Chapter 11:
Smallpox vaccine and vaccination in the Intensified Smallpox Eradication Programme
INTRODUCTION
Vaccination against smallpox had been practised in virtually every country of the world, and in many on a large scale, when the Intensified Smallpox Eradication Programme was launched in 1967 . By its use, smallpox had already been eliminated as an endemic disease from all but 31 countries, which constituted the hard core of the smallpox problem . It was clear that, in order to implement the programme, one of the first tasks of the WHO Smallpox Eradication unit would be to ensure that enough vaccine was available, of sufficiently high titre and sufficiently heatstable, to ensure that potent vaccine could be delivered to those needing vaccination in any place in the world, however remote and however adverse the environmental conditions.
Traditionally, smallpox vaccine had been distributed in liquid form, although laboratories in France and the Netherlands East Indies (now Indonesia) had produced airdried or freeze-dried vaccines from the 1920s onwards. Unless refrigerated, liquid vaccine did not retain its potency for more than a few days whereas, until it was reconstituted, freeze-dried vaccine remained highly potent for over a month at ambient temperatures, even under tropical conditions. During the period after the Second World War, freezedried vaccine prepared in France was being used in francophone Africa and by the mid- 1950s producers in several countries had developed freeze-dried vaccine production on a commercial scale . From 1959 onwards somewhat larger quantities of freeze-dried vaccine began to be used for smallpox vaccination in tropical countries, but its extensive use throughout the world dates from 1967- 1968. After 1971 it was the only kind of vaccine used in any country engaged in a national smallpox eradication programme.
In May 1980 the Thirty-third World Health Assembly, after it had declared that smallpox had been eradicated throughout the world, recommended that smallpox vaccination should be discontinued, except for investigators at special risk . By 1985, smallpox vaccine production had been stopped in most countries and in no country in the world is smallpox vaccination routinely conducted in the civilian population. Vaccine reserves are being kept for emergencies by WHO and by the health authorities of some 20 countries (see Chapter 28) . Thus smallpox vaccination has gone full circle. Introduced by Jenner in 1798, it came to be used all over the world until, with the eradication of the disease that it was designed to control, its use has now been abandoned, except for military personnel in some countries . Jenner’s prediction, in the paper reproduced in Chapter 6, that “. . . the annihilation of the Small Pox, the most dreadful scourge of the human species, must be the result of this practice” has been fulfilled, making smallpox vaccination redundant.
An interesting sequel to this history of the rise and fall of smallpox vaccination is that vaccinia virus is currently showing considerable promise as a vector for genes specifying protective antigens against a variety of other infectious agents (see box) . If the results of current research fulfil expectations, “smallpox” vaccine may make a comeback as a vehicle for providing simultaneous active immunization against a number of selected viral or protozoal diseases . It has the advantages of heat stability and ease of administration, but the risk of complications will need to be carefully weighed and perhaps a more attenuated strain sought (Quinnan, 1985) . In this chapter, various aspects of vaccine production and vaccination will be described in terms of practices that came into operation after the Intensified Smallpox Eradication Programme had begun in 1967. Many features were common to the periods before and after 1967, but that year marked a turning-point, in that the global efforts to eradicate the disease necessitated modifications in production methods and in quality control of the vaccine, and also in vaccination techniques . In this chapter, unless otherwise specified, “vaccine” means the freeze-dried smallpox vaccine .
Vaccination against smallpox had been practised in virtually every country of the world, and in many on a large scale, when the Intensified Smallpox Eradication Programme was launched in 1967 . By its use, smallpox had already been eliminated as an endemic disease from all but 31 countries, which constituted the hard core of the smallpox problem . It was clear that, in order to implement the programme, one of the first tasks of the WHO Smallpox Eradication unit would be to ensure that enough vaccine was available, of sufficiently high titre and sufficiently heatstable, to ensure that potent vaccine could be delivered to those needing vaccination in any place in the world, however remote and however adverse the environmental conditions.
Traditionally, smallpox vaccine had been distributed in liquid form, although laboratories in France and the Netherlands East Indies (now Indonesia) had produced airdried or freeze-dried vaccines from the 1920s onwards. Unless refrigerated, liquid vaccine did not retain its potency for more than a few days whereas, until it was reconstituted, freeze-dried vaccine remained highly potent for over a month at ambient temperatures, even under tropical conditions. During the period after the Second World War, freezedried vaccine prepared in France was being used in francophone Africa and by the mid- 1950s producers in several countries had developed freeze-dried vaccine production on a commercial scale . From 1959 onwards somewhat larger quantities of freeze-dried vaccine began to be used for smallpox vaccination in tropical countries, but its extensive use throughout the world dates from 1967- 1968. After 1971 it was the only kind of vaccine used in any country engaged in a national smallpox eradication programme.
In May 1980 the Thirty-third World Health Assembly, after it had declared that smallpox had been eradicated throughout the world, recommended that smallpox vaccination should be discontinued, except for investigators at special risk . By 1985, smallpox vaccine production had been stopped in most countries and in no country in the world is smallpox vaccination routinely conducted in the civilian population. Vaccine reserves are being kept for emergencies by WHO and by the health authorities of some 20 countries (see Chapter 28) . Thus smallpox vaccination has gone full circle. Introduced by Jenner in 1798, it came to be used all over the world until, with the eradication of the disease that it was designed to control, its use has now been abandoned, except for military personnel in some countries . Jenner’s prediction, in the paper reproduced in Chapter 6, that “. . . the annihilation of the Small Pox, the most dreadful scourge of the human species, must be the result of this practice” has been fulfilled, making smallpox vaccination redundant.
An interesting sequel to this history of the rise and fall of smallpox vaccination is that vaccinia virus is currently showing considerable promise as a vector for genes specifying protective antigens against a variety of other infectious agents (see box) . If the results of current research fulfil expectations, “smallpox” vaccine may make a comeback as a vehicle for providing simultaneous active immunization against a number of selected viral or protozoal diseases . It has the advantages of heat stability and ease of administration, but the risk of complications will need to be carefully weighed and perhaps a more attenuated strain sought (Quinnan, 1985) . In this chapter, various aspects of vaccine production and vaccination will be described in terms of practices that came into operation after the Intensified Smallpox Eradication Programme had begun in 1967. Many features were common to the periods before and after 1967, but that year marked a turning-point, in that the global efforts to eradicate the disease necessitated modifications in production methods and in quality control of the vaccine, and also in vaccination techniques . In this chapter, unless otherwise specified, “vaccine” means the freeze-dried smallpox vaccine .
Use of Vaccinia Virus as a Vector for Other Genes
The large genome of vaccinia virus contains a substantial amount of redundant DNA, as judged by the ability of mutants which have undergone large deletions to replicate in cultured cells and in animals (see Chapter 2) . Furthermore, recombination and marker rescue occur in doubly infected cells . These properties opened the way for the construction of vaccinia virus hybrids, which contain genes for specified polypeptides of other viruses, bacteria or protozoa.
