиники всё чаще оснащаются передовую аппаратуру. Это даёт возможность докторам ставить быстрые оценки. Современные приборы гарантируют комфорт и для пациентов, и для персонала. https://lifeposi.ru/apparaty-dlya-tejp-terapii-sochetanie-tehnologij-s-manualnymi-navykami-terapevta-66710/ Внедрение высоких технологий помогает результативное восстановление. Часто устройства имеют возможности для точного мониторинга состояния здоровья. Специалисты могут своевременно реагировать, основываясь на результатах аппаратуры. Таким образом, новейшее медицинское оборудование повышает эффективность лечебного процесса.
Michaeltut
October 30, 2025Нейросетевые боты для поиска информации становятся всё более удобными. Они помогают собирать открытые данные из интернета. Такие решения применяются для журналистики. Они способны оперативно систематизировать большие объёмы информации. глаз бога найти по фото Это позволяет сформировать более объективную картину событий. Некоторые системы также обладают удобные отчёты. Такие боты активно применяются среди аналитиков. Совершенствование технологий делает поиск информации доступным и наглядным.
Floydcop
November 12, 2025This online platform offers a lot of captivating and informative information. On this platform, you can explore a wide range of subjects that expand knowledge. Visitors will enjoy the materials shared on this site. Each section is organized clearly, making it simple to use. The content are relevant and engaging. There are recommendations on various fields. Whether your interest is in useful facts, this site has a lot to offer. Overall, this website is a valuable hub for curious minds. https://it-moves-new-media.de/
Floydcop
November 13, 2025This website provides a lot of engaging and informative information. Here, you can explore a wide range of subjects that provide insights. Visitors will benefit from the materials shared here. All topics is well-structured, making it convenient to use. The posts are presented professionally. There are recommendations on numerous themes. No matter if you seek educational content, this site has something for everyone. All in all, this site is a valuable hub for those who love learning. https://aiti-portal.de/