Chapter 7:
Developments in vaccination and control between 1900 and 1966
INTRODUCTION
By the year 1900 vaccination was in widespread use throughout the industrialized countries as well as in some cities in what were then the colonies of various European powers. Although variolation was no longer practised in Europe and North America, it was still widely employed in many parts of Africa and Asia. Smallpox persisted as an endemic disease in virtually every country of the world (see Chapter 8, Fig. 8.16), although its incidence in the industrialized countries was lower than in previous centuries.
The latter half of the 19th century saw the emergence of microbiology and immunology as scientific disciplines. Because of their familiarity with vaccination, many of the pioneers in these new sciences used vaccinia virus for their studies (see Chapter 2). In consequence, the empirical practices of Jenner and his early followers were placed on a more scientific basis. Vaccine production was no longer the province of the local physician, who had maintained the virus by arm-to-arm transmission, and small local “vaccine parks”, but was taken over by factory-type laboratories, the precursors of the great biological products supply houses of the 20th century.
There was a steady improvement in the quality of the vaccine, the methods for its distribution, and the public health infrastructure, which had reached such a level that by the early 1950s endemic smallpox had been eliminated from the industrialized countries of Europe and North America (see Chapter 8). In the decade after the end of the Second World War, several major advances were made in the production and methods of delivery of the vaccine, but further change and innovation were necessary before global eradication was feasible. This chapter summarizes the state of the art of vaccine production and inoculation, and the results of investigations into the complications of vaccination, as these stood in the period between the end of the Second World War and 1967, when the Intensified Smallpox Eradication Programme was initiated.
During the middle years of the 20th century quarantine measures, which had earlier operated on a national basis in some countries (see Chapter 6), were elaborated and applied on a global scale, with the development of the International Health Regulations by the World Health Organization. The present chapter concludes with an account of the introduction and eventual demise, in 1981, of the International Certificate of Vaccination or Revaccination against Smallpox.
Cowpox Virus and Vaccinia Virus
All orthopoxviruses exhibit cross-protection in laboratory animals. Among the orthopoxviruses that infect humans, cowpox and vaccinia viruses usually produce only local lesions, with minimal systemic disturbance, whereas variola and monkeypox viruses cause serious systemic diseases. Jenner's original "variolae vaccinae" was cowpox virus, and during the 19th century, on many occasions, virus for vaccination was derived from lesions in cows and sometimes horses in several European countries. Since the description of the biological characteristics of cowpox virus by Downie (1939a,b), it has been recognized that smallpox vaccines in use then, and probably for many years before the 1930s, consisted not of cowpox virus, but of another orthopoxvirus which had long been called "vaccine virus", but was shown by Downie to have biological properties different from those of cowpox virus (see Chapter 2). Although some smallpox vaccines were still said to be made from cowpox virus during the 1960s, it is doubtful, in the light of evidence from contemporary virological studies, whether this was so.
The origins of vaccinia virus are unknown. It may have arisen as a hybrid between cowpox virus and variola virus, it may have been derived from cowpox virus or some other orthopoxvirus by serial passage under artificial conditions of culture, or, as Baxby (1981) has suggested, it may be the laboratory survivor of a virus that is now extinct in nature. Whatever its origin, vaccinia virus is clearly a distinct species of Orthopoxvirus, and DNA maps of different strains of vaccinia virus are remarkably similar to each other and different from those of all other orthopoxviruses, including cowpox and variola virus (see Chapter 2, Fig. 2.7, 2.9 and 2.10, and Chapter 29, Fig. 29.1). However, like cowpox virus in the hands of Jenner and his followers, it provided inoculated subjects with a high degree of protection against smallpox, with little risk to either the individual or the community.
VACCINE PRODUCTION AND QUALITY CONTROL BEFORE 1967
Jennerian vaccination was the tool with which the incidence of smallpox was reduced and the disease eliminated from many countries of Europe and the Americas during the first half of the 20th century. However, at some time during the 19th century, for reasons which are obscure, the nature of the virus used for vaccination changed, in most parts of the world, from cowpox virus to another orthopoxvirus, which came to be known as vaccinia virus.
As scientific virology developed during the 20th century, it was applied to the study of smallpox vaccine and vaccination, and new methods of preparing, distributing and inoculating the vaccine were developed. The descriptions which follow refer to the best methods and procedures in use for vaccine production before 1967. However, because smallpox vaccine was produced in many countries, in which there were wide differences in the degree of scientific and industrial experience and skill, the procedures employed in some countries were much less satisfactory than those described here. Modifications to traditional production and assay methods, and—most important—the international quality control of vaccines, were introduced shortly after the Intensified Smallpox Eradication Programme began operations in 1967 (see Chapter 11).
Vaccine Pulp and Vaccine Lymph
By the end of the 19th century arm-to-arm vaccination had been made illegal in many countries and smallpox vaccine was obtained from animal skin. However, technical knowledge of vaccine production was still extremely poor. For example, even in an advanced industrialized country it was recommended that seed lymph could be obtained from (1) "smallpox direct", (2) cowpox, (3) horsepox, sheep-pox, goatpox, swinepox, or (4) vaccinia in the human body (England and Wales, Ministry of Health, 1928a).
Looked at with modern eyes, the production of smallpox vaccine, even in 1966, was a primitive operation. The method of production, in animal skin, made bacterial contamination inevitable, something that would not have been tolerated in a new viral vaccine in the 1950s.
A number of traditional expressions were used to describe the product obtained at different stages of the process: the animal in which the vaccine was produced was called the vaccinifer, the material used for its inoculation the seed, the material reaped from the scarified skin the vaccine pulp, and the clarified suspension obtained from this the vaccine lymph. The pulp was a mixture of epidermal cells and leukocytes, plasma, hair, bacteria and the virus. Clarification removed some of the coarser debris, but the vaccine lymph was milky in appearance on account of contamination with fragments of cells and bacteria. Purified virus, at the concentration found in the lymph, would have been a water-clear suspension.
Production of Vaccine Lymph
During the first half of the 19th century vaccine was usually made available by arm-to-arm transfer but was sometimes dispatched over long distances dried on threads, ivory tips, or glass slides (see Chapter 6). Cows were first used for the production of vaccine in Italy early in the 19th century; this practice gradually spread around Europe and was universal by the end of the century. Over the years, changes were made in the mode of preparation of both liquid and dried vaccine, but the initial production of vaccine pulp was the same for each.
Choice of vaccinifer
For historical reasons, calves were first used for vaccine production. During the First World War, the Lister Institute of Preventive Medicine, in Elstree, Hertfordshire, England, introduced the use of sheep, a practice subsequently adopted in some other countries. Because of their ready availability, water buffaloes were sometimes used in India, Indochina and Indonesia, and W. A. Collier (1953) considered them to be superior to other species for vaccine production. Attempts were made in several countries to produce smallpox vaccine in chick embryos and in cultured cells, to avoid the bacterial contamination that was inevitable when production was carried out by scarification of the skin of large animals. However, chick embryo vaccines were produced on a commercial scale only in Brazil, Sweden and the state of Texas in the USA, and only in Brazil were they in widespread use in an eradication campaign (see Chapter 12). Except on a very limited scale, tissue culture vaccines were never produced commercially.
During the 1920s the concept was developed that biological products prepared for use in man should be bacteriologically sterile. Legislation was introduced in some of the industrialized countries to regulate the production of vaccines and similar products. At that time smallpox vaccine grown on the flank of calves was already in wide use. Inevitably, it contained some bacteria, but there was no other practicable method of production. Thus, smallpox vaccine was the only vaccine for which both the public and the health authorities accepted the fact of contamination by exogenous microorganisms. Since bacterial contamination was inevitable, the aim in preparing vaccine lymph was to exclude pathogenic bacteria completely and reduce the level of contamination with the normal skin bacteria of the vaccinifer. Choice of a healthy vaccinifer was clearly of major importance. The requisite veterinary inspection was practised in some countries, but in other animals were bought in the market, or even rented to be returned afterwards to their owners. This last procedure circumvented one of the important precautions recommended by producers in developed countries-namely, that a thorough postmortem examination should be conducted when the animal was killed at the end of the incubation period and the pulp discarded if serious disease was found.
Under optimum conditions, every animal used for vaccine production was subjected to an examination by a veterinarian to ensure that it was free from tuberculosis, skin infection and ectoparasites. Only animals which had been free of fever and any sign of disease for at least 2 weeks were used, and during this observation period the animals were clipped and washed.

Plate 7.1. The preparation of smallpox vaccine at the Vaccine Production Institute, Dhaka, Bangladesh. A: Calves to be used for vaccination were held in quarantine for 7 days. B: Four days after scarification, the skin was scraped with a sharpened stainless-steel spoon and the vaccine pulp collected in a jar, for subsequent grinding and centrifugation to yield the vaccine lymph.
Preparation of the vaccinifer
The proper preparation of the skin of the vaccinifer was one of the most important steps in obtaining a vaccine of low bacterial content. After being anaesthetized or tranquillized, the animals were washed on an area on the flank or belly, preferably one not liable to soiling by excretions, and then shaved extensively enough to permit a reasonably large area to be scarified. Some producers treated the skin with an efficient skin disinfectant, such as a quaternary ammonium compound, and before vaccination the prepared area was well rinsed with sterile distilled water.
Scarification
The skin was prepared for vaccination by scarification, using the same principle as for the vaccination of humans (see below)—namely, to bring the virus into contact with cells in the Malpighian layer of the epidermis. However, the operation was on a much larger scale, the aim being to produce a confluent take on a large area of the flank and abdomen. The usual procedure was to make a series of parallel scratches about 1 cm apart on the cleansed flank and abdomen of the vaccinifer, followed by a similar series of scratches at right angles to the first. A wide variety of instruments was used in different laboratories, sufficient force being exerted to produce obvious erythema of the skin, but not bleeding. The prepared skin was then inoculated by rubbing seed virus of high infectivity into the superficial scarifications. Practical considerations limited the extent of the area scarified, because in some countries animals in rather poor health were used, and too extensive scarification might have caused their death.
Two important features which were subject to a great deal of variation in different countries, and sometimes even among different producers in the same country, were the choice of the strain of vaccinia virus and the mode of preparation and maintenance of the seed virus (see below).
Incubation period
Methods of maintaining the vaccinifer until the lesions were ready for harvest differed considerably. Ideally, the scarified area was loosely covered with a cotton cloth, which was changed at least once each day. In the better laboratories, the pen in which the animal was housed was so constructed that the vaccinated animal could not lie down. It had walls that could be washed frequently and a hard floor on which was placed a slatted wooden platform the length of the animal and sloping gently from front to back; the pen was cleaned thoroughly and often. In some countries, however, the vaccinifers were housed in ordinary stables and sometimes they were even turned out to pasture.
hey were even turned out to pasture. The pulp was harvested before crusts formed on the vaccinated area, at a time when both the viral titre and the quantity of pulp were at their maximum. Workers at the Lister Institute found in the 1920s that the viral yield was about the same on the 4th and 5th days after inoculation, but preferred the 4-day incubation period because the opportunity for bacterial contamination and hair growth was reduced by a day.
Harvest
The skin was rinsed with warm water, with or without soap, and the pulp was scraped from the skin with a curette, one form of which was made by sharpening the edge of a hemispherical stainless-steel spoon (Plate 7.1B). If the animals were killed, exsanguination before scraping ensured a less blood-stained product; if they were not killed, they were usually anaesthetized before scraping. Harvests from individual animals were kept separately in sterile containers closed tightly enough to prevent drying by evaporation, and stored below 0°C except when being processed.
Seed virus used for inoculation
The seed viruses used in various laboratories differed with regard to the strain of virus employed and the titre of the inoculum, as well as the methods by which they were maintained.
Strain of vaccinia virus. With a procedure that had been in use in many countries for over a hundred years, and with no effort at international standardization, it was not surprising that the strains of virus employed for vaccination in different countries differed in their biological properties. The choice of a particular strain was arbitrary, being based on the history of the vaccine production laboratory concerned. An examination of the situation in 1967 showed that many different strains were then in use (see Chapter 11). This can be understood when it is realized that during the 19th century vaccine production was an unregulated activity, undertaken by a large number of “backyard” producers. For example, as A. C. Hekker has commented (personal communication, 1981), in 1875 there were about 15 “parcs vaccinogenes” in a country as small as the Netherlands, with at least 1 in every province. By 1900 the number had been reduced to 3—in Amsterdam, Groningen and Rotterdam respectively. Finally, in 1954, production was centralized in the National Institute of Public Health, Bilthoven. The situation was similar in many other countries—for example, in Great Britain, where lymph maintained by arm-to-arm inoculation was used until the 1890s.
Since different strains of vaccinia virus vary considerably in their biological properties (see Chapter 2) and since the properties of the viral strain were probably important in determining the differences found in the rates of occurrence of postvaccinial encephalitis in different countries (see Table 7.8), this was not a trivial matter. However, no steps were taken to recommend internationally which vaccinia strain should be used for vaccine production until after 1967 (see Chapter 11), although some countries in which postvac-cinial encephalitis had been a serious problem had changed their production to the less pathogenic Lister strain before this.
Titre of inoculum. All authorities agreed that it was desirable to use an inoculum with as high a titre as possible. Two methods for measuring the titre had been used after the Second World War: scarification of the skin of a rabbit and pock counts on the chorioallantoic membrane of the chick embryo. The rabbit-skin assay, which consisted of a series of scarifications of 10—fold or sometimes 3—fold dilutions of the material under test, was not very precise, hence many producers could not make accurate estimates of the titre of either their inoculum or their product. Pock counting, although early shown to be very suitable for the assay of vaccinia virus (Keogh, 1936), did not come to be widely used by vaccine producers until the 1950s.
Maintenance of seed virus. As recently as 1966, many manufacturers still used some of the vaccine lymph prepared for distribution as the inoculum for the next series of vaccinifers, a procedure that over the years involved a large but unknown number of serial passages of the virus in these animals. Other manufacturers passed the virus in the skin of man or the rabbit after a certain number of subcultures in calves, or alternately in vaccinifer and rabbit. Meeting in 1958, a WHO Study Group on Requirements for Smallpox Vaccine (1959) recommended the use of the seed lot system. This involved, in principle, laboratory maintenance at refrigerator temperature of large stocks of a primary seed lot of a suitable preparation of vaccinia virus. From this primary seed lot enough virus had to be prepared for the production run. Depending on its titre, the extent to which it multiplied, and the number of vaccinifers used, the primary seed was passaged once or several times by the scarification of single calves to obtain the seed lot for the production run. The number of such serial passages of the virus in the vaccinifer was usually restricted to 5. With this method, possible alteration of the strain could be limited, the chance of extraneous contamination was reduced, and the inoculum could be standardized in terms of viral concentration and freedom from contamination.
The Stability of Vaccinia Virus
Stocks of all viruses, even after cloning (i.e., being grown from a single plaque or pock and thus a single viral particle), contain a small proportion of mutant virions (see Chapter 2). Most attenuated live virus vaccines were developed by selecting for certain of these mutants by continued serial passage of concentrated viral suspensions in a novel host. With an agent that was suitable to start with, such as vaccinia virus, the reverse sometimes occurred. The early vaccinators believed that serial passage in humans "weakened" the vaccine, and they resorted to periodic "retrovaccination"—i.e., passage of the virus in cows—to enhance its potency (see Chapter 6). Subsequently, some manufacturers observed that continued serial passage in calves led eventually to a fall in the take rates of the vaccine in man, although others (e.g., W. A. Collier, 1953) found that potency was maintained throughout 35 serial passages in water-buffaloes.
Manufacturers sought to overcome the fluctuations in potency by periodic passage of the seed virus in other animals, usually the rabbit but sometimes monkeys, donkeys, or even human subjects. These manipulations were rendered unnecessary by the development of the "seed lot" system.
Preparation of Liquid Vaccine
Traditionally, the vaccine pulp was processed so as to remove some of the extraneous material and reduce the bacterial count, and it was then dispersed as a liquid suspension called vaccine lymph.
Clarification of the vaccine pulp
The semisolid pulp was usually ground into lymph by comminuting it in a grinder, or later by homogenization in a blending machine. For this purpose, it was usually mixed with 40-60% glycerol. Storage of the glycerolated homogenate at low temperature over a period of months led to a steady fall in the number of viable bacteria, but it was later found that the same result could be more rapidly achieved by the addition of phenol (see below). Periodically bacteriological tests were made to determine when the lymph was suitable for distribution.
Use of glycerol
The introduction of glycerol to “stabilize” the vaccine virus and at the same time prevent bacterial multiplication was regarded as a major step forward in lymph production. This procedure made it feasible to change from arm-to-arm vaccination or the use of an itinerant vaccinated cow (see Chapter 6, Plate 6.12) to the distribution of liquid vaccine in capillary tubes.
Glycerol had three advantages: it acted as an antibacterial preservative, it helped to make the vaccine stick to the skin, and it permitted the maintenance of the vaccine in liquid form at—10°C and thereby ensured the long survival of active vaccinia virus. It also prevented ice formation, a process which, in the presence of phenol (contained in nearly all smallpox vaccines), led to the reduction of viral infectivity. However, although Copeman (1892, 1899) had claimed that glycerol was bactericidal but not virucidal, subsequent work showed that at temperatures above 0°C, it inactivated vaccinia virus rather rapidly. The deleterious effect of glycerol was of little importance in countries with temperate climates, especially if good refrigeration was available, but it was an important cause of vaccine failure in tropical areas.
Use of phenol
The bacterial count in glycerolated pulp stored in the refrigerator fell off very slowly. Several months were usually required before it was low enough to allow the vaccine to be issued for use, and repeated testing was necessary during this period. The procedure could be greatly accelerated by adding phenol to a final concentration of 0.5%, a procedure originally recommended by Gins (1924) and Lehmann (1937) and popularized by McClean (1949), whose protocol was as follows: Material harvested from the sheep was ground with twice its weight of a 1% solution of phenol in distilled water. After this had stood at 22°C for 48 hours, glycerol, equal in amount to the phenol solution, was added, so that the final concentration of phenol was 0.4%.