Several methods have been used to construct such hybrids . One method of general applicability (Mackett et al ., 1984) is to construct plasmid vectors which contain the vaccinia virus thymidine kinase (TK) gene interrupted by selected restriction endonuclease cleavage sites placed adjacent to an appropriate promoter . The continuous coding sequence for a foreign (non-vaccinial) protein is then inserted in the TK gene plasmid so that the transcriptional start site of the vaccinia promoter is adjacent to that of the foreign gene. Cells in which vaccinia virus is replicating are then transfected with this plasmid, and homologous recombination takes place at the vaccinia TK gene . Since these recombinants are TK-negative, they can be readily selected and then tested for the presence and expression of the foreign gene .
Genes for antigens which play a role in protective immunity against several important viral diseases were tested in 1983 and 1984 and a high level of expression was found in both cultured cells and laboratory animals . This method has considerable potential for providing cheap and effective vaccination against several different human diseases that are common in developing countries, as well as against some diseases of domestic animals .
VACCINE REQUIREMENTS FOR THE INTENSIFIED SMALLPOX ERADICATION PROGRAMME
Providing the Finance
The Intensified Smallpox Eradication Programme was launched in 1967 after the Nineteenth World Health Assembly in 1966 had voted an allocation of US$2 .4 million from the WHO regular budget. It was estimated that approximately 300 million persons would have to be vaccinated annually in the endemic and adjacent countries . At 1-2 US cents per dose, this would have cost US$3- 6 million for the vaccine alone, if WHO had provided all the vaccine required . Three other sources of vaccine were, however, already 11. VACCINATION IN THE INTENSIFIED PROGRAMME 541 available : (1) several of the endemic countries were producing large amounts of vaccine for local use ; (2) following a resolution of the Twelfth World Health Assembly in 1959, a few producer countries were already making donations to WHO ; and (3) vaccine was being supplied to developing countries through a number of bilateral aid programmes. WHO therefore decided that vaccine additional to that produced by the endemic countries or provided through bilateral aid and necessary for the implementation of the global smallpox eradication programme should be supplied entirely by voluntary donations . The annual allocation for smallpox eradication in the WHO regular budget could then be used exclusively for technical assistance : consultants, training, research and certain supplies and equipment including transport. This policy was retained throughout the Intensified Smallpox Eradication Programme . At the time (1967) it was thought that donated vaccine would constitute a relatively small proportion of the total requirement-namely, that needed to close the gap between the perceived need and available supplies . As the present chapter relates, the actual situation was found to be much more complex and difficult .
Donations of Vaccine, 1958-1966
Apart from encouraging the establishment of national smallpox eradication programmes, the only important consequence of the resolution on global smallpox eradication adopted by the Twelfth World Health Assembly in 1959 (see Chapter 9) was the initiation of donations of smallpox vaccine, both to WHO and on a larger scale in bilateral assistance programmes. Between 1958 and 1966 the vaccine donated to WHO totalled 47 062 500 doses (from Jordan, Madagascar, Mexico, the Netherlands, the Philippines, Switzerland, Thailand, the USSR and the United Kingdom) . Of this amount, 25 million doses of vaccine had been pledged by the government of the USSR in 1958 when it proposed that WHO should undertake a global smallpox eradication programme ; they were delivered between 1960 and 1964. In bilateral donations between 1961 and 1966 the USSR also provided some 700 million doses of vaccine, which met the annual requirements of Afghanistan, Burma, India and some other countries during this period .
Shortfalls in Vaccine-Quality and Quantity
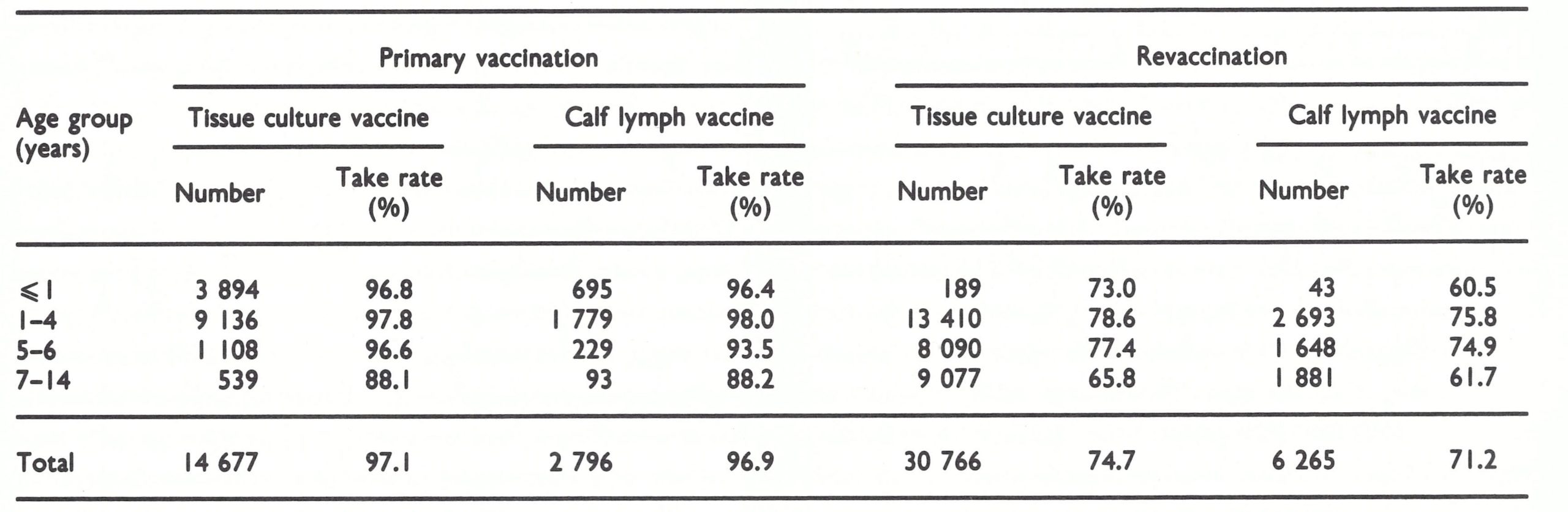
Since, although smallpox vaccine had been in use since 1800, standardized vaccine production procedures and reliable assay methods for quality control were not developed until the 1950s (see Chapter 7), the quality of the vaccine varied significantly in different countries. Although the quality of vaccines donated to WHO had been tested since 1959, until 1967 such vaccines accounted for an average of only 7 million doses per year (Table 11 .1), or less than 2% of the vaccine needed in endemic countries . The remaining vaccine was provided by domestic production or through bilateral aid programmes and by a number of producers who sold vaccine to the endemic countries . The titre of most of these vaccines was unknown to WHO and, even when known, the tests used to determine it were of uncertain reliability and conducted mainly by the production laboratories themselves. In many laboratories, the potency was tested by vaccinating 10 children and determining the proportion of takes, or by using the rabbit skin scarification method, which was imprecise; in addition, few laboratories tested the vaccine for heat stability.