Mode of distribution
Liquid vaccine was usually dispensed in glass or, later, plastic capillary tubes, in single dose lots or in amounts sufficient for about 20 vaccinations.
Preparation of Dried Vaccine
Liquid vaccine was satisfactory in temperate countries with well-developed health services, good transportation networks and reliable refrigerator capacity. Endemic smallpox was eliminated from Europe and North America by vaccination with liquid glycerolated vaccine, even though its potency was sometimes lower than desirable. At this time many authorities favoured the use of multiple sites of insertion (usually 2 or 4), a procedure which continued up to the 1960s in India and which sometimes produced a satisfactory take even with a substandard vaccine. However, liquid vaccine was totally unsatisfactory for tropical countries, as reports from medical officers in those areas attested. In 1919, for example, the complaint was made in Africa that even for primary vaccination “only 7-20% positive results were obtained with the best vaccine” (Tanganyika Territory, 1920). Likewise, in Madras, Hobday et al. (1961) obtained a 27% take rate in revaccinations with fresh liquid vaccine compared with a 63% take rate with freeze-dried vaccine. Because of its low heat stability, health authorities sometimes went to great trouble to try to maintain the potency of liquid vaccine. In Peru, for example, in the period before freeze-dried preparations became available, vaccine was carried in the field in kerosene refrigerators mounted on the backs of mules (C. Quiros, personal communication, 1984).

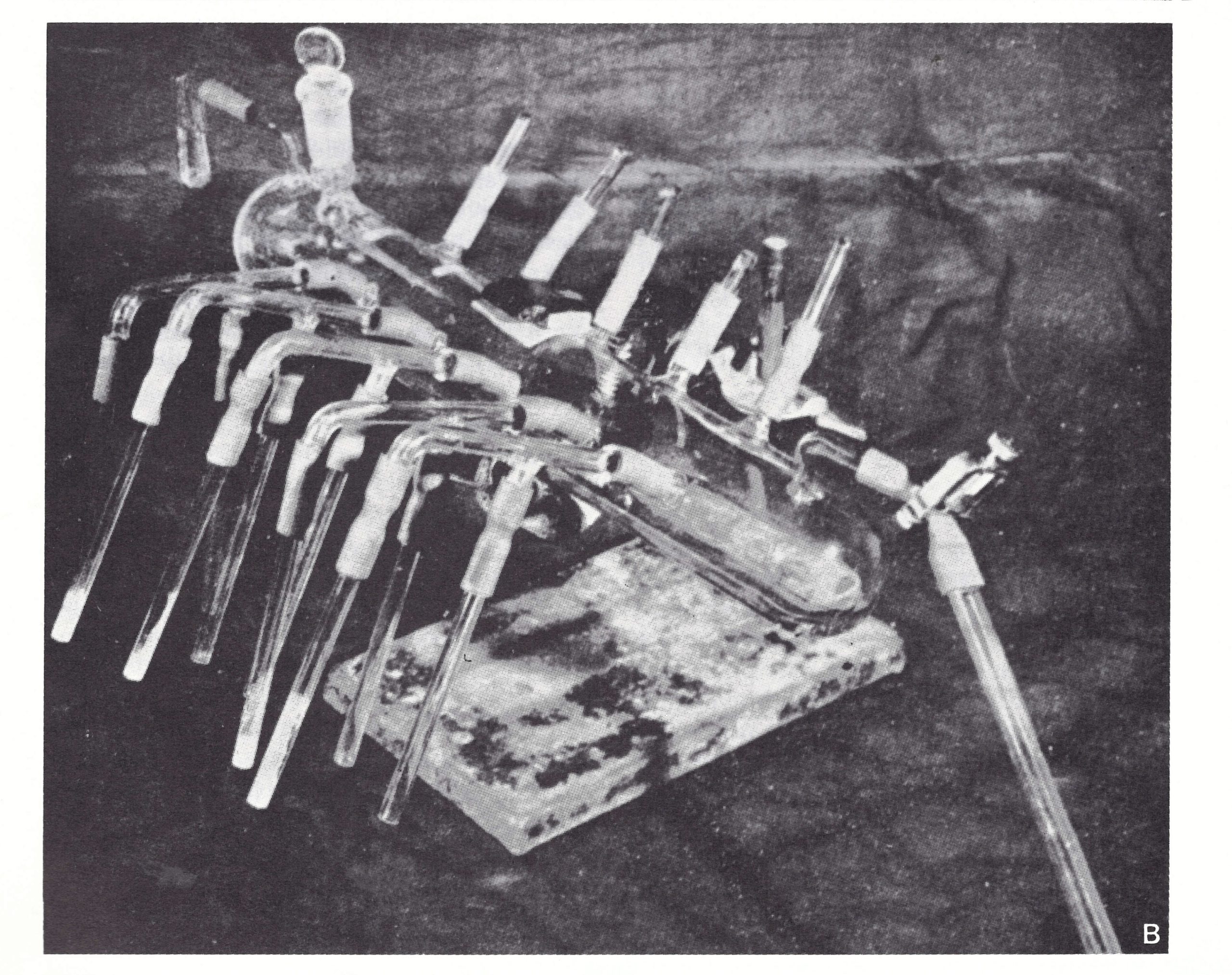
Plate 7.1. A: Apparatus developed by Camus and Fasquelle and used for the preparation of freeze-dried vaccine from 1917 onwards. B: Apparatus developed by Hornibrook (1949) for preparing freeze-dried vaccine. (From Hornibrook, 1949.)

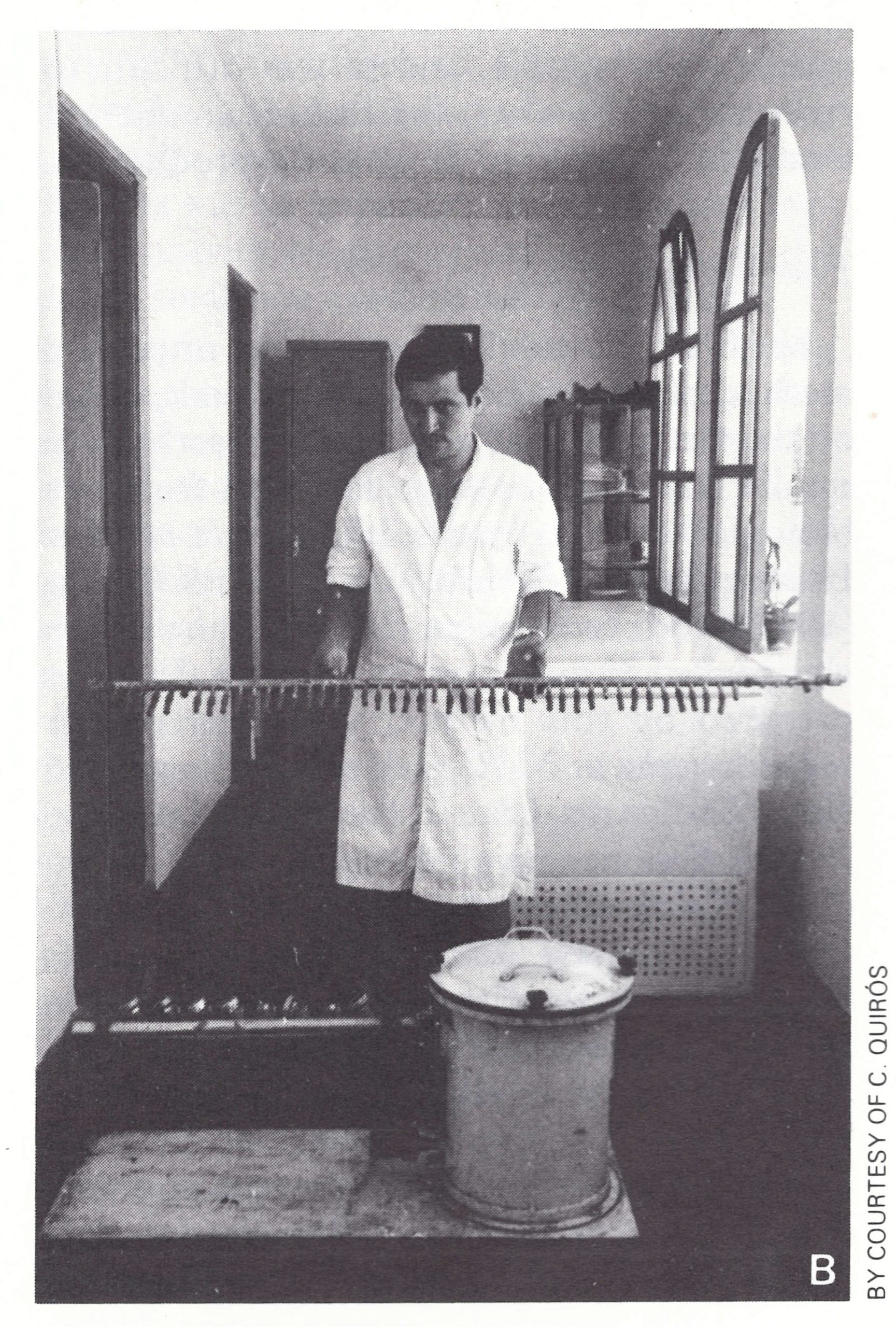
Plate 7.3. A: Apparatus used by Otten (1927) for evacuating ampoules containing airdried vaccine before sealing. (From Olivier et al ., 1932.) B: Commercial development of Hornibrook’s apparatus used for making freeze-dried vaccine in Peru in the early 1950s.
Air-dried vaccines
It was recognized very early that dried vaccine often survived far longer than liquid vaccine when both were maintained at ambient temperatures, and Jenner himself distributed vaccine dried on threads or between glass slides. Over the years a variety of methods for drying liquid vaccine were employed (L. H. Collier, 1954). Among the more effective were those devised by Camus (1909), working at the Vaccine Institute in Paris and concerned to produce an effective vaccine for use in French colonies in the tropics, and by Otten (1927), working in Batavia (Jakarta). Camus placed vaccine pulp in a thin layer under an evacuated bell jar, in the presence of sulfuric acid. The pulp was cooled and shaded from light, and drying was complete within a few hours. In tests in western Africa, after transport at ambient temperatures, it gave take rates in different trials of 66-100%. Otten used a similar procedure: buffalo lymph was dried in vacuo over sulfuric acid, at room temperature. Subsequently the ampoules were attached to a manifold and sealed under vacuum (Plate 7.3A). Although the results were variable, the majority (80%) of preparations of such dried vaccine produced takes in 75-100% of primary vaccinees, after storage at room temperature in Batavia for 16-30 months or at 37°C for 12 months (Otten, 1932; W. A. Collier, 1953).
Otten’s vaccine was an important factor in achieving the elimination of smallpox from the Netherlands East Indies (now Indonesia) in 1937 (see Chapters 8 and 13). However, there was considerable batch-to-batch variation, the vaccine was often heavily contaminated with bacteria, and it was difficult to reconstitute. Further, the technique did not lend itself readily to large-scale production.
Freeze-dried vaccines
In 1909 Shackell reported an improved method of drying biological materials, based on the fact that H2O could pass directly from a solid to a gaseous state. He deep-froze the biological material and sublimated it in vacuo. During the First World War, Wurtz & Camus (1919) produced a freeze-dried vaccine which was packed in tubes under vacuum. Samples prepared by this method were active after several weeks at 37°C and, when transported from France to Cote d’Ivoire, Guinea and French Guiana, gave take rates of 85-100% (Fasquelle & Fasquelle, 1949) (Plate 7.2A).
Approximately 10 million doses of this dried vaccine were sent every year between 1920 and 1940 to the French colonies in Africa for the smallpox vaccination campaign, and the Vaccine Institute in Paris continued to supply it to the francophone countries of Africa until 1966, in amounts ranging from 4 to 12 million doses annually (J. Fasquelle, personal communication, 1983). During the eradication programme in western Africa in 1967 (see Chapter 17), it was found that smallpox was less of a problem in countries that had been French colonies, in part because of their previous use of freeze-dried vaccine.
Meanwhile, from the 1930s onwards various laboratory workers in other countries had adopted freeze-drying for the preservation of vaccinia virus (Rivers & Ward, 1933, 1935; Lloyd & Mahaffy, 1935; Hahn, 1951; L. H. Collier, 1951). The technology was substantially improved by Flosdorf & Mudd (1938). A freeze-dried vaccine developed by Kaiser (1937, 1942) was said to have been used extensively by the German army during the Second World War (cited by L . H. Collier, 1954).
In 1948, the year in which the World Health Organization was established, a WHO study group on smallpox vaccine met in Paris and reported to the First World Health Assembly that freeze-dried vaccine such as that prepared at the Paris Vaccine Institute (using the method developed by Wurtz & Camus) had proved to be an effective heat stable vaccine in the French colonies for a number of years. Stimulated by these reports, Dr. Fred L. Soper, then Director of the Pan American Sanitary Bureau, encouraged the United States National Institutes of Health to carry out studies on appropriate methods for the production of freeze-dried smallpox vaccine, in order to fulfill the proposal made by the Executive Committee of the Pan American Sanitary Organization in 1949 that all countries in the Americas should cooperate in national programmes designed to eradicate smallpox (Soper, 1966). The technical investigations were undertaken by the Division of Laboratories, Michigan Department of Health, then a leading producer of glycerolated liquid vaccines (Ducor, 1947). They resulted in the development of a method for freeze-drying vaccine that had been dispensed in 0.5-ml lots in Pyrex tubes (Hornibrook & Gebhard, 1951), using a sulfuric-acid drier (Plate 7.2B) developed by Hornibrook (1949). Peru was the first country in the Americas to use this process on a commercial scale, beginning in October 1953.
In June 1952, WHO organized comparative studies on the heat stability of freeze-dried vaccine produced by the Vaccine Institute, Paris, the State Serum Institute, Vienna, the Division of Laboratories, Michigan Department of Health, Lansing, USA, and the Pasteur Institute, Bandung, Indonesia. The tests were carried out in the State Serum Institute, Copenhagen, the Division of Laboratories, Michigan Department of Health, the Vaccine Institute, Paris, the Lister Institute of Preventive Medicine, Elstree, and the New York City Department of Health. No laboratory performed the tests on vaccine that it had itself produced. The results showed that the heat stability of the vaccine provided by the Michigan Department of Health was the best of the 4 tested.

Plate 7.4. Leslie H . Collier (b. 1921). As a graduate student at the Lister Institute of Preventive Medicine, Elstree, Herts., England, in the early 1950s Dr Collier developed the method of freeze-drying of vaccinia virus that was subsequently adapted to large-scale freeze-dried vaccine production in many laboratories throughout the world.
The large demand for human plasma proteins for use in the Second World War had led to further advances in the technology of freeze-drying, notably the introduction of a centrifugal freeze-drying apparatus (Greaves, 1946). L. H. Collier (1951), working at the Lister Institute as a graduate student under the direction of D. McClean, applied this technique, with the addition of peptone as a stabilizing agent, to the preservation of vaccinia virus and subsequently developed it for large-scale commercial production (L. H. Collier, 1955). WHO decided to conduct an additional test to compare the new Lister Institute vaccine with the freeze-dried vaccine from Michigan. Samples maintained at 37°C and 45°C for various periods were titrated by pock counting on the chorioallantoic membrane and by scarification in rabbits and tested by primary vaccination of Royal Air Force personnel (Cockburn et al.,1957). The Lister freeze-dried vaccine was found to give 100% successful takes after storage for 64 weeks at either 37°C or 45°C. The Michigan vaccine was much less heat stable, the take rate falling to 72% after 16 weeks and 10% after 32 weeks at 37°C (Table 7.1), and to 47% after 4 weeks at 45°C. The results of titration of the two vaccines by pock counting on the chorioallantoic membrane, in an independent laboratory, were consistent with these results, in that the Lister vaccine maintained its titre whereas that of the Michigan vaccine fell off steadily after storage.
As further modified by C. Kaplan, by the substitution of fluorocarbon treatment for differential centrifugation as a method of partial purification, L. H. Collier’s method was eventually adopted by WHO for the global smallpox eradication programme (see Chapter 11).
Subsequently, freeze-drying technology was greatly improved for the large-scale commercial production of vaccine, particularly in the methods of filling and sealing ampoules. The availability of a method for the long-term preservation of vaccine in tropical climates was an important factor in encouraging countries to participate actively in the global smallpox eradication programme.
Vaccine pulp destined for freeze-drying was prepared as for liquid vaccine, except that glycerol was not added. In some laboratories, it was diluted or concentrated according to its potency on assay. In order to reduce bacterial contamination to an acceptable level, phenol was added to the bulk solution of the vaccine so that its final concentration did not exceed 0.5%.
Before 1967, the bulk solution was put into special ampoules or vials containing 0.25, 1.0 and 5.0ml, corresponding respectively to 25, 100 and 500 doses. These were then placed in a freeze-drier, of which there were two types the centrifugal drier and the shelf drier (see Chapter 11, Plate 11.7). The former required the use of a secondary drier, which consisted of a number of manifolds mounted over phosphorus pentoxide as a desiccant, while the latter provided both primary and secondary drying and sealing of the containers under vacuum inside the drier.
Development of Freeze-dried Smallpox Vaccine
"In 1948, I was told by the then Director [of the Lister Institute], Dr (later Sir) Alan Drury, that there was a great need for a smallpox vaccine that would be stable at tropical temperatures. He asked if I would be interested in this problem, and suggested that freeze-drying in which he himself was concerned as a means of preserving blood plasma might be a useful technique.
"At the outset, I aimed for a method that would yield consistent heat stability results and set myself these additional criteria: the vaccine must still comply with the official standard of potency after at least one month at 37°C (a requirement later adopted by the British and European Pharmacopoeias and by WHO); if dried, it must be safe and easy to reconstitute in the field by vaccinators with little technical training; and it must be possible to produce it economically on a large scale. In addition, it would of course have to comply with the official regulations on bacterial contamination.
"In my field experiments, I compared the stabilities of the glycerinated and lanolinated vaccines in current production; although the latter was the more stable at 22°C, it completely lost its potency within a month at 37°C. I then tried Otten's method of drying crude vaccine from the liquid state over sulphuric acid; vaccine made in this way was widely used in Indonesia and some batches had survived at ambient temperatures for remarkably long periods. There was however considerable batch-to-batch variation, a finding that I confirmed; furthermore, the bulk-dried vaccine was cumbersome to handle and distribute into ampoules.
"Without further ado I then started to explore the possibilities of drying from the frozen state, using a large centrifugal dryer of the type that had recently been invented by R. Greaves at Cambridge. This machine was made in the Institute's workshops and although primitive in appearance, it worked very well; the main difficulty was the making of vacuum-tight seals, at that time rather more of an art than a science.
"The general plan was to prepare experimental vaccines in various ways, to freeze-dry part of the batch and then to compare the keeping properties of the dried and corresponding liquid preparations at 4°C, 22°C and 37°C. Each batch was tested monthly, sometimes for more than two years. It was soon clear that dried lots of routinely produced animal skin vaccine varied considerably in stability; materials partly purified by differential centrifugation yielded much more uniform results.
"The next problem was rather more difficult. Phenol was added to destroy contaminating bacteria; this had to be done before drying, otherwise, the bacteria would have been preserved along with the virus. Although phenol in low concentration does not harm vaccinia virus in liquid suspensions, vaccines containing it lost much of their potency on freeze-drying; this was eventually traced to the tendency of phenol to come out of solution during rapid cooling and become concentrated to a degree that killed the vaccinia virus. This effect could however be prevented by adding peptone to a concentration of 5 before drying the vaccine. Fortunately, among the many additives tried, peptone also proved by far the best for preserving potency at all temperatures tested; it had the further advantages of being non-antigenic, cheap and easy to reconstitute.
"After much experimentation, a satisfactory method was devised for purifying animal vaccine and freeze-drying it after adding peptone to a concentration of 5%. Vaccine thus prepared consistently maintained its original potency for at least three months at 37°C; in later experiments, batches stored at the high temperature of 45°C still gave 100% successful primary vaccination after four years. The criterion for the permissible minimum content of vaccinia virus was fixed by determining the amount of virus needed to achieve 100% successful vaccinations; the final step in the development stage was the devising of a simple and safe method for reconstituting the dried vaccine in the field. It then remained only to scale up all the processes to a point at which full production could begin; this was accomplished by 1953." (L. H. Collier, personal communication, 1980.)
Quality Control
At the national level
Smallpox vaccine had been used for many years before the concept developed that it should be subject to national quality control, in addition to whatever steps the manufacturer might take to protect the reputation of his products. By the mid-1920s a few countries had developed regulations governing biological products destined for medical use, such as vaccines, toxoids and sera.
In January 1926 the Smallpox and Vaccination Commission of the Health Organisation of the League of Nations decided to collect information from vaccine producers regarding the production, testing, standardization, storage and delivery of smallpox vaccine. France, Germany, the Netherlands, Switzerland and the United Kingdom participated in this survey, which was completed in 1927. On the basis of the results, the United Kingdom incorporated standards into the Therapeutic Substances Regulations 1927 (England and Wales, Ministry of Health, 1928a). A brief review of these regulations provides some information on how the quality control of smallpox vaccine was then being conducted in the United Kingdom. The regulations defined certain conditions concerning the qualifications of staff, the animals to be used, the housing of such animals, precautions to be observed during the production process, specifications for final containers for the use of the vaccine, and labelling. The provisions relating to the purity of the vaccine indicated that each batch should be tested to ensure the absence of anaerobic organisms and streptococci: bacterial counts should be less than 5000 per ml. Scarification of the cornea of guinea pigs or the skin of rabbits with diluted vaccine lymph was the principal method for potency testing, typical vaccinia lesions being regarded as the criterion for adequate potency. It seems likely that the quality control of vaccine lymph was being carried out along these lines in many other countries of Europe and perhaps in North America at that time. During this period the application of the control system depended on the interest and capabilities of the production laboratory, since there was no mechanism for independent assessment.
At the international level
In 1946 the Interim Commission of the World Health Organization was set up, taking over the functions of the Health Organisation of the League of Nations. The Commission established a section (unit) of Biological Standardization in the Secretariat. The responsibilities of this section included the establishment of international standards for biological assay, the formulation of requirements for biological products, the coordination of research on biological standardization and the encouragement of Member States to set up national control laboratories for biological substances (Outschoorn, 1973).
WHO standards for smallpox vaccine were first established in 1958, at a meeting in which 6 experts from Europe, 2 from North America, 2 from Asia, and 1 from North Africa participated (WHO Study Group on Requirements for Smallpox Vaccine, 1959). This meeting suggested the criteria that should be established for smallpox vaccine, in relation to the strain of vaccine, methods of preparation of the vaccine lymph, bacterial counts, use of the seed lot system, methods of potency assay and standards for the heat stability of freeze-dried vaccine. In 1965 these requirements were revised in the light of experience accumulated during the intervening period (WHO Expert Group on Requirements for Biological Substances, 1966). Meanwhile, after extensive collaborative studies, the first reference preparations of smallpox vaccine were established in 1962 (ampoules containing 14 mg of freeze-dried smallpox vaccine; Krag & Bentzon, 1963) and of antismallpox serum in 1965 (ampoules containing 84.3 mg of freeze-dried pooled human serum 1000 IU per ampoule; WHO Expert Committee on Biological Standardization, 1967). These reference preparations were produced with a view to facilitating the laboratory investigations needed in order to improve vaccine quality. The modifications proposed in 1965 reflect the progressive improvement in vaccine production and assay.
Seed lot system. In the 1959 WHO requirements, up to 10 serial passages of the seed virus were permitted. Meanwhile, advances in viral genetics and practical experience with poliovirus vaccine had drawn attention to the need to minimize the serial passage of seed virus if genetic stability was to be maintained. In response to this new knowledge, the acceptable maximum number of passages of vaccinia virus secondary seed lots was reduced to 5.
Bacterial count. In 1959 the acceptable count was set at less than 1000 non-pathogenic bacteria per ml, whereas in 1965 this number was reduced to less than 500 per ml, because manufacturers had improved their techniques for the handling of animals and the collection of the vaccine pulp.
Potency. Cockburn et al. (1957) found that vaccine with a titre of 1.5 x 107 pock-forming units per ml gave 100% successful takes in groups of about 100 primary vaccinees, and in a more elaborate statistical analysis of their data (published as an annex to the article cited), C. C. Spicer showed that vaccines with a potency of 108 pock-forming units per ml should give less than 1 failure in 1000 primary vaccinations. In 1959 a titre of 5 X 10 7 pock-forming units had been recommended as the minimal acceptable potency, but in 1965 the level was doubled, to 108 pock-forming units per ml. This change was based on observations made by the WHO Expert Committee on Smallpox (1964), to the effect that:
“…vaccines of relatively poor potency are adequate for successful primary vaccination but inadequate for successful revaccination. Their use for revaccination gives a false sense of security, since a negative response is often taken as evidence of immunity. Failure in successful revaccination explains in part the continued presence of smallpox in some endemic countries where vaccination is regularly practised.”
Changes were also made in the recommended method of titration. Following the observations by Cockburn et al. (1957) that pock counting was considerably more precise than rabbit scarification (see Table 7.1) and that pock counts correlated well with take rates (Fig. 7.1), the 1959 recommendation that rabbit-skin scarification and pock counting were acceptable alternatives was changed so as to eliminate scarification.
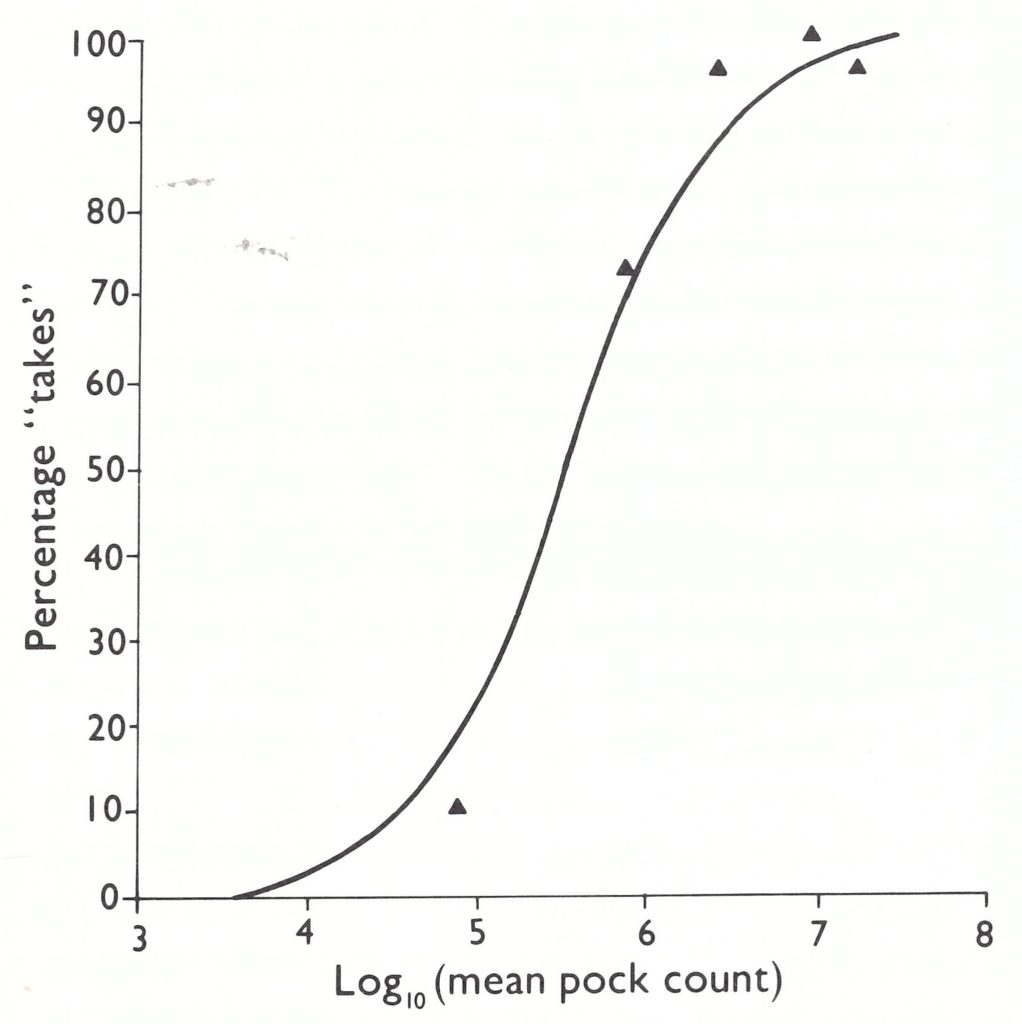
Fig. 7.1. Relationship between pock counts of vaccinia virus and the percentage of successful takes on primary vaccination. The solid line represents a theoretical curve based on probit analysis assuming a 50% probability of a take at a dose of 3 x 10 5 pock forming units per ml. (From Cockburn et al .,1957.)
Heat stability. The requirements for heat stability remained unchanged; vaccine should maintain the minimum acceptable potency (now 10 8 pock-forming units per ml) after incubation at 37°C for 4 weeks.
Implementation of the WHO recommendations. The WHO requirements for smallpox vaccine established in 1965 remained in effect throughout the Intensified Smallpox Eradication Programme. However, the promulgation of standards by WHO in 1959, and again in 1965, and their application in laboratories in diverse countries were very different matters, especially before the WHO Smallpox Eradication unit organized an international quality control system in 1968. Before that, many laboratories lacked competent staff, adequate facilities and interest. Some production laboratories did no potency assays at all; others relied on tests in unvaccinated children, regarding 9 takes out of 10 as satisfactory (a result that could be obtained with a vaccine whose titre was as low as 10 6.5 pock-forming units per ml; see Fig. 7.1). For producers who did wish to titrate their production lots, reference preparations of known titre were required. However, only 2 or 3 ampoules of the international reference vaccine, prepared in 1962, were supplied to production laboratories, from which the laboratories were expected to produce their own working standards. In developed countries this was no problem, but it was beyond the capacity of many laboratories in developing countries. Further, some developed countries did not adopt the standards recommended by WHO. For instance, in the USA, the rabbit skin scarification method was still used as the method for estimating the potency of smallpox vaccine until 1971. In Japan, a titre of 5 x 10 7 pock-forming units per ml remained the officially accepted minimum level of potency for several years after 1965. National quality control of smallpox vaccines was exercised in only a few countries.
From 1959 to 1966, WHO’s participation in monitoring the quality of vaccine was limited to material which had been donated to the Organization for use in the global eradication programme. This amounted to only about 7 million doses a year (total over the period: some 46 million doses; see Chapter 11, Table 11.1). Material supplied through bilateral assistance or by local production was not tested by WHO. This often resulted in the use of substandard vaccine. As A. S. Outschoorn, former Chief Medical Officer of the WHO Biological Standardization unit, said:
“It has never been feasible for W.H.O. laboratories to be established which would undertake control of biological products on behalf of Member States. There would perhaps be a place in the future for private enterprise to make available sources of expertise or advisory services and even testing facilities from which Member Governments could call for assistance outside of the World Health Organization.” (Outschoorn, 1973.)
The absence of effective quality control was one of the first obstacles that had to be overcome when the Intensified Smallpox Eradication Programme was established in 1967 (see Chapter 11).
VACCINATION TECHNIQUES BEFORE 1967
During the long history of vaccination, practices, and techniques were gradually developed that differed from country to country. However, by the 1950s they shared many features, which are summarized below.
Vaccination Site
The most commonly used site for primary vaccination and revaccination was on the extensor surface of the upper arm, over the deltoid muscle. In some parts of India, however, revaccination was carried out on the flexor surface of the forearm. The skin site was usually rubbed with alcohol or acetone. Although studies described in Chapter 11 showed that such skin preparation was unnecessary and could, if the alcohol did not completely evaporate, partially inactivate the vaccine, the practice continued to prevail in many countries.
"Vaccination" and "Immunization"
The term "vaccination" sometimes causes confusion because it has both a specific and a general meaning. Vaccination against smallpox was the only form of preventive immunization against an infectious disease until 1880, when Pasteur developed techniques of attenuation of virulence and protective inoculation of animals against anthrax and chicken cholera. In 1881, in order to recognize Jenner's contribution to the concept of inoculation with attenuated microbes as a method of protection against infectious diseases, Pasteur proposed that this procedure should be called "vaccination" and the product used a "vaccine", whatever its nature. This terminology remains in use, though the general procedure is now usually called "immunization"; the product is still called a vaccine—e.g.,polio vaccine, measles vaccine. In this book "vaccine" always means smallpox vaccine unless specified in some other way.
Methods of Vaccination
Several methods were used, all involving the introduction of the virus into the Malpighian layer of the epidermis, with a variety of instruments (Plate 7.5). In most countries, either the skin was scarified by a single linear incision or scratch, or vaccine was introduced into the epidermis by the multiple pressure method, but in a few places a rotary lancet was used.
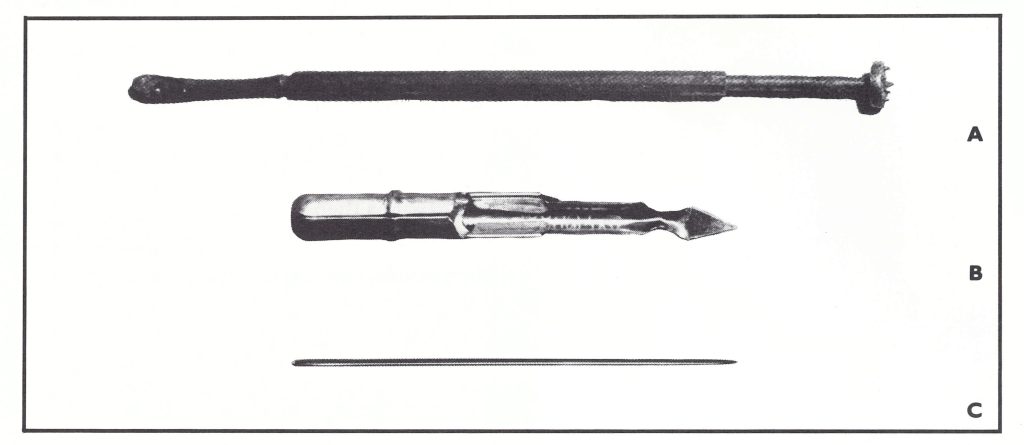
Plate 7.5. Instruments used for vaccination (actual size) before the introduction of the bifurcated needle. A: Rotary lancet. B: Vaccinostyle, used for scratch vaccination. C: A straight surgical needle is used for vaccination by scratch or multiple pressure method (See Fig . 7.2).
Methods of Vaccination
Several methods were used, all involving the introduction of the virus into the Malpighian layer of the epidermis, with a variety of instruments (Plate 7.5). In most countries, either the skin was scarified by a single linear incision or scratch, or vaccine was introduced into the epidermis by the multiple pressure method, but in a few places a rotary lancet was used.
Dermal scarification
A scratch about 5 mm long was made in the skin with a needle, a lancet or a small knife and the vaccine suspension was rubbed into the site. A single cut or cross cuts were made, in 1, 2 or 4 different sites. This was essentially the same method as had been used for variolation in Europe during the latter part of the 18th century.
Multiple pressure method
From very early times attempts were made to deposit virus between the skin layers by methods other than the scratch method or intradermal inoculation with a syringe and needle. Leake (1927), in a pamphlet offering official advice on vaccination to physicians in the USA, recommended the “multiple pressure” method (Fig. 7.2), which was subsequently adopted in several other countries (Parish, 1944; England and Wales, Ministry of Health, 1956). A small drop of vaccine was placed on the skin and the side of a straight surgical needle, held tangentially to the skin surface, was pressed firmly and rapidly into the drop by an up-and-down motion, about 10 times for primary vaccination and 30 times for revaccination. Excess vaccine was wiped off. Only 1 insertion site was recommended, for both primary vaccination and revaccination. This method was less traumatic than scarification, while still producing an adequate take. However, it was difficult to train vaccinators to exert sufficient pressure, and this sometimes caused unsuccessful vaccinations.