Thus WHO was faced, not with the relatively small problem of closing a gap between the total requirement and supplies obtained from existing vaccine donors and local production of vaccines in the endemic countries, but with the much larger task of developing a production-donation system to serve most of the endemic countries and their neighbours.
As is shown in Table 11 .1, vaccine donations to WHO were substantially increased after the Intensified Smallpox Eradication Programme was initiated in 1967. In the early days of the programme, one of the difficulties in increasing the amounts donated was that laboratories producing smallpox vaccine in most countries had only a limited capacity that was barely enough to meet their national programme requirements, let alone help to supply the very much larger amounts of vaccine needed for a global eradication programme.
Before 1967 provisions for testing the vaccine were complicated and slow, arrangements for shipping vaccine from the producers to the recipient country were complex and resulted in many delays, and WHO had no access to reserve supplies of vaccine for immediate shipment in an emergency . The problem was overcome when distribution was centralized through the Smallpox Eradication unit in Geneva .
WHO SURVEY OF VACCINE PRODUCERS
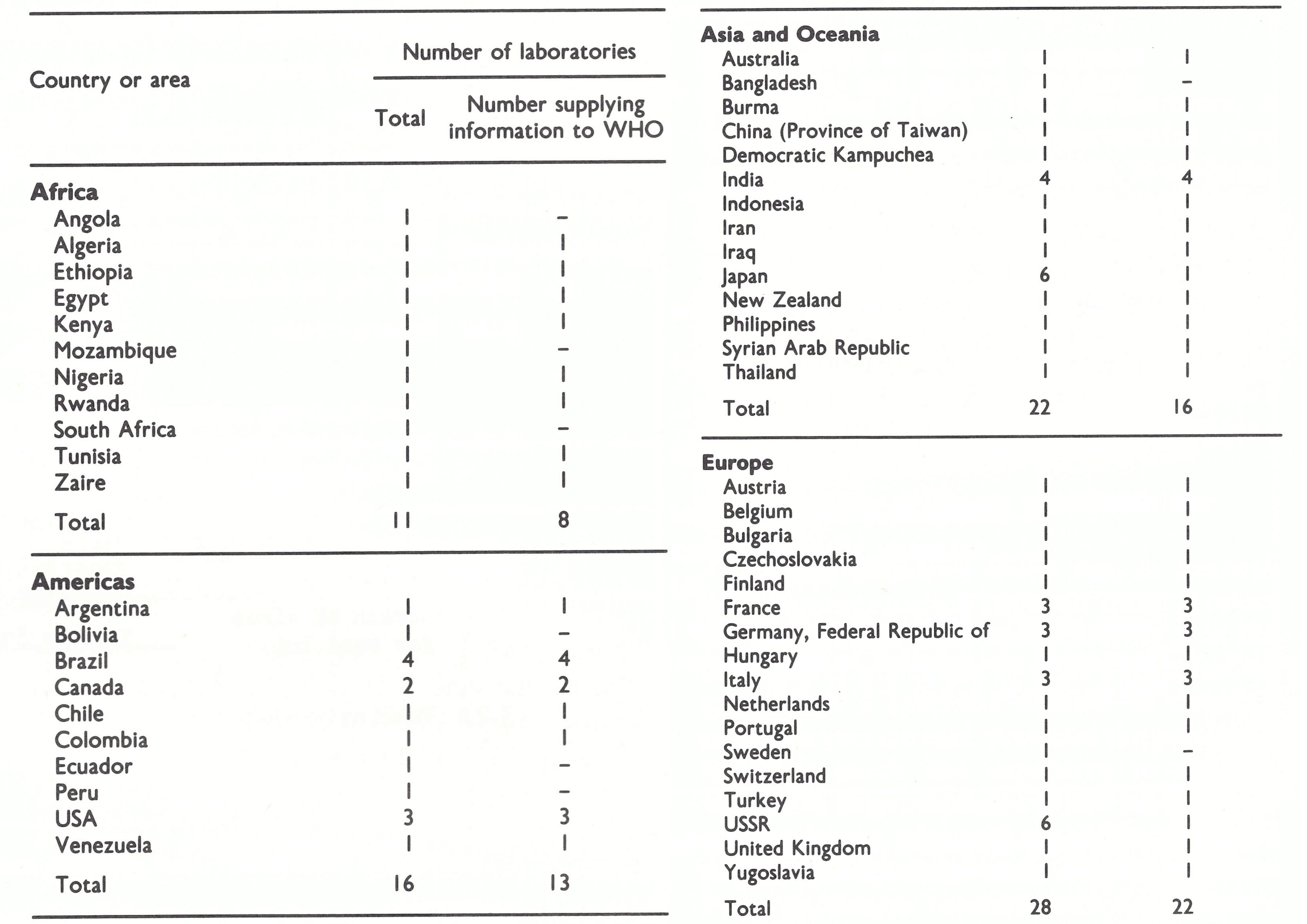
Faced with the problems of meeting the basic requirement for smallpox eradication namely, adequate amounts of a potent and stable vaccine and a system for distributing it to the countries in which it was neededthe Smallpox Eradication unit decided to conduct a survey of vaccine producers throughout the world . The objective was to gather information on the quality of the smallpox vaccines then in use and on the production capacity, production methods and the kind of quality control practised in different countries . This would provide a factual basis for introducing into vaccine production and use the changes needed to ensure that sufficient quantities of vaccine of suitable quality were available for the global eradication programme. Arita, then medical officer with the unit, took primary responsibility for this task.
In February 1967 the unit tried to identify all present or planned production facilities for freeze-dried vaccine and dispatched questionnaires (Plate 11.1) to the laboratories concerned, requesting information on the method of freeze-drying, the strain of vaccinia virus used, the method of growing the virus, the number of doses per vial or ampoule, and the results of testing . Of 77 laboratories contacted in 52 countries, 59 in 44 countries replied (Table 11.2) . The information obtained by means of the survey is summarized below.

Plate 11.1. Sample questionnaire circulated by the Smallpox Eradication unit in 1967 to all identified smallpox
vaccine producers in countries accessible to WHO.