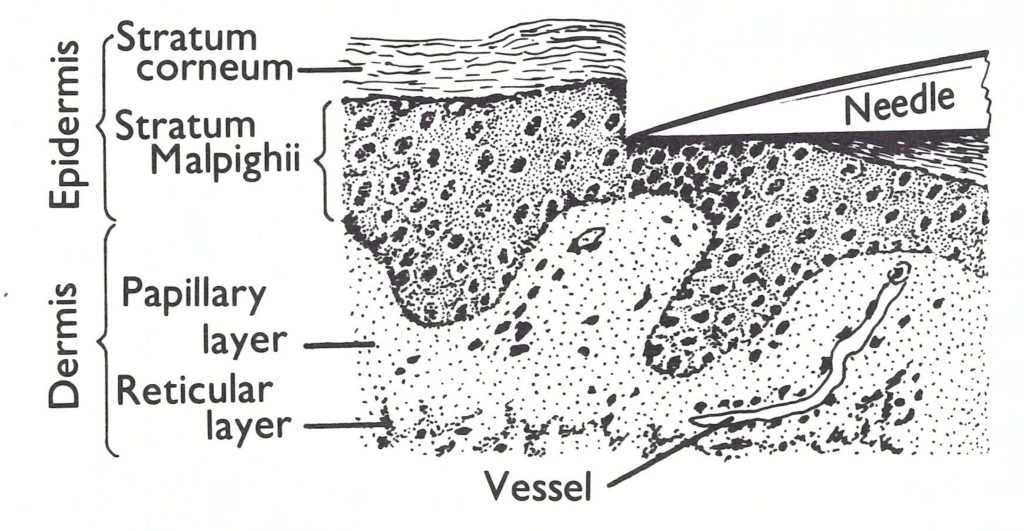
Fig. 7.2. The principle of the multiple pressure method of vaccination. Virus on the tip of the needle is deposited within the deeper layers of the epidermis.
Rotary lancet
Vaccination using the rotary lancet (Plate 7.5) was carried out in many parts of the Indian subcontinent. A rotary lancet designed for vaccination was first described by Rose (1871) and was manufactured in the United Kingdom and later in India. The vaccine was placed on the skin with the small spoon on one end of the instrument. The disc, anchored by the slightly longer central spike, was rotated in such a way that the small lateral spikes abraded the epidermis. If potent vaccine was used, the ensuing trauma produced a large and severe vaccinial reaction, which was a frequent cause of refusal to undergo vaccination. Further, the vaccines then in use were often heavily contaminated with bacteria and the use of the rotary lancet could produce scarring even when the infection had been due to pyogenic bacteria rather than vaccinia virus. Failures sometimes occurred when vaccinators did not allow the lancet to cool adequately after the flaming that was carried out after each vaccination.
Although the WHO Expert Committee on Smallpox (1964) recommended that this method should not be used, it was still routinely employed in some parts of the Indian subcontinent long after this. During the smallpox eradication programmes in Pakistan and India (see Chapters 14 and 15), the programme staff hunted for rotary lancets and confiscated a large number of them from vaccinators who were reluctant to abandon the method but were thus obliged to use the bifurcated needle. However, many remained in use, particularly in the municipal corporations in India, as late as 1974.
Age for Primary Vaccination
Throughout the 19th century, stress was placed on the primary vaccination of infants as the optimum method of providing protection against smallpox, and laws for compulsory vaccination during the first 3-6 months of life were introduced in many countries. However, in the St Petersburg Foundling Hospital, infants aged 7-8 days were vaccinated with lymph maintained by arm-to-arm vaccination from 1801, when the Empress of Russia obtained lymph from Jenner, until 1867, when vaccine from cows began to be used. There were no serious complications, but a few infants required repeated inoculation before a take was obtained. Compulsory registration made it possible to keep track of foundlings until they were 25 years old, so that observations on the efficacy of vaccination were possible. There were 17 epidemics of smallpox in St Petersburg between 1826 and 1846; out of about 15 000 foundlings, only 34 had smallpox, with only 1 fatality (Frobelius, 1869; Donnally & Nicholson, 1934). Elsewhere, the vaccination of newborn infants exposed to cases of smallpox was occasionally recommended, but routine vaccination of newborn children in a hospital appears to date from the early 20th century, in some centres in France and Germany. The compulsory vaccination of neonates was first practised in Detroit, USA, at the time of an epidemic of variola major in 1925 (Lieberman 1927).
Espmark & Rabo (1965 a,b) examined the effect of maternal antibody on the response of infants vaccinated under the age of 1 month. They found that more potent vaccines were needed for these younger infants than for infants vaccinated at 5-12 months of age, but that with potent vaccines (1080 TCID 50 per ml or higher) take rates and neutralizing antibody responses were equally good in both groups, and the local signs and constitutional symptoms were milder in the younger infants.
In most countries of Europe and North America, infants were vaccinated during the second 6 months after birth. In the United Kingdom (Conybeare, 1964b) and in the USA (Neffet al., 1967; Lane et al., 1969), investigations into the complications of vaccination indicated that the risk of post vaccinial encephalitis in infants vaccinated before the age of 1 year was greater than that in those vaccinated later. This led to recommendations that vaccination should be postponed until the 2nd year of life. However, in countries in which the maternal antibody level was reasonably high—e.g., Sweden—it was shown that vaccination in the first 3 months of life was effective, and was usually attended by less severe signs than vaccination later in infancy (Espmark et al., 1973)
Other considerations were important in endemic countries, in which the case-fatality rate for smallpox was very high in young infants. Noting that in Madras, India, most infants were born in hospitals and were accessible as neonates but often difficult to trace after they left hospital, Rao carried out a pilot study in 1959-1960 in which 2500 infants were vaccinated with liquid vaccine on the 3rd day of life, with a take rate of 82% (Rao & Balakrishnan, 1963). Subsequently, take rates of 100% were obtained with freeze-dried vaccine, and the practice of neonatal vaccination was extended to a number of urban areas throughout India (WHO/SE/71.30, Rao).

Plate 7.6. Lesions in a Pakistani child due to use of the rotary lancet, with insertions at 4 separate sites.
Interpretation of the Results of Vaccination
In susceptible individuals, smallpox vaccination produced a typical Jennerian pustule at the inoculation site and frequently swelling and tenderness of the draining lymph node. At the height of the reaction there was usually slight fever, and the subject might feel indisposed for a few days. A feature of smallpox vaccine, which among the variety of agents now used for immunization against infectious diseases is shared only with BCG vaccine, is that successful vaccination produced a characteristic skin reaction which could be readily observed, and which usually left a permanent and characteristic scar. This had both immediate and long-term consequences. Observation of the nature of the cutaneous lesion after recent vaccination or revaccination enabled the vaccinator to decide whether the virus had replicated, and the patient had thus been rendered immune to smallpox. In the longer term, the vaccination status of an individual or a population could be determined, with considerable accuracy, by visual examination for vaccination scars, thus obviating the need for a serological survey. For these reasons, special attention was devoted to skin reactions after both primary vaccination and revaccination.
The clinical features of smallpox in unvaccinated and vaccinated persons and the pathogenesis and immune responses in smallpox and after vaccination are described in Chapter 1 and Chapter 3 respectively. It may be useful to recall here the salient features of immunity to smallpox (see box).
Major reaction in primary vaccination
A typical Jennerian pustule was termed a “major reaction”, and constituted evidence that the vaccinee would be protected against smallpox. The course of the reaction is illustrated in Plate 7.7A and C. A papule appeared at the vaccination site on the 3rd day after vaccination, and within 2 or 3 days this became vesicular, to constitute the umbilicated and loculated “Jennerian vesicle”. As in smallpox (see Chapter 1), the vesicle soon became pustular, owing mainly to the entry of polymorphonuclear cells, the migration of which was stimulated by the viral infection itself, and the surrounding area became erythematous and indurated to a much greater extent than was found in the skin lesions of smallpox. The area of erythema reached a maximum between the 8th and 12th days (usually on the 9th or 10th day), and at this time the draining lymph nodes were enlarged and tender and the subject often sustained a mild fever and may have felt unwell. The pustule dried from the centre outwards to become a dry brown or black scab which fell off about 3 weeks after vaccination, to leave a typical pitted scar.
For routine inspection, observation of the pustule on the 7th day confirmed whether vaccination had been successful. The reaction to vaccinia virus could be readily distinguished from reactions due to bacterial infection, both by its time course and its characteristic appearance.
Revaccination
As described in Chapter 3, successful primary vaccination elicited not only humoral immunity, but also a longer-lasting cell-mediated immunity, which conditioned the response to revaccination. Interpretation of the results of revaccination was sometimes difficult, in terms of evaluating their significance in relation to protection against smallpox. Sometimes there was no reaction at all, a result which was usually due to the use of vaccine of low potency, and which was impossible to interpret correctly (WHO Expert Committee on Smallpox, 1964). If it did occur, the reaction to revaccination could be maximal at any time between the 2nd and the 8th day. During the period before the global smallpox eradication programme, 3 varieties of response used to be distinguished (see, for example, van Rooyen & Rhodes, 1948):
-
Immediate reaction.
Although it used to be described as an "immediate" reaction, dermal hypersensitivity to vaccinial protein produced erythema during the first 24-48 hours after vaccination. It was a classical delayed hypersensitivity reaction, which could be elicited by non-infectious vaccine as well as by active vaccinia virus. Such a response might be given by highly immune individuals even when potent vaccine was used, but it could also occur in individuals with little or no residual immunity who were given inactive vaccine.
-
Accelerated reaction.
Persons with some residual cell-mediated immunity but not enough to inhibit viral replication experienced erythema and the development of a vesicle and sometimes a pustule, which evolved in a sequence more rapid than that seen in a primary vaccination reaction (Plate 7.7B and D). Those with substantial immunity experienced little more than an immediate reaction, whereas those with minimal residual immunity experienced a reaction almost indistinguishable from that seen after primary vaccination. The result of revaccination was dependent on the balance between the potency of the vaccine and residual immunity. A highly potent vaccine could provoke a major reaction, perhaps slightly accelerated, in an individual who had failed to respond to a less potent vaccine; revaccination on the flexor surface of the forearm was more often followed by a substantial reaction than revaccination over the deltoid muscle.
-
Major reaction in revaccination
If a long period had elapsed after a primary vaccination, revaccination could produce a reaction similar to that described for a primary vaccination. This was called a major reaction.
The WHO Expert Committee on Smallpox (1964) considered that the traditional classification of the reaction to revaccination as “immediate”, “accelerated” or “major”, as just described, could be misleading, in terms of judging whether an individual might be susceptible to smallpox. The Expert Committee therefore recommended that revaccination should be recorded as successful if, on examination 6-8 days later, there was a pustular lesion or an area of definite induration or congestion surrounding a central lesion, which might be a scab or an ulcer. Such a response was termed a “major reaction”. All others were termed “equivocal reactions”—i.e ., the persons concerned could not be presumed to be immune to smallpox and were revaccinated.
How Vaccination Protected against Smallpox
A large part of the DNA of all orthopoxviruses is very similar and codes for polypeptides that have a close resemblance in all orthopoxviruses. Vaccination against smallpox consisted in the production of an infection of the skin with vaccinia virus, with extension to the lymph nodes and spleen, organs concerned with the immune response. Because vaccination involved infection of the skin with a relatively large dose of a virus that replicated rapidly, generalization of infection and therefore the immune response occurred more quickly than they did in naturally acquired smallpox. This explains why vaccination during the incubation period of smallpox sometimes aborted or modified the clinical course of the disease.
The immune response to vaccination results in the development of cell-mediated immunity, probably to antigens expressed on the surfaces of infected cells, and a number of humoral antibodies, some of which can neutralize infectivity and may persist for long periods. There are also memory cells for both cell-mediated and humoral immune responses, which wane slowly.
For a few years after vaccination, the level of immunity may have been sufficient to prevent completely the replication of variola virus. A somewhat lower level of immunity may have allowed limited replication of variola virus, short of symptom production, but with an anamnestic immune response (subclinical infection). Still lower levels of immunity may have allowed the generalization of variola virus to occur, but stimulation of the memory cells would have produced an accelerated immune response which would have modified the clinical manifestations of smallpox (modified-type smallpox). After many years the immunity provided by vaccination might have waned to such an extent that the attack of smallpox might not have been modified in any way and might indeed have been fatal.
COMPLICATIONS OF VACCINATION
Smallpox was such a dire disease and the effects of vaccination by comparison so trivial that for a hundred years after vaccination was introduced little account was taken of its complications in places in which smallpox was still endemic. There was a strong antivaccinationist movement in several countries, but this was based less on concern about the risks of the procedure than on moral and philosophical objections to compulsory vaccination (see Chapter 6). Among those who supported vaccination there was a reluctance to admit that there were any risks. For example, in the United Kingdom during the first quarter of this century, 103 deaths among 4 275 109 primary vaccinations were recorded as being “associated with vaccination”, but the official view was that these “may merely indicate that the child has been vaccinated before death and that death is really attributable to some current illness” (England and Wales, Ministry of Health, 1924). However, smallpox vaccination consisted in the infection of the human host with a virus that must replicate and produce lesions if it was to evoke immunity. Any such procedure that is used on a sufficiently large scale will be associated with occasional cases in which more severe lesions result. The pustular skin lesion illustrated in Plate 7.7 and the fever and lymphadenitis described earlier were the normal results of infection with vaccinia virus. Vaccination was sometimes complicated by much more severe symptoms and was sometimes fatal. These severe complications became a matter of much concern in many industrialized countries, in which endemic smallpox had been eliminated but routine vaccination programmes were maintained in order to protect the community in the event of the importation of smallpox from an endemic area.
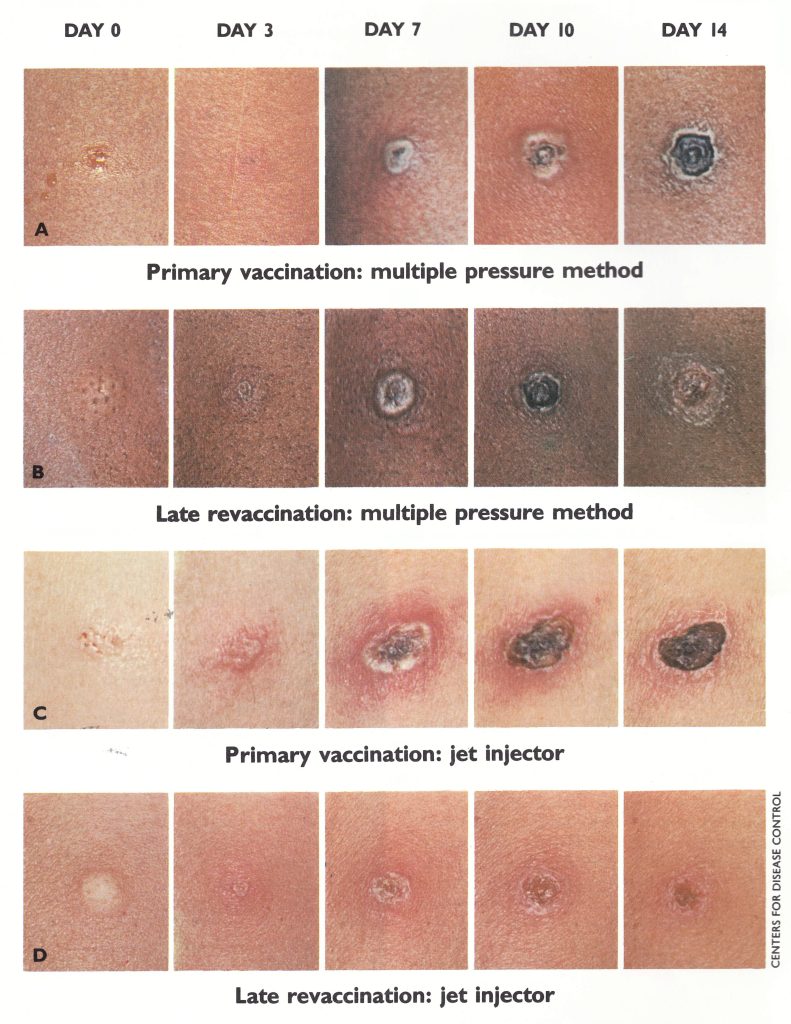
Plate 7.7. Skin reactions after primary vaccination and late revaccination (several years after primary vaccination) by the multiple pressure method and jet injector.