Methods of Vaccine Production
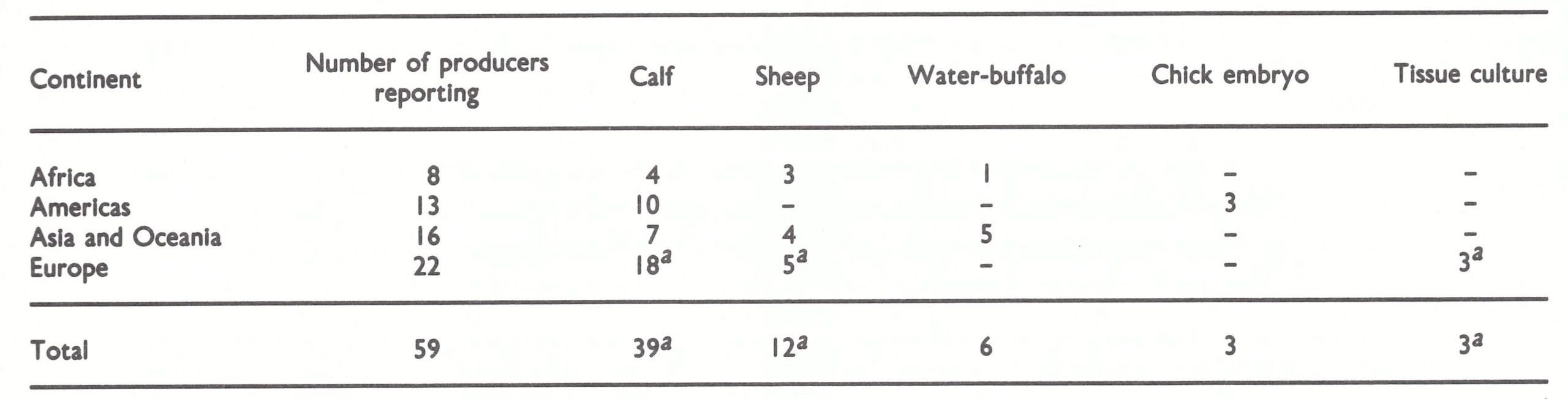
In 51 of the 59 laboratories, vaccinia virus was harvested from the skin of calves (39) or sheep (12); 6 laboratories were producing vaccine from water buffaloes, 3 on the chorioallantoic (CA) membrane of chick embryos and 3 in tissue culture (bovine embryo fibroblasts). These results confirmed that smallpox vaccine of animal skin origin was by far the most extensively used throughout the world, in both developed and developing countries (Table 11.3).
Lyophilization equipment used in the different laboratories had been produced by at least 11 different manufacturers-in Czechoslovakia, France, the German Democratic Republic, the Federal Republic of Germany, Japan, the United Kingdom and the USA. 3 laboratories (Table 11.4) . Furthermore, it was doubtful whether all the strains were correctly described. For example, in a laboratory in Africa, it was found that the strain used was a mixture of vaccinia and cowpox viruses. Moreover, the different passage histories of what were nominally the same strains of vaccinia virus undoubtedly resulted in substantial differences in their biological properties.
Strains of Vaccinia Virus
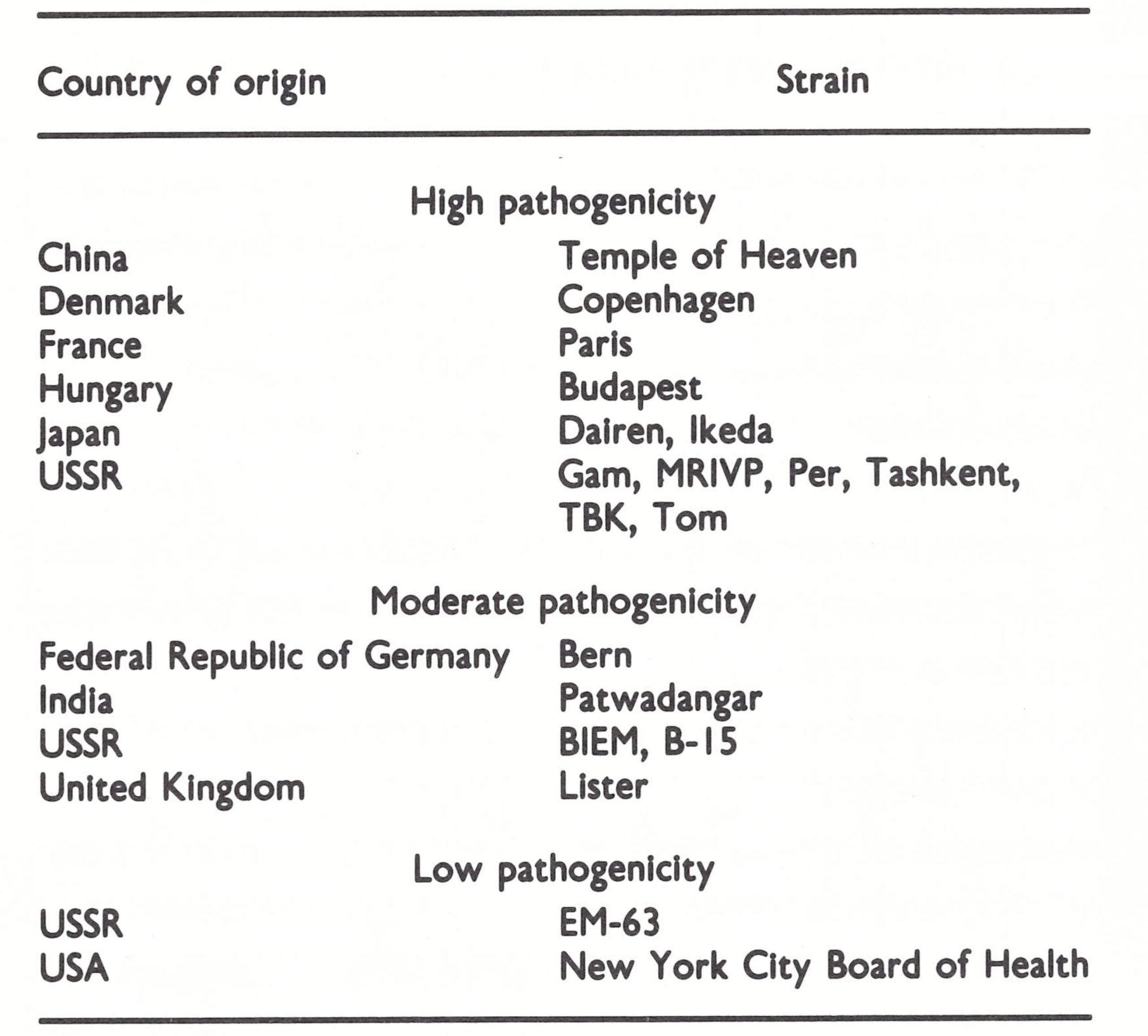
Many different strains of vaccinia virus were in use for vaccine production, although it is probable that some of these had a common ancestry. Of the 59 laboratories that responded, 23 employed the Lister strain, 7 the New York City Board of Health strain, and 7 the Paris strain . The remaining 22 laboratories used a number of different strains, none of which was used by more than a One laboratory employed both calves and sheep as vaccinifers as well as cultured bovine embryo fibroblasts, while 2 employed calves and bovine embryo fibroblasts. Table 11.4. WHO survey, 1967 : strains of vaccinia virus used for vaccine production a Includes the following strains: Aosta, Bohemia, Bordeaux, Chambon, Hamburg, Ikeda, Massachusetts 999, Vienna. 3 laboratories (Table 11.4) . Furthermore, it was doubtful whether all the strains were correctly described. For example, in a laboratory in Africa it was found that the strain used was a mixture of vaccinia and cowpox viruses. Moreover, the different passage histories of what were nominally the same strains of vaccinia virus undoubtedly resulted in substantial differences in their biological properties .