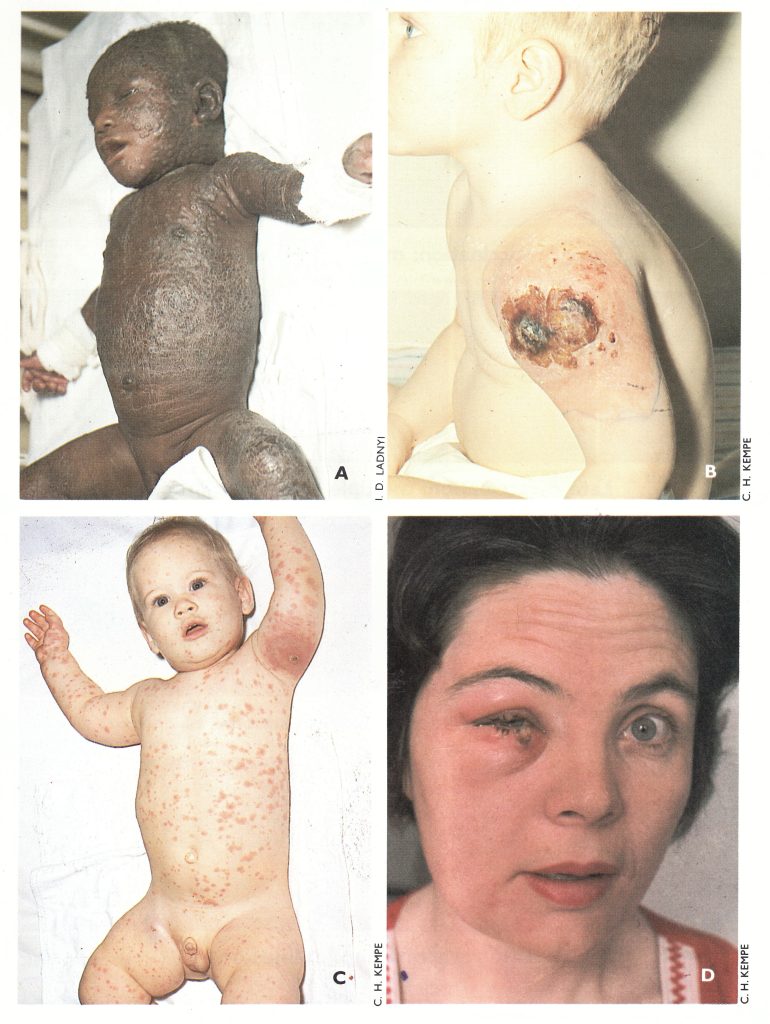
Plate 7.8. Severe complications of vaccination. A: Eczema vaccinatum in the unvaccinated contact of a vaccinated sibling. B: Progressive vaccinia, which was fatal, in a child with an immunodeficiency. C: Generalized vaccinia, 10 days after vaccination; benign course, no scarring. D: Auto-inoculation with vaccinia virus.
Types of Complication
Three groups of complications occurred among vaccinated subjects: abnormal skin eruptions, disorders affecting the central nervous system, and a variety of other rarer or less severe complications.
Abnormal skin eruptions
Three kinds of abnormal skin eruptions were recognized. Two of them, eczema vaccinatum and progressive vaccinia, were associated with abnormal host reactions; true generalized vaccinia occurred in otherwise healthy persons.
Eczema vaccinatum. This complication occurred in vaccinated persons or unvaccinated contacts who were suffering from or had a history of eczema. Either concurrently with or shortly after the development of the local vaccinial lesion (or after an incubation period of about 5 days in unvaccinated eczematous contacts) a vaccinial eruption occurred at sites on the body that were at the time eczematous or had previously been so. These areas became intensely inflamed, and sometimes the eruption later spread to healthy skin. Constitutional symptoms were severe, with high temperature and generalized lymphadenopathy, and the prognosis was grave in infants in whom large areas of skin were affected (Plate 7.8A).
It was to allow the safe vaccination of children with eczema that Kempe (1968) promoted the use of the CVI-78 attenuated vaccine (see Chapter 11), and vacciniaimmune globulin was often used to treat these cases, in both vaccinated persons and those accidentally infected. The risk of this infection remains, since military personnel continue to be vaccinated in some countries. Several cases of vaccinial infection (and thus potentially of eczema vaccinatum) in contacts of recently vaccinated military personnel were reported in Canada and the United Kingdom, and in the USA as recently as 1985 (Journal of the American Medical Association, 1985).
Progressive vaccinia (vaccinia necrosum). This complication occurred only in persons who suffered from a deficient immune mechanism, such as agammaglobulinemia, defective cell-mediated immunity or immunodeficiency associated with tumours of the reticuloendothelial system or the use of immunosuppressive drugs (see Chapter 3, Fig .3.8) . In these cases the local lesion at the vaccination site failed to heal (Plate 7.8B), secondary lesions sometimes appeared elsewhere on the body and all lesions spread progressively until—as was likely—the patient died, usually 2-5 months later.
Generalized vaccinia. As mentioned earlier, vaccinia virus produced a systemic infection in man, with transient viraemia. Very rarely a generalized vaccinial rash, sometimes covering the whole body, occurred 6-9 days after vaccination (Plate 7 .8C). The course of the individual skin lesions resembled that of the lesion at the vaccination site, but if the rash was profuse the lesions sometimes varied greatly in size. The generalized eruption usually did not have the “centrifugal” distribution which was characteristic of the rash of smallpox (see Chapter 1). Generalized vaccinia was not associated with severe immunodeficiency, and the prognosis was good.
Accidental infection. This was the most common complication of vaccination, but it was usually not serious and was certainly greatly underreported. The commonest sites, in cases that were seen by physicians, were the eyelids (Plate 7.8D), vulva and perineum. Two categories were included in a national survey carried out in the USA (see below and Table 7.3): accidental infection of secondary sites on vaccines and accidental infection of normal contacts of vaccinated persons.
Postvaccinial tonsillitis
Some Soviet authors (Braginskaya et al.,1971; Gurvich et al.,1974) regard postvaccinial tonsillitis as a not uncommon complication of primary vaccination. Gurvich et al. (1979) reported the recovery of vaccinia virus from pharyngeal swabs in 49% of children with postvaccinial tonsillitis, compared with 7% of those with no complications.
Postvaccinial encephalitis
This was the most serious complication of vaccination in persons in whom there was no contraindication for the procedure. There is an extensive literature on the subject, reviewed by Wilson (1967); the histopathology is described in Chapter 3. One difficulty in assessing postvaccinial encephalitis is that the term encompassed 3 conditions, only 2 of which were due to vaccinial infection (see box).
Concomitant disease. A temporal association does not prove causation, and some cases of encephalitis after vaccination were undoubtedly merely coincident disease due to other causes. For example, Greenberg & Appelbaum (1948) noted that of 49 cases diagnosed ante mortem as postvaccinial encephalitis, 4 out of the 8 fatal cases proved on autopsy to have been due to other causes (2 cases of tuberculous meningitis, 1 brain tumour and 1 case of hypertensive vascular disease). Some of the non-fatal cases may also have been coincidental.
Cases of encephalitis may be expected to occur in any large population of children within a defined period even if no vaccines of any kind are administered. A survey in New Jersey, USA, in 1965 showed that in the absence of vaccination 2.86 cases of encephalitis occurred per million children 1-9 years old per 28-day period; an investigation in Florida in 1968 reported a figure of 2.28 cases per million, in the same age group and during the same time span (Landrigan & Witte, 1973). Figures of a like magnitude were found in the National Childhood Encephalopathy Study in the United Kingdom, undertaken in 1976 because of widespread public and professional concern over the safety of pertussis immunization (England and Wales, Department of Health and Social Security, 1981). The results showed that the majority of the severe neurological disorders studied were not associated with recent immunization (within 28 days) and must be attributed to other causes. Nevertheless, serious neurological complications were sometimes caused by smallpox vaccination, 2 forms being recognized.
Encephalopathy and encephalitis. The pathological distinction made by Vries (1960) between encephalopathy, in infants under 2 years of age, and postvaccinial encephalitis or encephalomyelitis, with characteristic perivenous demyelination, in individuals over 2 years of age, is important and can be tentatively recognized by consideration of the age of the subject, the symptomatology and the incubation period. According to Spillane & Wells (1964), the onset in cases of encephalopathy was often violent, characterized by convulsions; hemiplegia and aphasia were common, the period of amnesia was short, and the spinal fluid, though under increased pressure, was often normal. Recovery was frequently incomplete, the patient being left with cerebral impairment and hemiplegia.
On the other hand, in the perivenous demyelinating microglial encephalitis following vaccination in subjects over 2 years of age the onset was usually abrupt, with fever, vomiting, headache, malaise, and anorexia, succeeded by such symptoms as loss of consciousness, amnesia, confusion, disorientation, restlessness and delirium, drowsiness, convulsions and coma, with incontinence or retention of urine, obstinate constipation, and sometimes meningismus. Paralysis, when it occurred, tended to be of the upper neuron type. The spinal fluid showed an increase in the concentration of protein and the number of lymphocytes. The case-fatality rate was usually about 35% and in fatal cases death usually occurred within a week. In patients who survived, recovery often set in within a few days and was complete within 2 weeks.
Weber & Lange (1961) found that almost all cases of postvaccinial central nervous system disease in children less than 2 years old and diagnosed as encephalopathy had incubation periods of 6-10 days (mean, 8.6 ± 2.3 days) from the date of vaccination (Table 7.2). By contrast, 96% of 161 cases of postvaccinial encephalitis were in individuals over 2 years of age and most had incubation periods of 11-15 days (mean, 12.3 ± 2.1 days), an interval compatible with an immunopathological basis for the syndrome. In fatal cases, the day of death in infantile encephalopathy was on average 10.5 ± 3.0 days after vaccination; the corresponding figure for encephalitis was 16.3 ± 3.9 days.
Postvaccinial encephalitis began to cause concern in the 1920s and 1930s (Hurst, 1953), when it appeared to be especially frequent in several European countries. As smallpox became less common, the importance of this serious complication of vaccination increased, mainly because there was no way of assessing the risk of postvaccinial encephalitis beforehand—unlike progressive vaccinia and eczema vaccinatum, for which there were known predisposing conditions. No means were known then, or in fact are known now, of predicting the occurrence of post-infection encephalitis, whether attributable to measles, vaccination against rabies, or smallpox vaccination (Johnson, 1982).
Other complications of vaccination
Very rarely, the fetus carried by a woman who had been vaccinated was infected in utero. Although a possible example of this was described by Jenner (1809), up to 1978 only 28 cases had been reported in the scientific literature, almost all as single case reports (A. Gromyko, personal communication, 1978). Of the 25 cases for which data are available, 21 occurred in mothers who had been vaccinated during the first 6 months of pregnancy and 4 occurred in mothers vaccinated later. In the first group, a fetus with generalized vaccinia was delivered on average 8 weeks after the vaccination, and in the second group about 4 weeks after vaccination. Many cases were fatal, the fetus being stillborn or dying a few days after birth. There is no convincing evidence that congenital malformations ever resulted from vaccination of the mother; the infected fetus usually died, but if it survived it recovered completely (Tondury & Foukas, 1964). The mother usually sustained a normal vaccination reaction; the occurrence of fetal vaccinia was additional evidence that viraemia occurred during normal vaccination.
Until smallpox was eradicated, smallpox vaccination was the most widespread immunization procedure in the world. It was thus inevitable that there should sometimes be a temporal coincidence between vaccination and the onset of certain other rare conditions of unknown etiology, such as multiple sclerosis. One rare association that probably had some etiological significance was the occurrence of a malignant skin tumour, such as melanoma, in the smallpox vaccination scar, usually many years after vaccination (Marmelzat, 1968). Vaccinial osteomyelitis was occasionally reported, the diagnosis of which was sometimes confirmed by the recovery of vaccinia virus (Sewall, 1949).
Sometimes vaccines were contaminated with pyogenic cocci, or the introduction of staphylococci into the skin during the vaccination led to localized bacterial infection.
Frequency of Complications
Apart from a multitude of case reports, estimates of the frequency of severe complications-especially postvaccinial encephalitis, which was widely recognized in the 1930s—were produced in many European countries during the period 1940-1965. These have been summarized by Wilson (1967). More useful, because they are more comprehensive, are the two surveys carried out in the USA (nation-wide and in 10 states) by the Communicable Disease Center (CDC—subsequently renamed the Centers for Disease Control), Atlanta, Georgia. Using different methodologies, these surveys were designed to estimate the incidence of all complications of smallpox vaccination in the USA for the year 1968. The methodologies of these two surveys will be described before the frequency of various complications is discussed.
Methodology of the CDC surveys
Following a nationwide retrospective survey of complications occurring during the year 1963 (Neff et al.,1967), CDC undertook a comprehensive prospective national survey, and a confirmatory, less extensive, survey through physicians in 10 selected states of the frequency of complications of smallpox vaccination in the USA in the year 1968.
National survey (Lane et al., 1969). Efforts were made to identify all possible complications, on a national scale, using 8 methods to obtain information.
-
The American Red Cross Vaccinia- Immune Globulin (VIG) distribution system supplied the names of physicians who had been provided with VIG, which was in short supply, for patients. It was distributed from some 10 centres and released to the attending physician only after a telephone conversation with the physician at the Red Cross centre and due justification of its use had been made. The attending physicians were asked for clinical and epidemiological information on their patients shortly after receiving VIG.
-
Consultants to the Red Cross VIG Program supplied the names of patients suspected of having complications of vaccination who came to their attention but did not receive VIG.
-
The National Encephalitis Surveillance Program of CDC received reports from each state of all cases of encephalitis, no matter what the cause.
-
Death certificates citing complications of vaccination were scrutinized.
-
Casual reports reaching state and territorial epidemiologists regarding vaccination complications that had not been officially reported were transmitted to CDC.
-
The Burroughs Wellcome Company supplied information on patients who received the drug metisazone (Marboran) for the treatment of complications of vaccination.
-
Reports were provided by smallpox vaccine producers who had received complaints from patients with complications allegedly attributable to their products.
-
The Viral Exanthems Unit of CDC reported on specimens submitted to its diagnostic laboratory for the confirmation of vaccinia virus.
The results obtained in this national survey, expressed in absolute numbers and as cases per million vaccinations, are set out in Tables 7.3 and 7.4 respectively.
Ten-state survey (Lane et al., 1970b). Ten states, whose populations constituted 11.6% of the total population of the USA, were chosen, and information was sought there through mail surveys directed to practising physicians. The states in question were selected partly because of their size (not too large) and partly because of the interest shown by their state epidemiologists and the presence of epidemic intelligence service officers assigned by CDC. Some 84% of physicians responded in 8 of the states and 44% and 49% respectively in the other 2. The questionnaires sought information on any patients with complications of vaccination.
The physicians’ diagnoses were accepted for minor complications, but clinical data for more severe complications were thoroughly reviewed and accepted only if the information was considered adequate. The results of this survey are presented in Tables 7.5 and 7.6.
Comparisons of data from different sources
Lack of generally accepted diagnostic criteria within countries and between countries, as well as variability in the completeness of reporting, makes it difficult to arrive at comparative judgments. Problems of various kinds were associated with each of the important complications. For example, some physicians reported patients who had erythema multiforme accompanying vaccination, or even accidental infection of a site or sites elsewhere on the body, as “generalized vaccinia”. Others restricted this term to cases with a generalized rash (Plate 7.8C).
Most important, there were no generally accepted criteria for the diagnosis of postvaccinial encephalitis. In some countries all reports of postvaccinial encephalitis were reviewed by an experienced team; in others the report was based on the views of the attending physician, who might report as encephalitis cases in which convulsions, or some other single symptom occurred, or cases with high fever and drowsiness after vaccination. In addition, there were always a few cases of encephalitis due to other causes which were merely coincident with vaccination, a problem that was dealt with in some countries by national encephalitis surveys.
As regards the USA in 1968, significantly more cases of all types of complication were found in the 10-state survey than in the national survey (compare Tables 7.4 and 7.6). This presumably resulted from the more intensive effort to discover all cases of complications made possible by the methodology of the 10-state survey, the data from which provide the better basis for comparison of complication rates in the USA with those in other countries.
Eczema vaccinatum. The CDC national survey recorded 58 cases of eczema vaccinatum among primary vaccinees, 8 among persons who had undergone revaccination and 60, with 1 death, among persons infected by contact. The frequencies were over 3 times higher than this in the 10-state survey (38.5 compared with 10.4 per million after primary vaccination and 3 .0 compared with 0 .9 per million after vaccination).
Apart from the CDC survey, few data on the frequency of this complication are available. In the mass vaccination campaign that followed the importation of smallpox into New York in 1947, during which 5-6 million people were vaccinated (but childhood eczema was accepted as a contraindication to vaccination), 10 cases of eczema vaccinatum occurred among vaccinees and 28 cases among persons who had not been vaccinated but had been in intimate contact with a recently vaccinated subject (Greenberg, 1948). There was an outbreak of smallpox in South Wales in 1962-1963 and about 900 000 persons were vaccinated. Waddington et al. (1964), who probably saw all patients with severe skin eruptions, recorded 35 cases of eczema vaccinatum (10 in vaccinees, 25 in unvaccinated contacts), with 2 deaths. The authors emphasized that the severity of eczema vaccinatum was independent of the activity of the underlying eczema, which in most patients was quiescent at the time of infection. This suggests that the virus often reached the skin by the haematogenous route, rather than as a result of the direct infection of eczematous lesions.
Progressive vaccinia (vaccinia necrosum). Eleven cases of progressive vaccinia were recorded in the CDC national survey. Four of these-all of whom had received vaccinia-immune globulin were also identified in the 10-state study. In the national survey 6 cases were seen in revaccinated individuals, all of whom showed evidence of serious pre-existing illness, including leukaemia, Hodgkin’s disease and lymphoma. Only 1 case occurred in a child, a 22-month-old infant with a congenital immune disorder—Bruton’s hypogammaglobulinemia. There were 4 deaths, despite treatment with vaccinia-immune globulin and metisazone.
Conybeare (1964a) reported 8 cases of progressive vaccinia (with 7 deaths) among 5 million vaccinees in England and Wales over the years 1951-1960. All occurred after the primary vaccination of infants less than 6 months old, most of whom were probably suffering from congenital immunodeficiencies.
Generalized vaccinia. Being a relatively mild condition, generalized vaccinia was recorded much more frequently in the 10-state than in the national survey in the USA (241.5 per million primary vaccinations compared with 23.4 per million among the same group in the national survey). Generalized vaccinia was rare in persons undergoing revaccination.
Conybeare (1964a) recorded 162 cases in about 5 million vaccinations and revaccinations carried out in England and Wales in 1951-1960. The lesion at the vaccination site matured normally, but 9-14 days after vaccination papular lesions appeared elsewhere on the body; these became vesicular and then pustular before scabbing. On any individual patient, the lesions usually varied considerably in size. One hundred and fifty cases occurred in primary vaccinees and 106 in infants less than 1 year old. The accompanying constitutional disturbance was mild.
Accidental infection. Not surprisingly, many cases of accidental infection were not reported and therefore did not figure in the CDC national survey. The rates in the 10-state survey were about 20 times higher (25.4 per million primary vaccinations in the national survey; 529.2 per million in the 10-state survey). Very few cases occurred after vaccination.
A special kind of accidental infection on which some statistics were collected was that associated with ocular lesions (Plate 7.8D). Sedan et al. (1953) recorded 19 cases among 850 000 vaccinees in Marseilles in 1952 (22.3 per million), and Waddington et al. (1964) noted 11 cases of vaccinia on the eyelid among 900 000 vaccinees in South Wales in 1962. The most comprehensive report (Ruben & Lane, 1970), derived from cases detected by the perusal of requests for vaccinia-immune globulin addressed to the American Red Cross, records 348 cases, most of which came to light in the years 1963 and 1968 when special national surveys had been conducted. There were also some cases that occurred in 1964, 1965 and 1969. Corneal involvement occurred in 22 cases and produced a much higher rate of residual ocular defect (Table 7.7).
Postvaccinial encephalitis. Although comparisons of data were difficult because of the variability of the criteria for making the diagnosis and the extent of reporting, the frequency of postvaccinial encephalitis varied greatly in different countries. Table 7.8 presents data from 5 countries (9 series of studies) which illustrate dramatically the large differences reported, especially for postvaccinial encephalitis after the primary vaccination of individuals over 2 years of age. All the statistics presented probably include a few cases of encephalitis that were merely coincident with, rather than caused by, vaccination, but the differences in frequencies are so great that they could not have been due solely to diagnostic or statistical eccentricities.
Postvaccinial encephalitis was much more common in European countries than in the USA, and more common in the countries of continental Europe than in the United Kingdom. There were also differences in the age incidence. In most countries postvaccinial encephalitis was considered mainly a risk of primary vaccination in adolescents or young adults, but in the USA the higher incidence of encephalopathy in infants under the age of 1 year led to a recommendation that compulsory vaccination should be postponed until the 2nd year of life. More detailed analysis of the data used in Table 7.8 for various age groups showed that the frequency of postvaccinial encephalitis was greatest in the age group 6—2 years.
Wilson (1967) summarized data from many sources which demonstrate that the case fatality rate for most series of cases of postvaccinial encephalitis was about 30%, with a reported low of 9% and a high of 57%.
Although host factors were certainly involved in a disease believed to have an immunopathological basis (see Chapter 3), the strain of virus appears to have been important in determining the frequency of postvaccinial encephalitis. The New York City Board of Health strain, which was the principal strain used in the USA, appears to have been the least likely of the widely used strains to cause it, closely followed by the Lister strain. Indeed, the frequencies recorded by Neff et al. (1967) and Lane et al. (1969) for individuals over 1 year of age were so low that they could almost be accounted for by the coincident occurrence of encephalitis due to other causes. However, the rate was much higher in the 10-state survey reported by Lane et al. (1970b).
Although there is no single laboratory test that can satisfactorily determine the virulence of vaccinia virus for man or assess the likelihood that vaccination will cause postvaccinial encephalitis, investigations by Marennikova et al. (1969) in mice and rabbits were consistent with the following classification of several strains in terms of their pathogenicity: mildly pathogenic— New York City Board of Health and EM-63; moderately pathogenic—Lister, Bern and Patwadangar; and highly pathogenic— Copenhagen, Tashkent and Ikeda (see Chapter 11, Table 11.20).
Several different strains of vaccinia virus were used in Belgium and the Netherlands during the 1950s; the incidence of postvaccinial encephalopathy in infants dropped substantially after the Lister strain was introduced in the Netherlands in 1963 (Table 7.9). In Switzerland and Austria the number of reported cases of postvaccinial encephalitis fell dramatically after the Bern strain was replaced by the Lister strain in 1962 and 1967 respectively (C. Fleury, personal communication, 1967; Berger & Heinrich, 1973).
Contraindications to Vaccination
During smallpox eradication programmes in areas in which smallpox was endemic, WHO recommended that no contraindications to vaccination should be envisaged—for two reasons. First, the risk of smallpox infection was considered to be significantly greater than the risk of complications and, secondly, most vaccinations were carried out by personnel without medical training, who could not be expected to diagnose correctly conditions such as eczema. However, it was recommended that very sick individuals should not be vaccinated, because their subsequent death might be attributed to vaccination.
On the other hand, in countries in which smallpox was not endemic, special attention was paid to contraindications to vaccination. Instructions as to what physical conditions should be regarded as constituting contraindications varied according to national health legislation, but the following 4 conditions were generally accepted:
-
Immune disorders.
These included agammaglobulinemia, hypogammaglobulinemia, neoplasms affecting the reticuloendothelial system and the use of immunosuppressive drugs. Such conditions substantially enhanced the risk of the most serious complication of vaccination—namely, progressive vaccinia
-
Eczema.
Vaccination was contraindicated in persons who were currently suffering from eczematous skin lesions or whose family included an individual with eczema, because of the risk of eczema vaccinaturn in either the vaccinee or the family member concerned. However, it was always difficult to exclude from vaccination persons who had suffered from eczema in the past but did not have active skin lesions; such persons sometimes experienced eczema vaccinatum.
-
Pregnancy.
It is a general principle to avoid immunization in pregnancy. This policy was followed with smallpox vaccination, because of the risk (admittedly rare) of fetal vaccinia, a usually fatal condition.
-
Disorders of the central nervous system.
In order to minimize the risk of postvaccinial encephalitis, disorders of the central nervous system in the person to be vaccinated, or in family contacts, were accepted as contraindications in many countries. However, there is no evidence that the exclusion of such subjects affected the incidence of that complication.
In addition to the above 4 generally accepted contraindications, in some countries vaccination was considered undesirable for persons with an acute infectious disease (influenza, measles, etc.), a chronic infectious disease (e.g., tuberculosis) or an allergic constitution, or for persons who had been immunized against other diseases such as yellow fever, cholera, or typhoid fever on the same day as the scheduled smallpox vaccination. The justifications were, respectively, the hypothetical risk of complications arising from infection with vaccinia virus in persons whose physical condition was not normal at the time, and the belief that an interaction between the different immunizing agents might prevent the occurrence of a proper immunological response.
In most countries in which smallpox was endemic, leprosy was also present. Since primary smallpox vaccination often precipitated reactions such as erythema nodosum leprosum or neuritis in leprosy patients (Webster, 1959), health officers in these areas had to determine whether patients with lepromatous symptoms should be vaccinated. The WHO recommendation was that leprosy, including lepromatous leprosy, should not be a contraindication in such circumstances, since the risk of smallpox infection was substantially greater than that posed by the reactions concerned (Browne & Davis, 1962).
Prevention and Treatment of Complications
Immunoprophylaxis and immunotherapy
Human antivaccinia immunoglobulin (vaccinia-immune globulin), prepared from the plasma of recently vaccinated persons, became available in the early 1950s. Potency requirements were that it should contain at least 500 IU of vaccinia antibody per ml, in terms of the International Standard for Anti-Smallpox Serum (WHO Expert Committee on Biological Standardization, 1967).
It was thought that since some of the more severe complications of vaccination were caused by defects in antibody production, the administration of vaccinia-immune globulin at the time of vaccination might prevent such complications, without inhibiting an active immune response. Nanning (1962) reported on a field experiment in the Netherlands in which about 100 000 army recruits were vaccinated, half of them given vaccinia immune globulin and the other half a placebo. There were 3 cases of central nervous system complications in the vaccinia-immune globulin group and 13 in the placebo group. No study of similar magnitude was conducted elsewhere, but in the Netherlands vaccinia immune globulin was subsequently used in all primary vaccinations of adults (Polak, 1973). Vaccinia-immune globulin was also recommended for the prevention of eczema vaccinatum, being administered at the time of vaccination, whenever an eczematous child had to be vaccinated because of special circumstances, such as exposure to a case of smallpox (Sharp & Fletcher, 1973).
Kempe (1960) analysed 300 cases of severe complications of vaccination treated with vaccinia-immune globulin. There was no evidence that it influenced the course of postvaccinial encephalitis, but observations on 132 treated cases of eczema vaccinatum suggested that it reduced the mortality from the usual level of 30-40% to 7%. It was also useful in treating 28 cases of accidental autoinoculation with vaccinia virus and led to the prompt cessation of the development of new lesions in 62 patients with generalized vaccinia. However, vaccinia-immune globulin was much less effective in treating cases of progressive vaccinia, because the major immunodeficiency in these cases was in cell-mediated immunity. Vaccinia-immune globulin administration was associated with the production of neutralizing antibody in cases of eczema vaccinatum, generalized vaccinia and progressive vaccinia. Kempe treated all subjects who presented, holding that the cases were few in number and occurred only sporadically and that the early indications of the value of the treatment made it ethically difficult to withhold its use. In another study, Sharp & Fletcher (1973) observed rapid improvement within 48 hours of the administration of vaccinia-immune globulin to cases of generalized vaccinia, eczema vaccinaturn, and severe local vaccinial reactions.
Chemotherapy
In the 1950s and 1960s, several chemicals were found to inhibit vaccinia virus replication in cell cultures and in laboratory animals. In addition to being investigated for the treatment of smallpox (see Chapter 1), they were tested for the treatment of complications of vaccination. The most effective substances in experimental systems were derivatives of cytosine arabinoside, rifampicin, a urea derivative of diphenyl sulfone, and a thiosemicarbazone derivative termed metisazone (N-Methylisatin-β-thiosemicarbazone). There was no evidence that any of them was effective, although Brainerd et al. (1967) reported that the use of metisazone had a beneficial effect in progressive vaccinia, a condition in which vaccinia-immune globulin was useless.
Reconsideration of Vaccination Policies in Non-endemic Countries
As smallpox transmission was interrupted in various countries, the occasional untoward results of routine vaccination gradually emerged as a public health problem, which concerned both the general public and health administrators. In essence, in smallpox-free countries routine vaccination was maintained or mass vaccination campaigns were undertaken in order to maintain a protective barrier against the spread of smallpox in the population, but this community benefit was gained at the expense of a few individuals who suffered from the complications of vaccination. For example, the last case of smallpox in the USA was reported in 1949 but routine vaccination continued until 1972. The national survey of vaccination in the USA during 1963 carried out by CDC (Neff et al.,1967) showed that among the 14 million persons who were vaccinated against smallpox in that year (6.2 million primary vaccinations and 7.8 million revaccinations), there were 132 cases of severe complications and 7 deaths. From the results of this survey, it could be roughly estimated that between 1949 and 1972 there may have been about 3000 cases of severe vaccinial complications, with some 150 deaths.
Lane & Millar (1969) compared the projected number of deaths from routine vaccination in the USA for the 30 years from 1970 to 2000 with what might be expected if this procedure were replaced by vaccination of only high-risk groups (military draftees, hospital workers and international travellers), in combination with ring vaccination of contacts (at most 5000 per importation) and occasional mass vaccination campaigns to control smallpox outbreaks resulting from importations. They concluded that there might be 210 vaccination deaths if routine vaccination were continued, compared with 60 deaths from the vaccination of high-risk groups. There would have to be 21 separate importations of smallpox to make up the difference of 150 deaths, but no cases had been imported into the USA during the previous 20 years and the risk was constantly diminishing. Dick (1973) carried out a similar kind of calculation for the United Kingdom and concluded that on the basis of the mortality due to vaccination in that country there would be 150 such deaths if routine vaccination were continued—a figure that would be equalled by the number of smallpox deaths only if there were 50 importations of variola major over the 30 years between 1970 and 2000, compared with the 13 which had occurred during the preceding 20 years. It is clear that routine vaccination in the United Kingdom and the USA, and by analogy in other countries which had long since eliminated smallpox, entailed a cost which had been acceptable when there was a substantial risk of smallpox, either endemic or imported, but which was not justified when the risk had become very slight.

Plate 7.10. C. Henry Kempe (1922-1984). As Professor and Chairman of the Department of Pediatrics at the University of Colorado Medical Center, Denver, USA, Kempe was a major figure in the investigation of complications of vaccination in children in the USA during the 1950s and 1960s. He also participated in the extensive investigations of smallpox carried out with Dr A. R. Rao in Madras, India, over the same period.
Lane & Millar (1969) compared the projected number of deaths from routine vaccination in the USA for the 30 years from 1970 to 2000 with what might be expected if this procedure were replaced by vaccination of only high-risk groups (military draftees, hospital workers and international travellers), in combination with ring vaccination of contacts (at most 5000 per importation) and occasional mass vaccination campaigns to control smallpox outbreaks resulting from importations. They concluded that there might be 210 vaccination deaths if routine vaccination were continued, compared with 60 deaths from the vaccination of high-risk groups. There would have to be 21 separate importations of smallpox to make up the difference of 150 deaths, but no cases had been imported into the USA during the previous 20 years and the risk was constantly diminishing. Dick (1973) carried out a similar kind of calculation for the United Kingdom and concluded that on the basis of the mortality due to vaccination in that country there would be 150 such deaths if routine vaccination were continued-a figure that would be equalled by the number of smallpox deaths only if there were 50 importations of variola major over the 30 years between 1970 and 2000, compared with the 13 which had occurred during the preceding 20 years. It is clear that routine vaccination in the United Kingdom and the USA, and by analogy in other countries which had long since eliminated smallpox, entailed a cost which had been acceptable when there was a substantial risk of smallpox, either endemic or imported, but which was not justified when the risk had become very slight.
Aside from the risks inherent in routine vaccination, complications posed a severe problem when a country in which smallpox was not endemic conducted mass vaccination for the containment of an outbreak following an importation. For example, in 1963 smallpox was imported into Sweden from an unidentified Asian country. Twenty-seven cases resulted. A voluntary mass vaccination campaign was performed in which about 300 000 persons in central Stockholm were vaccinated in the course of a few weeks (Strom & Zetterberg, 1966). As a result of the vaccination campaign, 77 persons suffered vaccinial complications (neurological complications, 14; carditis, 7; eczema vaccinatum, 7; and postvaccinial exanthema, 49). Without vaccination, of course, there would have been many more than 27 cases of smallpox.
These experiences provoked substantial debate among research workers and health planners during the 1960s as to whether it was justified to continue routine smallpox vaccination in countries in which smallpox was no longer endemic (Benenson, 1974; Dick, 1966, 1971, 1973; Kempe & Benenson, 1965; Langmuir, 1974). This was not a new debate. In the geographically isolated country of Australia compulsory vaccination had been adopted in only 2 of the 6 states, and Cumpston (1914) calculated that only about 30% of the children born in Australia between 1860 and 1910 had ever been vaccinated. “Conscience clauses” introduced into legislation enacted in the states in which compulsory vaccination of infants was operative reduced the proportion of newborn infants vaccinated each year to less than 10% by 1923 (Cumpston & MacCallum, 1925). The responsible state and Commonwealth health officers repeatedly inveighed against this laxity in vaccination, but the state governments took no action. The situation in New Zealand was similar. The threat of smallpox was never perceived as great enough to justify the enforcement of an unpopular measure, and even the large outbreak of variola minor in New South Wales in 1913-1917 (see Chapter 8) was not enough to cause the parliament of that state to pass a compulsory vaccination act.
By the late 1960s, several countries of Europe and those of North America had already discontinued compulsory routine smallpox vaccination programmes, but it was still common for paediatricians to recommend that children should be vaccinated, usually during the 2nd year of life. The reason for this recommendation was that it was believed that complications were less common when primary vaccination was carried out in childhood; moreover, the International Health Regulations, as applied in many non-endemic countries, made the possession of a valid smallpox vaccination certificate almost a sine qua non for international travel.
Complications: the Overall Picture
The complications arising from vaccination against smallpox clearly posed a serious public health problem in the non-endemic countries. Several steps were taken to mitigate its effects, notably the substitution of “milder” strains of vaccinia virus, such as the New York City Board of Health and the Lister strains, that caused fewer complications, the postponement of vaccination until the 2nd or 3rd year of life (which greatly reduced the risk of postvaccinial encephalopathy), the use of vaccinia-immune globulin for the treatment of complications and the development of more attenuated strains of vaccinia virus (see Chapter 11). Nevertheless, there was an irreducible minimum rate of complications, and even normal primary vaccination was associated with unpleasant local and general reactions. The ultimate solution was the discontinuation of vaccination, which could be done without argument only if smallpox were eradicated globally. In addition, universal discontinuation of smallpox vaccination would result in a substantial saving in health budgets covering vaccination programmes and the medical care of patients suffering from complications. These considerations had begun to preoccupy the world scientific community during the 1950s and 1960s and constituted one of the reasons for promoting the development of the global smallpox eradication programme.
PROGRAMMES FOR VACCINATION AND REVACCINATION
Vaccination and Revaccination of the General Public
By the middle of the 20th century, smallpox vaccination had become part of the routine immunization programme of most countries in the world. The recommended age for vaccination varied from country to country, but it was common practice for primary vaccination to be done during the first 2 years of life and revaccination performed when children entered and left primary school. The first report of the WHO Expert Committee on Smallpox (1964) recommended that in countries in which smallpox was endemic, primary vaccination should be carried out as early as possible, preferably in the neonatal period, and repeated about 12 months later. For revaccination, a 5-10 year interval was recommended for the nonendemic countries and one of 3 years for the endemic countries. In North America and some European countries, hospital staff were, in theory, obliged to be vaccinated every 3 years, but as smallpox faded from memory this requirement was rarely enforced.
Because of the relatively high incidence of postvaccinial encephalopathy among infants in the USA revealed by the CDC surveys, it was recommended that primary vaccination should not ordinarily be carried out during the first 2 years of life. On the other hand, public health experts in many European countries, noting that the highest incidence of postvaccinial encephalitis was in adolescents undergoing primary vaccination, recommended that this immunization should be performed during the 2nd year of life.
Simultaneous Vaccination with Several Antigens
Smallpox vaccination was often carried out at approximately the same time as immunization with diphtheria and tetanus toxoid or with inactivated antigens of pertussis, typhoid and poliomyelitis since it was considered that such a practice was safe and did not interfere with the immune responses to each agent (Winter et al ., 1963). However, it was important not to use the same arm for the injection of bacterial antigens as had been used for the inoculation of smallpox vaccine a few days earlier, in order to avoid the severe febrile reactions that occasionally resulted (A. S. Benenson, personal communication, 1982).