Number of Doses per Container
In most countries there was, in general, little communication between those producing vaccine and those administering it in the field . Government laboratories were called on to produce a specified number of doses of smallpox vaccine each year, and the producers found it much less expensive to dry the vaccine in large ampoules. Likewise, commercial producers, who sold vaccine by the dose, preferred to package it in large containers . Few producers understood the logistic problem that this posed for field staff, nor did they consider that, after reconstitution, much of the vaccine in large ampoules might be kept for a long time before being used (and thus lose potency), or else be discarded.
Ampoules and vials of various sizes were used in the different laboratories. Most producers regarded a “dose” as being 0 .01 ml of reconstituted vaccine . About one-third of the laboratories supplied vaccine in containers of 2 or more different sizes, holding from 10 to more than 500 doses. More than 70% of vials on which the number of doses was indicated contained 100 doses or more.
Initial Potency
Information was requested from each laboratory on the initial potency, heat stability and bacterial counts of the last 3 batches of vaccine produced . Of the 59 laboratories, only 31 reported that the initial potency of the vaccine met WHO standards (see Chapter 7) in all 3 production batches on which they were asked to report (Table 11 .5). Even this figure may have been an overestimate, since the assays were carried out in the production laboratories and were not independently verified. In 48 laboratories, potency was measured on the CA membrane, 1.0 X 108 rock forming units per ml or higher being regarded as a satisfactory titre. Of these 48 laboratories, 2 recorded the potency in such a manner as to suggest that this assay procedure was not well understood, while 5 recorded results determined by the rabbit scarification technique, which was much less accurate than pock counting on the CA membrane.
Heat Stability
The results of tests to determine the heat stability of the vaccine were much less satisfactory than those for initial potency, although heat stability was very important since most of the vaccine required for the global smallpox eradication programme was to be used in tropical regions . Only 16 out of 59 laboratories recorded satisfactory results for all lots tested (titre of not less than 10 80 pock-forming units per ml after incubation of dried vaccine at 37°C for 4 weeks); 23 laboratories reported some or all lots to be unsatisfactory (Table 11.6), while 15 failed to report on heat stability, probably because the necessary tests were never carried out. Of the batches with unsatisfactory heat stability, the titre of about half was reduced to less than 10 7.5pock-forming units per ml after heating.
Bacterial Count
Since most smallpox vaccine was produced in animal skin, bacteriological sterility was not attainable. WHO standards required that the bacterial count should be less than 500 microorganisms per ml and that no pathogenic bacteria should be present. Satisfactory results were obtained in most laboratories (Table 11.7); of 138 batches on which reports were supplied, in only 12 was the maximum acceptable count exceeded. These came from 8 different laboratories, distributed over 4 continents. Of the 50 lots for which a bacterial plate count of zero (per ml) was recorded, 10 were produced in chick embryos or tissue culture, in 3 countries.
ESTABLISHMENT OF THE WHO REFERENCE CENTRES FOR SMALLPOX VACCINE
The results of the survey confirmed early misgivings about the quality of the vaccine being supplied for use in endemic countries. It was clear that WHO needed a mechanism for the periodic testing of vaccine supplied to the eradication programme, whether by donation to the Organization, through bilateral assistance agreements or by local production in the endemic countries.
WHO does not have laboratories attached directly to it, either at Headquarters or in the regional offices. For purposes of quality control of the vaccine, therefore, the Organization had to make contractual service agreements with appropriate laboratories whereby they would undertake to conduct specified tests.
In December 1966 the Connaught Medical Research Laboratories, University of Toronto, Canada, had accepted a contract from the WHO Regional Office for the Americas for the provision of technical services to improve the quality of smallpox vaccine being produced by laboratories in that region (see Chapter 12). The services involved included an evaluation of existing smallpox vaccine production facilities and their personnel in the region, the provision of training for production personnel, and advice on the selection of equipment necessary for the production and testing of smallpox vaccines being produced in the region. Dr Robert Wilson and Dr Paul Fenje, experts from the Connaught Laboratories, visited production laboratories in the region, especially in South America, where 11 laboratories were producing smallpox vaccine, many of them providing material that did not meet WHO standards, as judged by the survey conducted in 1967.
In 1967, similar arrangements were made by WHO with the National Institute of Public Health, Bilthoven, Netherlands, of which the Director-General was Dr Jan Spaander. The head of the vaccine laboratory was Dr Anton Hekker.
In 1969 the Connaught Laboratories were formally designated as the WHO Regional Reference Centre for Smallpox Vaccine in the Region of the Americas, and the National Institute of Public Health in Bilthoven as the WHO International Reference Centre for Smallpox Vaccine. The services provided by the International Reference Centre were as follows:
- To test smallpox vaccines submitted to WHO from different production laboratories.
- On the basis of the results of tests and special studies, to advise appropriately on the improvement of vaccine production methods.
- To collect, maintain and study, as indicated, strains of vaccinia virus from different parts of the world.
- To provide seed virus and national reference vaccine when required.
- To conduct research which could contribute to the improvement of vaccine production and testing methodology.
- To train virologists in the production and testing of smallpox vaccine.
Shortly after the 1967 survey, 20 laboratories in 20 different countries submitted samples of their vaccines to the WHO International Reference Centre for Smallpox Vaccine for testing. They included 4 laboratories in Asia, 8 in Europe, 3 in North Africa, 1 in south-western Asia, 3 in sub-Saharan Africa and 1 in the USA. The samples from 6 of these laboratories were intended for use in the eradication programme, through donation to WHO or bilateral assistance; the samples from the other 14 laboratories were from experimental production runs. It is reasonable to assume that most laboratories submitted samples that they expected would meet WHO standards.
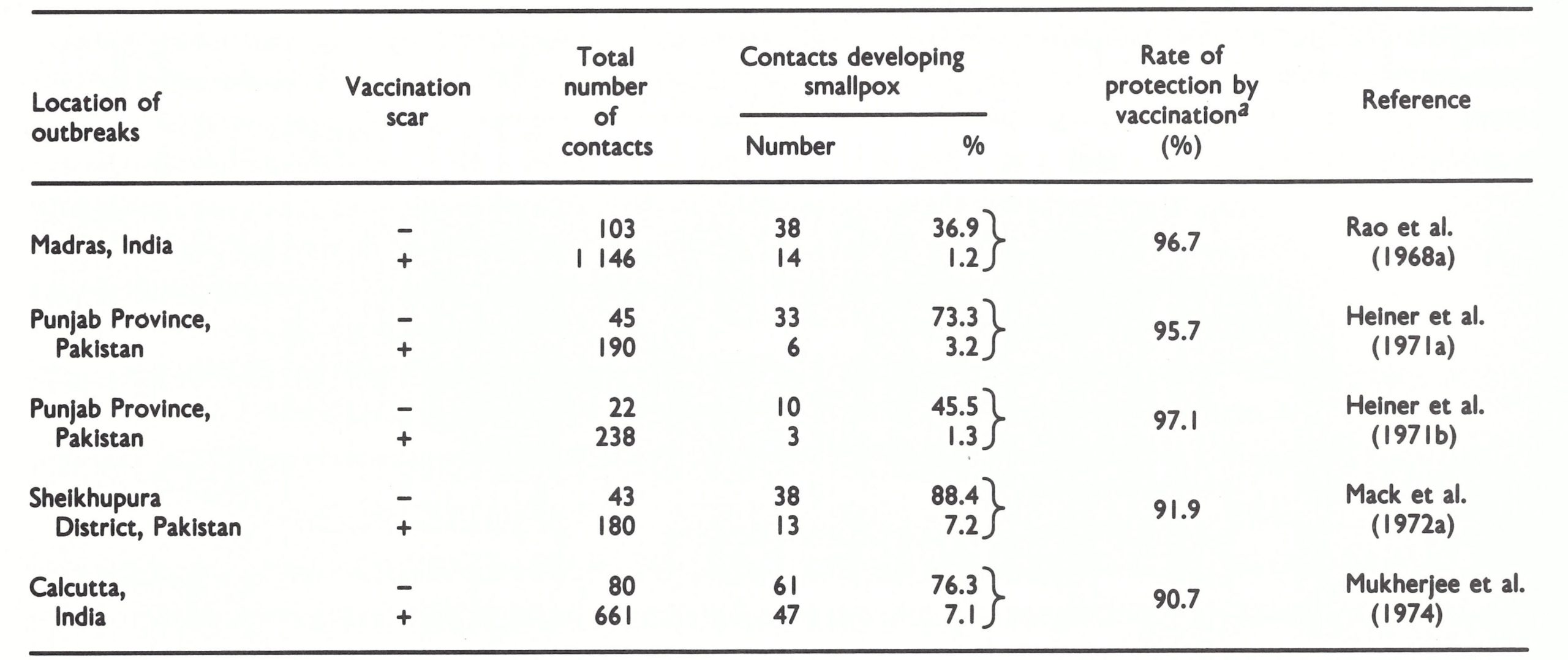
Of the 35 batches actually proposed for use in the eradication programme, 22 failed to meet the initial potency or heat-stability requirements (Table 11.8). Some of these were from potential donors in the industrialized countries. Of the 39 batches submitted by producers intending to develop freeze-dried vaccine for use in their own countries or as a contribution to the global eradication programme, 25 failed to meet the WHO requirements. The conclusion was that in 1967 not more than 10% of the vaccine in use in endemic countries met WHO requirements, while the quality of the experimental batches was equally unsatisfactory.


Plate 11.2. Charles Value Chapin (I856 – 1941) was Superintendent of Health of Providence, Rhode Island (USA), from 1884 to 1932. A nationally renowned figure, he imaginatively translated newly emerging concepts of the epidemiology of infectious diseases and methods for their prevention into public health administrative practice.
Plate 11.3. Jan Spaander (b.1914). Director-General of the National Institute of Public Health in Bilthoven, Netherlands, 1950-1980. He greatly facilitated the operations of the WHO International Reference Centre for Smallpox Vaccine established in the Institute.
DEVELOPMENT OF IMPROVED VACCINES
The results of the survey, together with those of other tests on samples carried out by the WHO reference centres, indicated the urgency of strengthening vaccine production methods and quality control so that potent, heat-stable vaccine could be made available to the global programme.
A number of steps were therefore taken by the Smallpox Eradication unit to improve the quality of the vaccine and to ensure an adequate supply:
- organization of a Travelling Seminar on Vaccine Production in March 1968, which resulted in the production of a WHO document on production methodology;
- arrangement of visits to production laboratories by WHO programme staff and consultants;
- establishment of a reference vaccine;
- production of seed lots of Lister strain vaccine by the WHO International Reference Centre for Smallpox Vaccine;
- development of a rapid heat-stability test for the vaccine; and
- regular checking of vaccine potency and heat stability by the WHO reference centers.
Meeting of Experts (March 1968)
Although the basic principles of vaccine production and testing had already been described in two issues of the WHO Technical Report Series (WHO Study Group on Requirements for Smallpox Vaccine, 1959; WHO Expert Group on Requirements for Biological Substances, 1966), the steps in the production of smallpox vaccine had mostly been empirically developed. Little specific information about production methods had been published in the scientific literature. Furthermore, the survey had shown that unsatisfactory batches of vaccine were being produced in laboratories in developed and developing countries alike.
Report Series (WHO Study Group on Requirements for Smallpox Vaccine, 1959; WHO Expert Group on Requirements for Biological Substances, 1966), the steps in the production of smallpox vaccine had mostly been empirically developed. Little specific information about production methods had been published in the scientific literature. Furthermore, the survey had shown that unsatisfactory batches of vaccine were being produced in laboratories in developed and developing countries alike.
Table 11.8: Independent testing In 1967 by the WHO International Reference Centre for Smallpox Vaccine In Bilthoven of production batches Intended for use In the eradication programme or in experimental productiona
a In this and later tables, the designation of resulta as "satisfactory" or "unsatisfactory" was based on the following criteria for a satisfactory product (titres expressed In pock-forming units ml):
Initial potency: ≥108.0
Heat stability, titre after 4 weeks at 37°c: ≥108.0
Bacterial coun (colonies per ml): < 500
b Failed to meet WHO requirement In terms of Initial potency or heat stability.
Development by WHO of Quality Control of Smallpox vaccine
In 1958 a resolution of the Eleventh World Health Assembly had stressed the importance of "thermostable smallpox vaccine" for use in tropical areas, and in 1962 the Fourteenth World Health Assembly invited countries to contribute freeze-dried vaccine to the WHO eradication programme . Thus WHO had to test donated vaccine before accepting it and began to do so in 1962 . However, the arrangements did not cover other, much larger, amounts of vaccine being donated through bilateral assistance programmes or vaccine being produced in endemic countries for national programmes . In fact, the WHO Secretariat held the view that bilateral contributions and the way that they were used were the concern solely of the two countries involved, and that WHO should not intervene and had no authority to impose international quality control on domestic vaccine producers. These problems were addressed after the 1967 survey revealed that so many producers, in so many countries, were producing substandard vaccine. Using these data, the Smallpox Eradication unit initiated a continuing exchange of information with individual producers, who came to understand the importance of international quality control.
Another consequence of the WHO survey of vaccine quality was that the results led governments of many countries to recognize the deficiencies of the system by which producers themselves evaluated the quality of their products, and thus helped in the establishment in several countries of independent systems for the quality control of all biological products.