Studies were carried out during the 1950s and 1960s to find out whether the combined use of various live vaccines would affect their safety and efficacy. The simultaneous administration of oral polio vaccine and smallpox vaccine caused no particular problems either in terms of increased side-effects or reduced take rates (Karchmer et al.,1971; Winter et al., 1963). Other studies showed that combinations of smallpox vaccine, BCG vaccine (Lin, 1965), yellow fever vaccine (Meers, 1960), and measles vaccine (Breman et al.,1975) were safe and produced good immune responses. In these studies, separate vaccines were administered simultaneously at different sites. However, when smallpox, measles and yellow fever vaccines were combined and inoculated by jet injection into the same site, there were no untoward effects but the immune response for yellow fever virus was reduced (Meyer et al.,1964b). When smallpox, yellow fever, measles and diphtheria-pertussis- tetanus (DPT) vaccines were administered at separate sites by jet injection, the response to smallpox vaccine was unaffected but the measles seroconversion rate dropped from 89% (if DPT was not given) to 70% (Ruben et al., 1973). However, this apparent decrease was not observed in later studies, and simultaneous administration of the antigens commonly used in developing countries in the 1970s was generally regarded as safe and efficacious (Foege & Foster, 1974). In practice, the commonest combinations were smallpox and yellow fever vaccines (in much of francophone Africa) and simultaneous immunization against smallpox and measles, in the CDC-assisted programme in western and central Africa (see Chapter 17). Smallpox and BCG vaccinations were given simultaneously but in opposite arms in many countries in central and eastern Africa.
Vaccination of International Travellers
The 1944 International Sanitary Convention (United Nations Relief and Rehabilitation Administration, Health Division, 1945) amended the previous Convention, formulated in 1926, and devised a vaccination certificate (Plate 7.11), which required that the vaccinial reaction should be inspected 8-14 days after the primary vaccination and 48 hours after revaccination. A certificate was not valid for more than 3 years.


Plate 7.11. The first International Certificate of Vaccination against Smallpox developed by the International Sanitary Convention in 1944.
Plate 7.12. A: The form proposed to the Fourth World Health Assembly in 1951, by the committee drafting the International Sanitary Regulations.
Plate 7.12. B: The revised form approved by the Health Assembly.
On 9 April 1951, a special committee on the drafting of the International Sanitary Regulations (the former International Sanitary Conventions) met in Geneva (World Health Organization, 1951a). During the Committee’s sessions, the original draft form of the vaccination certificate was extensively amended (World Health Organization, 1951b; Plate 7.12A). As revised (Plate 7.12B), this form was adopted by the Fourth World Health Assembly on 25 May 1951. When, in 1981, the vaccination of travellers against smallpox was no longer a requirement a stamp was used to cancel the smallpox vaccination certificate but the form was still in use for other vaccination requirements. In 1983 it was replaced by a new form lacking any provision for smallpox vaccination (see Chapter 28, Plate 28.1).
As early as 1945, the authorities concerned with the International Sanitary Convention stipulated that, in relation to vaccination certificates, “recent vaccination shall be taken as meaning evidence of successful vaccination not more than 3 years or less than 14 days previously, or evidence of an immune reaction” (United Nations Relief and Rehabilitation Administration, Health Division, 1945). In 1956, the WHO Committee on International Quarantine noted that although these rules “may lack a firm scientific basis, they are nevertheless administratively expedient in order to avoid delay to persons on an international voyage” (World Health Organization, 1956). The maximum interval of 3 years, in particular, lacked a carefully documented basis, although Marsden (1936) had discovered no cases of variola minor in persons vaccinated less than 7 years earlier, and before that Hanna (1913) had found that variola major was rare and mild in children under 4 years of age who had been vaccinated in infancy, but severe (case-fatality rate, 45%) in unvaccinated children in that age group (see Chapter 1, Table 1.11). Subsequently, Rao (1972) reported that only 90 out of 2181 cases of variola major in children under 4 years of age occurred in those with a vaccination scar.
Nyerges et al. (1972) reported that the levels of neutralizing antibody 3 years after the last revaccination were as high as those found 3 weeks after primary vaccination. Taking the absence of rises in the level of haemagglutinin-inhibiting antibodies after revaccination as evidence of resistance to viral infection, these investigators found that such resistance increased with each successive 3-year revaccination, and suggested that the revaccination interval of health personnel at high-risk could be prolonged beyond 3 years after 2 successful revaccinations. This result suggests that for international travellers the 3-year interval was conservative, but, as the WHO Committee on International Quarantine had concluded, it was administratively convenient, and continued to be accepted as long as vaccination certificates were required.
The day on which the certificate became valid after primary vaccination or revaccination was often debated at meetings of the WHO Committee on International Quarantine. The times agreed on in 1951—namely, 8 days after primary vaccination and the day after revaccination—were consistent with the earliest expected development of the immune response. Modifications, such as 12 or 14 days after the date of both primary vaccination and revaccination, were suggested, and the obligatory inspection of the vaccinial reaction was questioned. In a sense, this debate is a reflection on the ways in which international committees sometimes operate, especially in the absence of a real problem.
It is clear from these discussions that national health administrations attached considerable importance to the use of vaccination certificates, regarding them as one of the most important measures for preventing importations of smallpox. However, although all agree that because possession of a vaccination certificate was mandatory more travellers were vaccinated than would otherwise have been the case, the importance of this requirement is challenged by some critics, who point out that it was subject to abuse. For example, certificates were not always examined by the appropriate health officer at the point of entry, and if a passenger had transferred at an intermediate port in a smallpox-free country his vaccination certificate might not have been examined at all. Worse, perhaps, were instances of deliberate falsification, or the provision of certificates by travel agencies to unvaccinated customers. Persons who had an apparently valid certificate might nevertheless contract smallpox if the vaccine or technique used had been faulty.
The situation was at its worst during the 1960s and 1970s, a period of greatly increased air travel, which reduced the travelling time between even the most distant endemic and non-endemic countries to a matter of hours. In earlier times, the inspection of all crew and passengers on ships from overseas ports to detect patients with smallpox had proved a useful method of limiting importations, especially into countries situated at some distance from endemic areas.


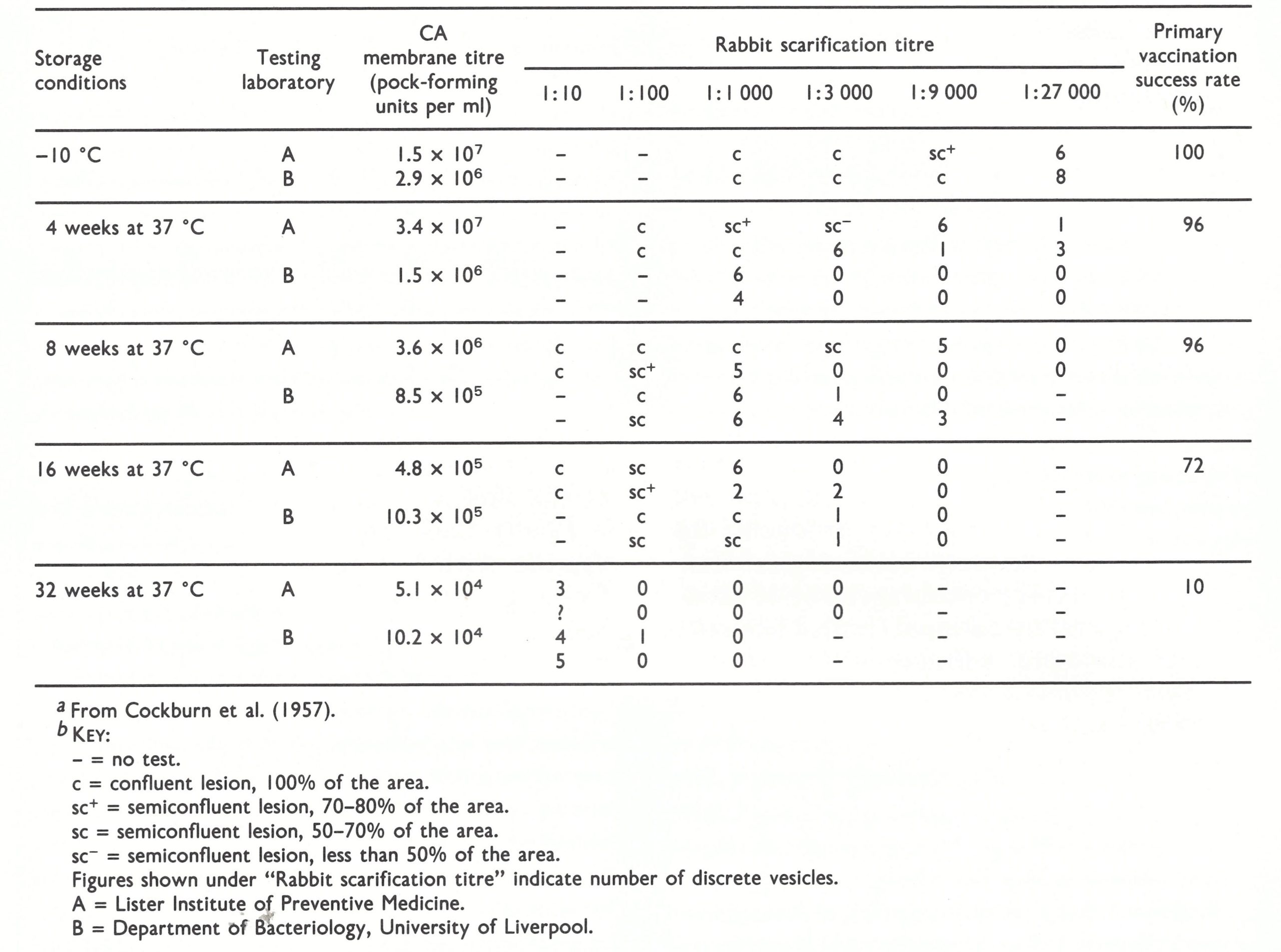
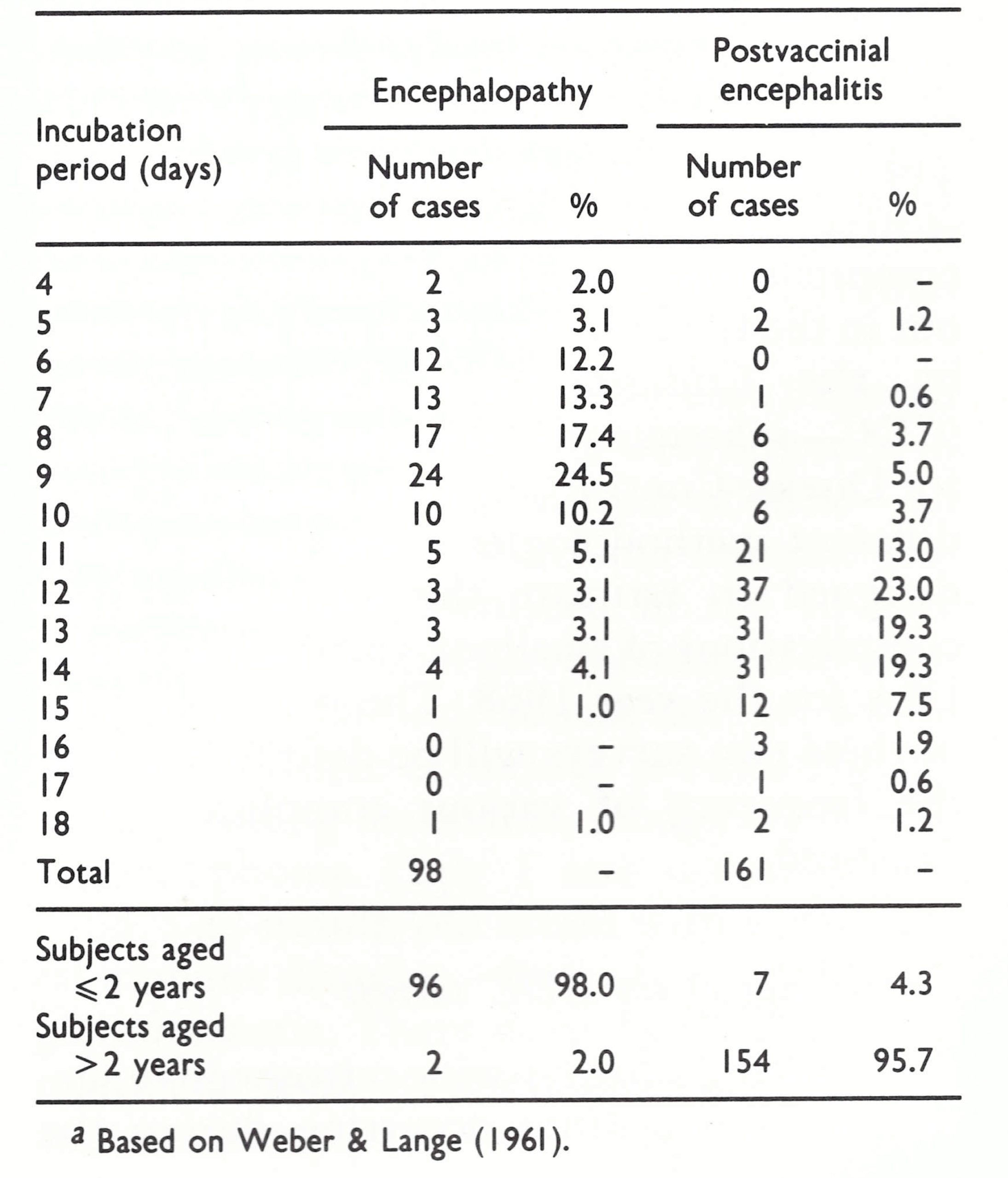
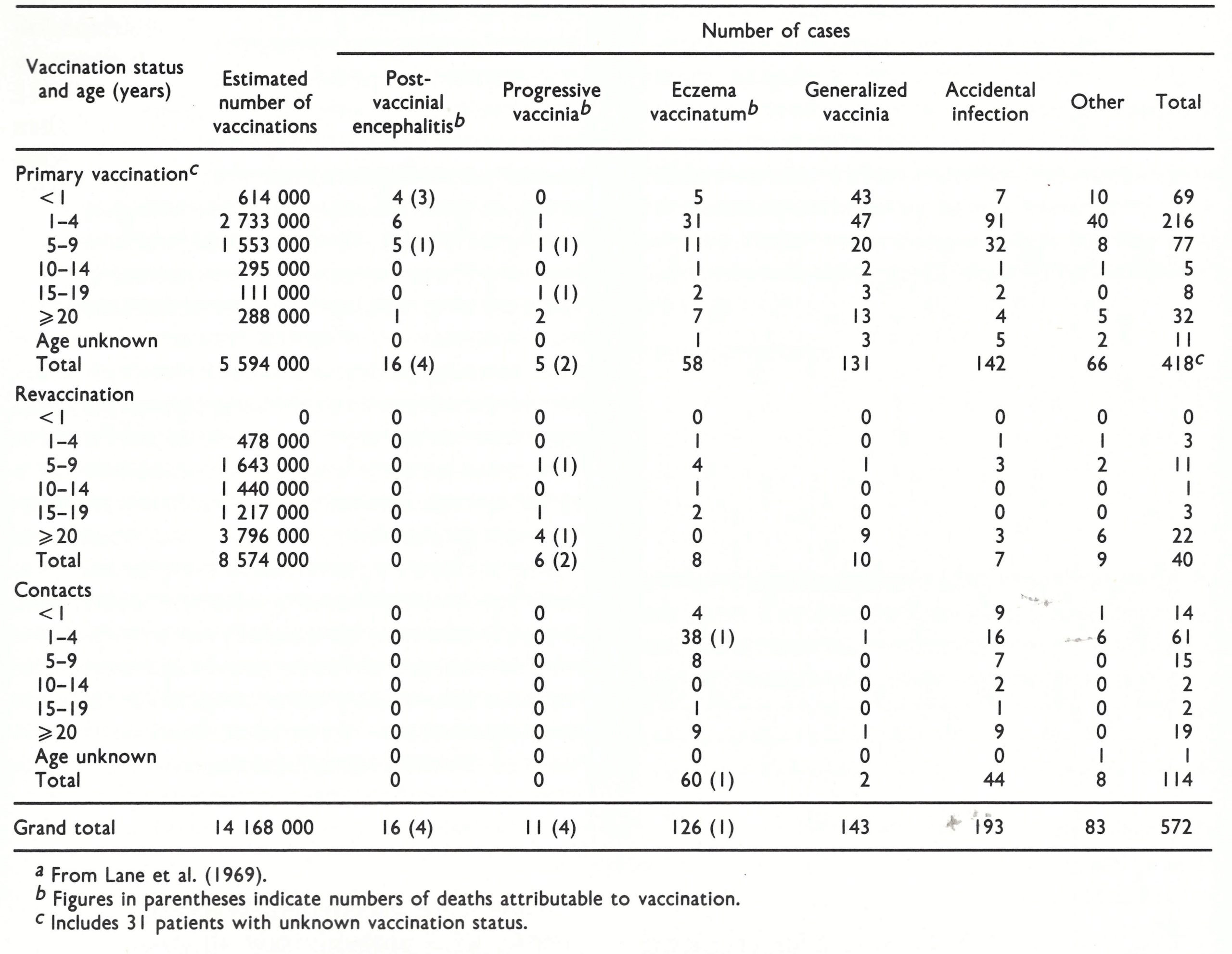
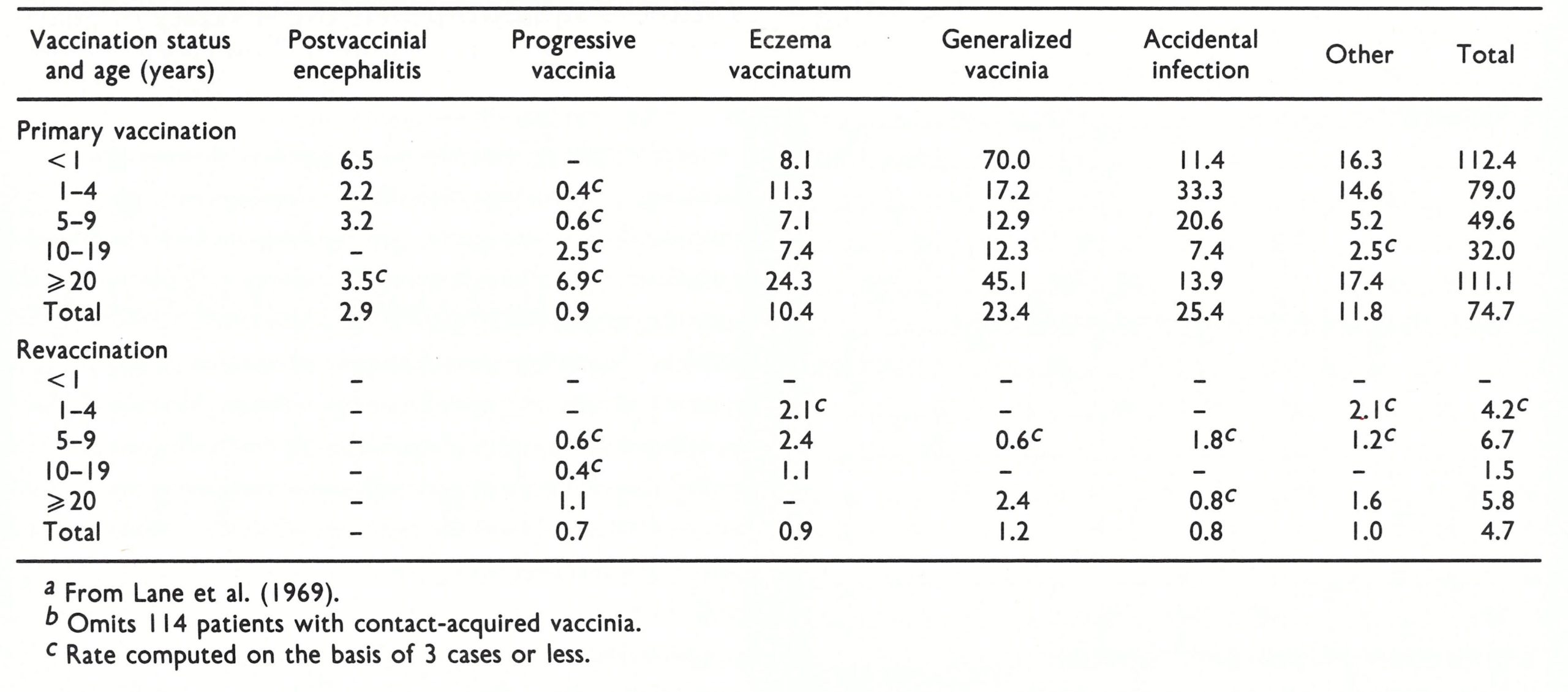
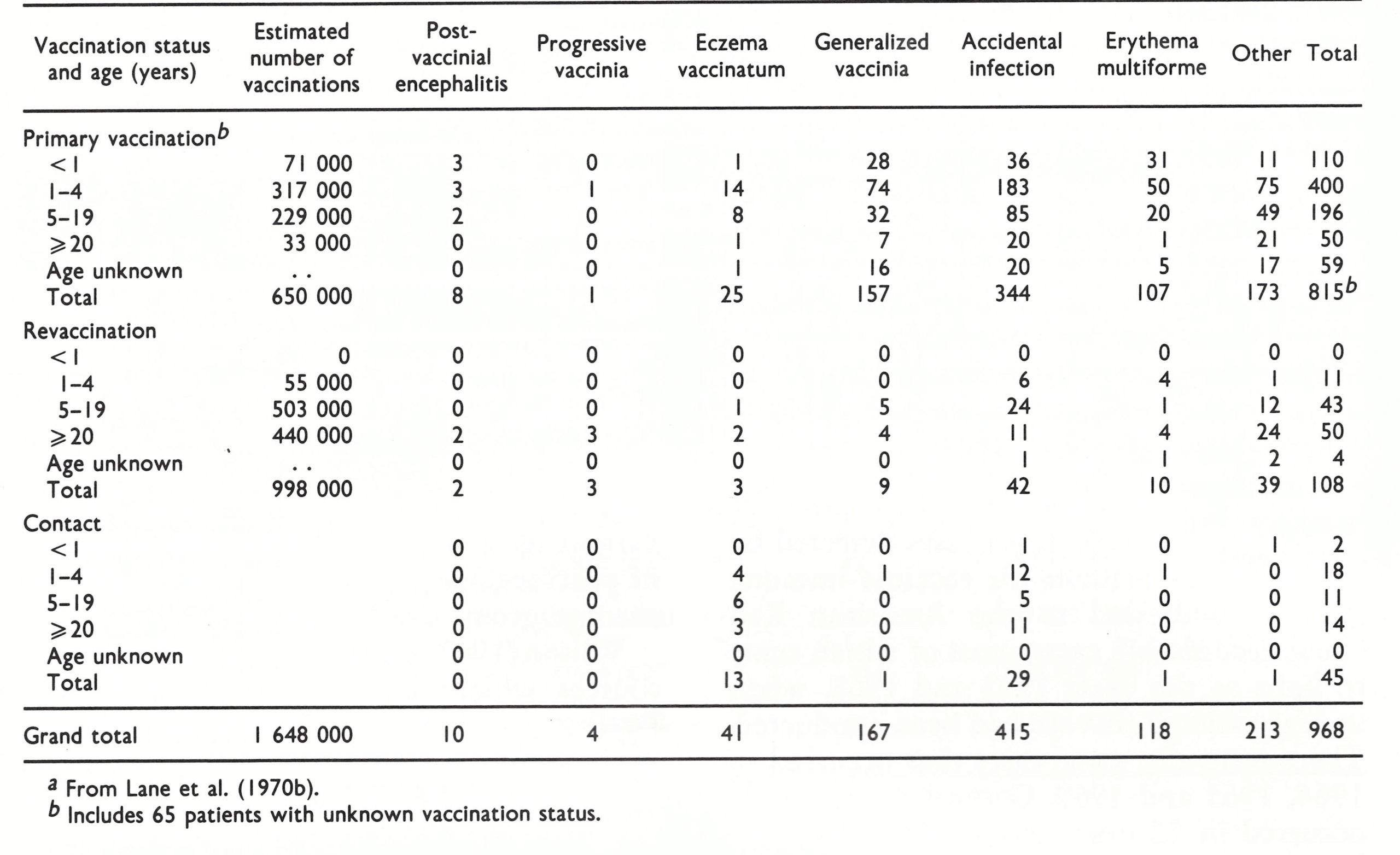
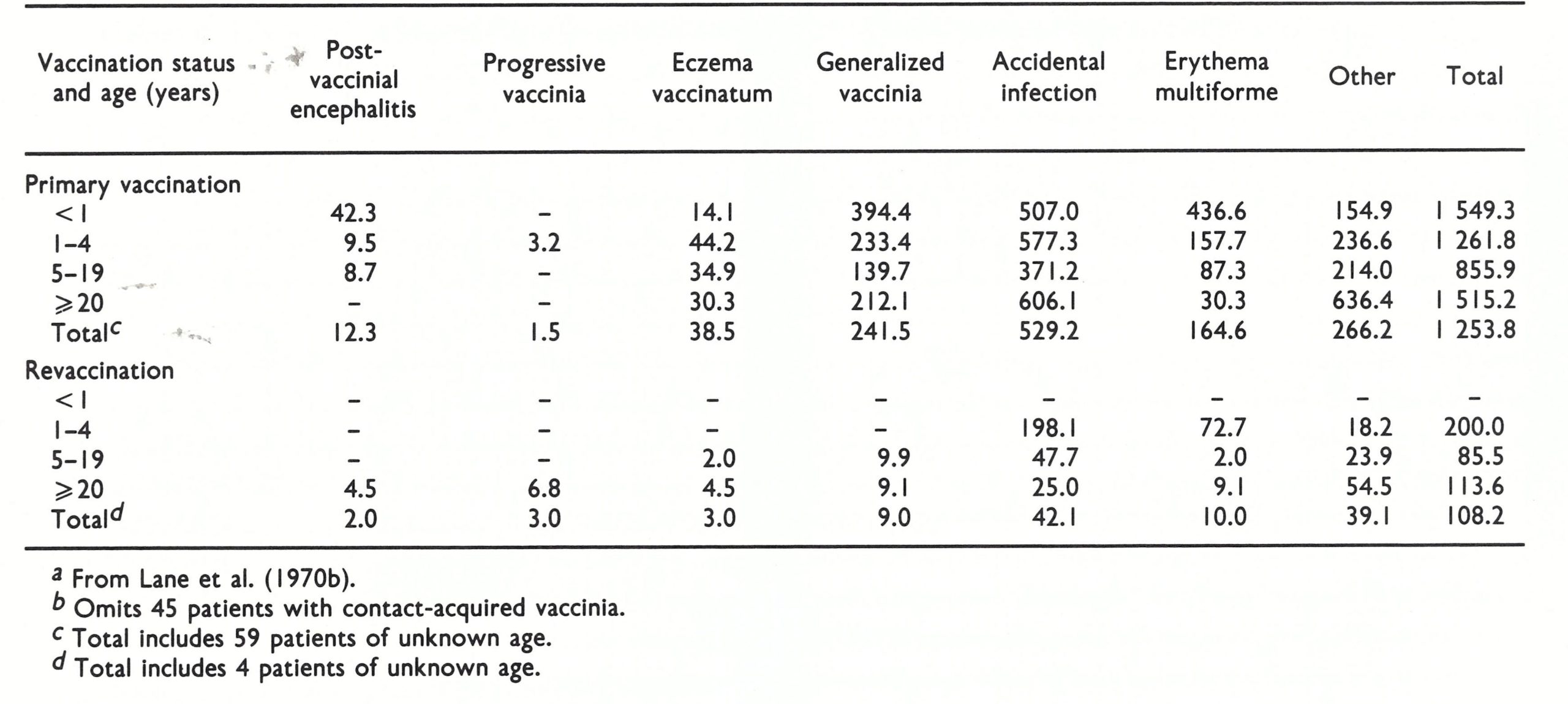
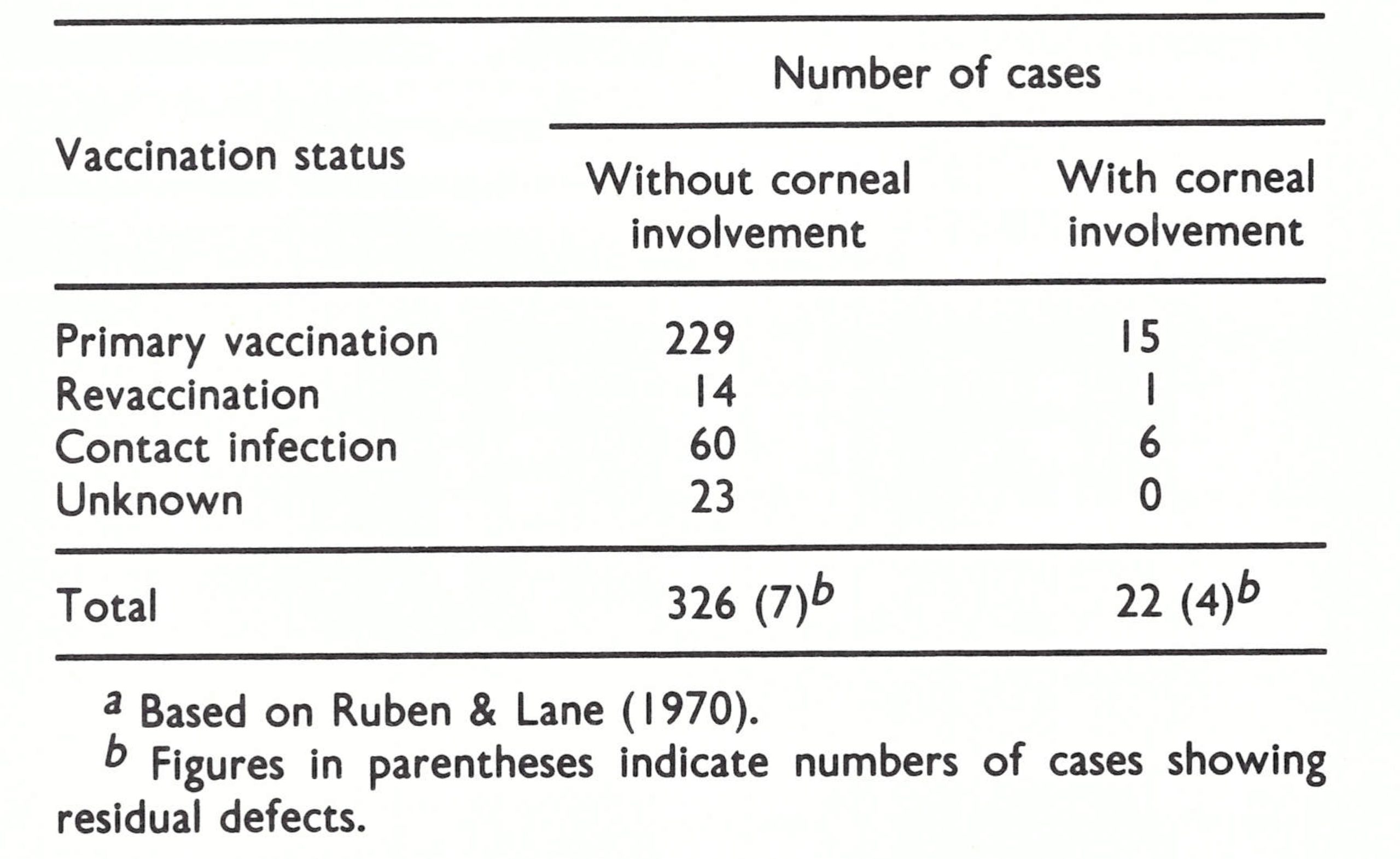
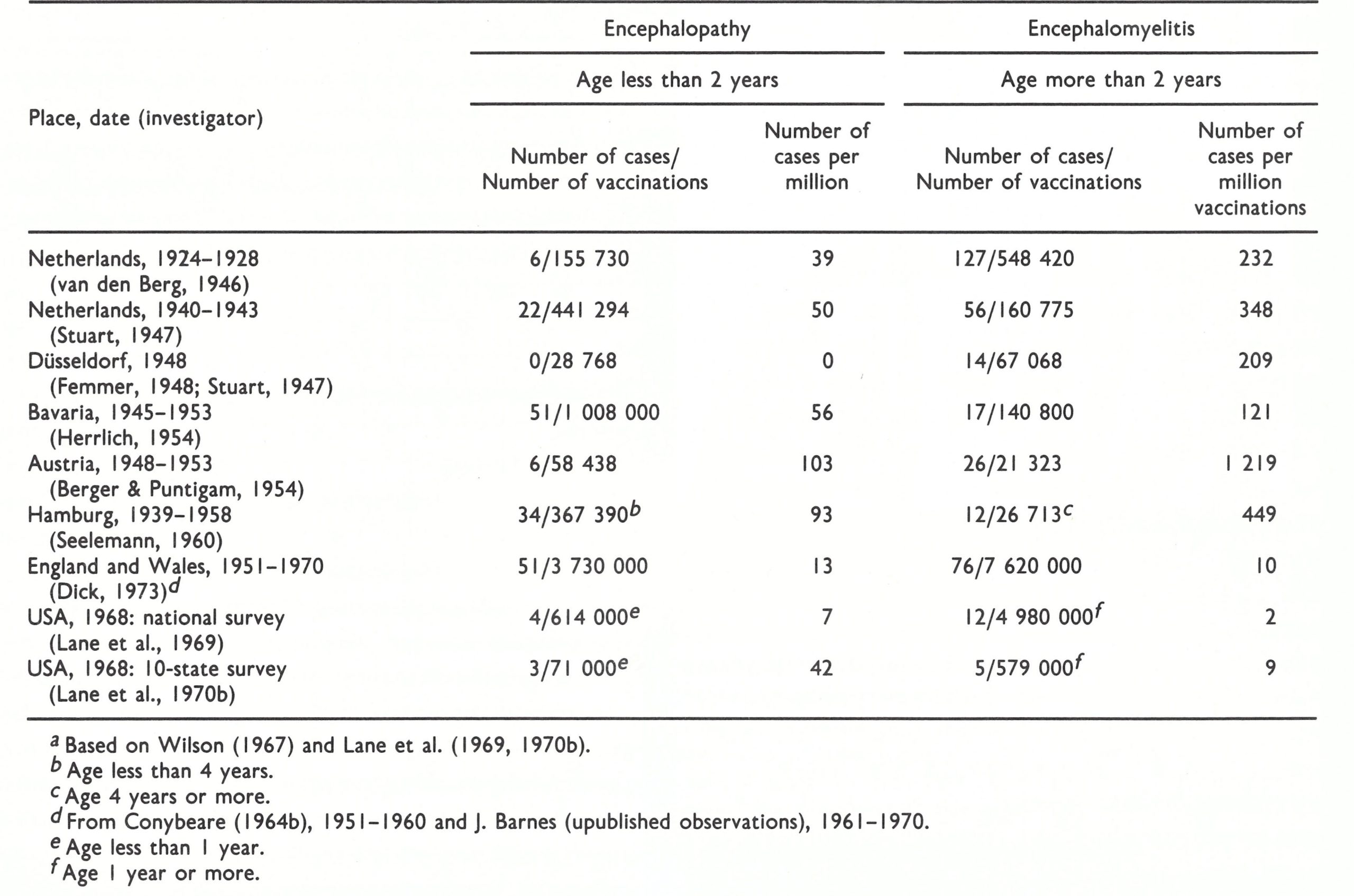


rxIc
August 26, 2025816902 146116Thanks for the excellent post against your weblog, it genuinely provides me with a appear about this subject.??;~.?? 474946