Panel of experts
Selected experts from the following laboratories were chosen to participate in a seminar held in Geneva in March 1968 and in activities related to the preparation of the manual on vaccine production:
- Connaught Medical Research Laboratories, University of Toronto, Toronto, Canada
- Moscow Research Institute for Viral Preparations, Moscow, USSR
- National Institute of Public Health, Bilthoven, Netherlands
- Research Institute of Immunology, Prague, Czechoslovakia
- Wyeth Laboratories, Philadelphia, USA
At that time the first and the third laboratories were directly assisting the WHO eradication programme in testing vaccine samples; the fourth laboratory—in Prague—had published an important developmental study on smallpox vaccine; and the laboratories in the USSR and the USA were major producers of freeze-dried smallpox vaccine for the global eradication programme.
In the 1960s the Lister Institute, in the United Kingdom, was the leading laboratory in the technology of freeze-dried smallpox vaccine (see Chapter 7). Because funds for its research were derived partly from the proceeds of the sale of its vaccine, the Institute turned down WHO’s request to permit the participants in the seminar to visit the Institute, believing that the full disclosure of its technical knowledge might reveal trade secrets. However, the Director of the Lister Institute agreed to the appointment of Dr Colin Kaplan, then Director of its Vaccine Production Department, as a special consultant to the group. Early in 1968 exploratory discussions were held in Bilthoven between Dr Kaplan, Dr A.C. Hekker (Bilthoven), and Henderson and Arita (WHO), and a working paper on production methods, based mainly on those used in the Lister Institute, was prepared as a basis for discussions on the proposed manual.
First meeting (19-23 March 1968)
The first meeting of the group of experts, held at WHO Headquarters in Geneva, reviewed various aspects of production and testing. Working papers submitted by the participants provided information from each laboratory on the following topics: nature of seed lot used for production, virus titre at each production stage from seed lot to final product, virus yield, heat-stability studies of final product, and level of bacterial contamination at various stages of production. Photographs showing the various production processes were examined, with a view to their inclusion in the proposed manual.
Visits to laboratories and final meeting (28-30 March 1968)
Immediately after the first meeting, the group of experts visited the Moscow Research Institute for Viral Preparations and Wyeth Laboratories, Philadelphia, two of the major vaccine contributors, to observe the production process and examine the production data. The final meeting was held at Wyeth Laboratories. The draft of the manual on the methodology of vaccine production was carefully reviewed in the light of the visits. Agreement was reached on a document entitled Methodology of Freeze-dried Smallpox Vaccine Production, the first report to describe simple and practical methods for the production and testing of freeze-dried vaccine. Although never officially published, it contained all the necessary information at a level of detail not provided elsewhere and was made widely available as a document of the Smallpox Eradication unit (SE/68.3 Rev. 2), being distributed to all laboratories interested in the production of freeze-dried smallpox vaccine. Its contents are summarized below .

Plate 11.4. Members of the Seminar on Vaccine Production, at the final meeting from 28 to 30 March 1968, at the Wyeth Laboratories, Philadelphia, Pennsylvania, USA. Left to right, front row: P. Fenje (Canada), S.S . Marennikova (USSR), A.C. Hekker (Netherlands), J.H . Brown (USA), R.J. Wilson (Canada) and I. Arita (WHO); back row: F.M . McCarthy (USA), M.Z. Bierly (USA), H. Tint (USA), V.N. Milushin (USSR), A. Bernstein (USA), C. Kaplan (United Kingdom), D.A. Henderson (WHO), B A. Rubin (USA) and A.K. Fontes (USA).
Production of Freeze-dried Vaccine
Choice of vaccinifer
Although in 1968 the successful production of smallpox vaccine in chick embryos and in cultured cells had been reported, it was decided that the document should concern itself only with the traditional method, in which animal skin was used for the production of vaccine pulp, since this was far simpler and more straightforward. It was thought better to upgrade the method familiar to producers in developing countries than to suggest techniques that were comparatively new and that failed at that time to give a product of satisfactory heat stability.
Strain of vaccinia virus
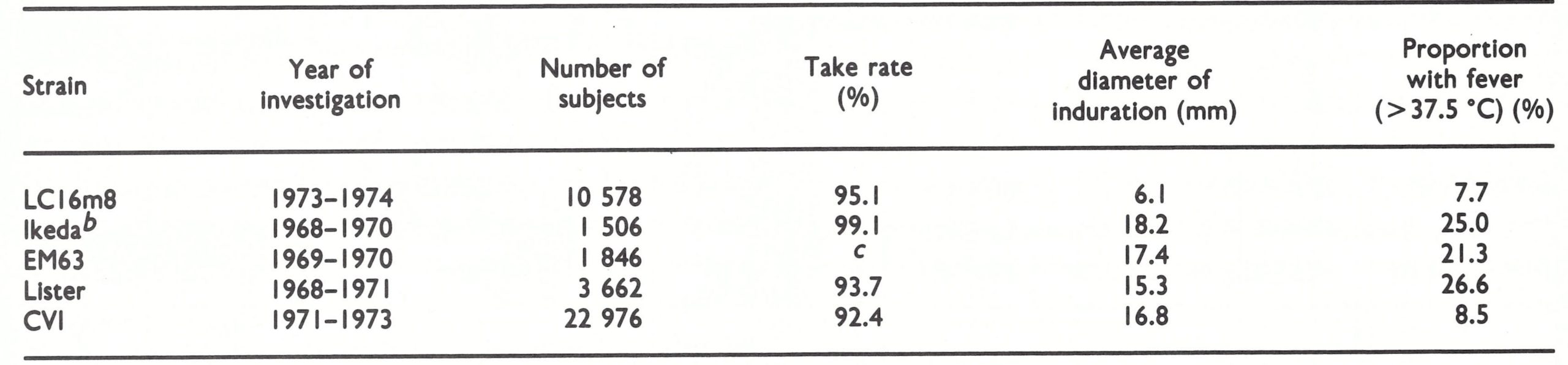
It was recommended that a strain of vaccinia virus should be used which would induce adequate immunity in man with as few ill effects as possible, that it should produce compact, clearly visible white pocks on the chorioallantoic membrane, thus making assay easier, and that unsuitable strains should be discarded. No particular strain was officially recommended but, in response to inquiries, the Smallpox Eradication unit advised that either the Lister or the New York City Board of Health strain should be used. The Lister strain was more widely used, because it produced pocks on the CA membrane that were easier to count and because the WHO International Reference Centre later produced seed lots of this strain for distribution to vaccine producers in developing countries (see below).
Seed lots
The principle of using seed lots (WHO Study Group on Requirements for Smallpox Vaccine, 1959; see Chapter 7) was recommended and a practical method for preparing them was described. Slonim et al. (1969) showed that the viral concentration in the harvested pulp was 3 times higher when vaccinifers were scarified with an inoculum containing 108.3 pock-forming units per ml than with one containing 107 pock-forming units per ml. To provide a safety margin, the document stipulated that the titre of the seed lot should not be less than 108.7 pock-forming units per ml. This was substantially higher than the acceptable titre for vaccination, but was essential if adequate yields of high-titer vaccine were to be obtained.
Preparation of vaccinifer
A rigorous schedule for cleansing the animal skin was recommended, which substantially reduced the bacterial count to the extent that very few or no viable bacteria (and no pathogens) were found in the aliquot of vaccine (usually 1 ml) cultured.
Scarication
The recommended method of scarification was based on the experience of the participating producers, and an instrument for scarification of the vaccinifer in use in Wyeth Laboratories was produced by WHO and distributed to producers on request (Plate 11.5).
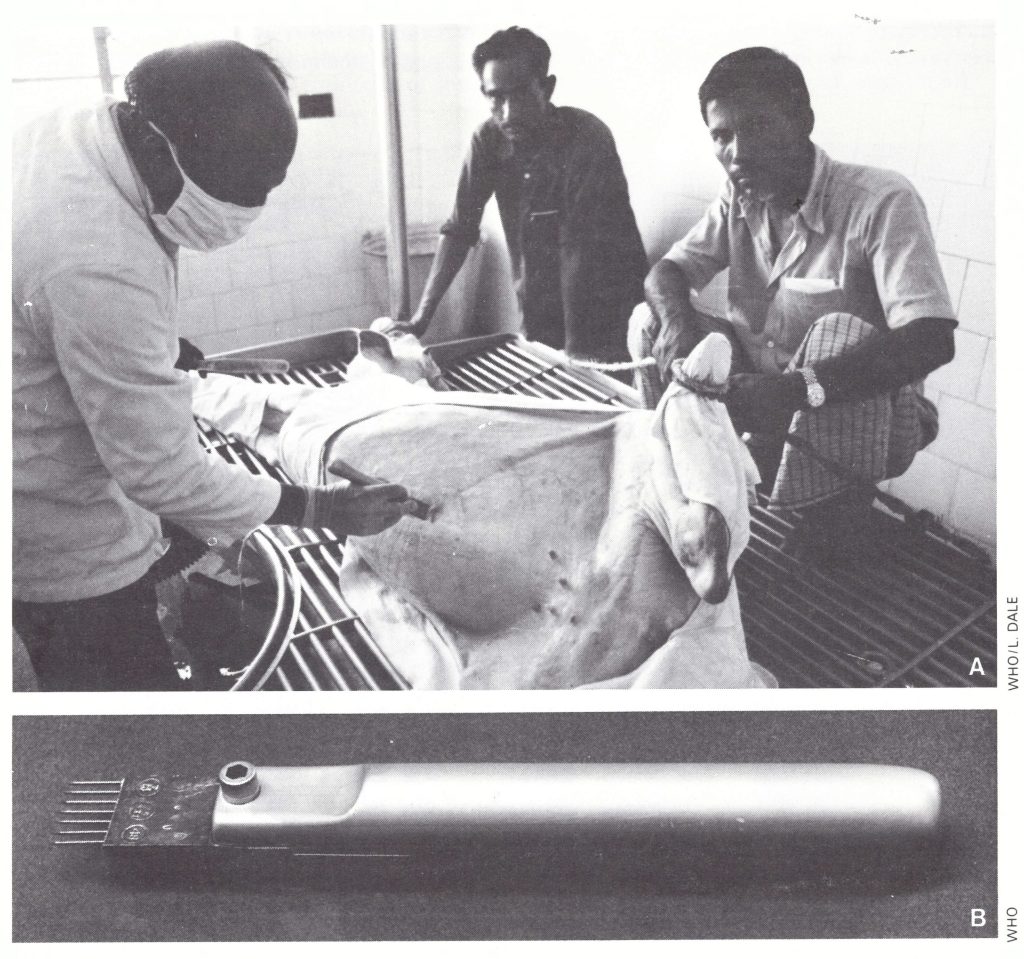
Plate 11 .5. A: Vaccine production in calf skin in Bangladesh, using the scarifier developed by Wyeth Laboratories,
Philadelphia, Pennsylvania, USA. B: Enlarged view of scarifier.

Plate 11 .6. Dimitrij Slonim (b. 1925), of the Institute of Sera and Vaccines, Prague, Czechoslovakia, contributed to methods for the accurate assay of vaccine and was a member of the Seminar on Vaccine Production.
Preparation of vaccine
The method of extraction and treatment of the viral suspension was described with special reference to the specification of the required potency at each stage of the production. Phenol was added to a concentration of 0.5% by weight, following observations by Hekker & van Ramshorst (1969), who had investigated the phenol content and bacterial counts of 51 lots of freeze-dried vaccine from 23 laboratories in several countries and shown that this was the maximum concentration that would reduce the bacterial count without affecting initial potency or heat stability.
Before being dispensed into ampoules, the viral suspension was required to have a titre of at least 108.7 pock-forming units per ml, since the processes of freeze-drying and incubation at 37°C for 4 weeks would somewhat reduce its potency. The final vaccine, after incubation, was required to have a titre of not less than 108 pock-forming units per ml, in line with the standards established by WHO in 1965.
Size of container used for vaccine distribution
The volume of fluid to be dispensed into each final container was specified as between 0.15 ml and 0.25 ml, which would provide 15-25 doses per ampoule by the conventional scarification or multiple pressure techniques and 60-100 doses for vaccination with the bifurcated needle. Although ampoules providing fewer doses were requested by field workers, production experts agreed that 0.15-ml lots were the smallest practicable volumes that could be dispensed and dried.
Reconstituting fluid
Traditionally, glycerol had been used at a concentration of 40-60% in the suspending fluid of liquid vaccines. It had to be omitted from the fluid used to suspend the lymph for freeze-drying, but its properties made it useful for the reconstituted vaccine. Studies by Slonim & Roslerova (1969) showed that, at temperatures above 0°C, glycerol inactivated vaccinia virus at a rate that was proportional to temperature and glycerol concentration. It was suggested in the document that the reconstituting fluid should consist of a solution of 50% (v/v) glycerol in 0.004 M Mcllvaine’s buffer.
Freeze-drying equipment
The document provided advice on the advantages and disadvantages of the different types of freeze-drier available commercially (centrifugal and shelf ; Plate 11.7), and the ampoules, vials and rubber stoppers appropriate for them.

Plate 11.7. Freeze-driers of the type used for the manufacture of smallpox vaccine. A: Centrifugal freeze drier and secondary drier, with manifolds. B: Centrifugal carrier plate assembly. C: Sealing headers for ampoules on secondary drier. D: Shelf-type freeze-drier.
CONTINUING QUALITY CONTROL OF SMALLPOX VACCINE
Having established methods for the production of smallpox vaccine suitable for use in tropical countries, it was important that both WHO and national authorities should continue to test the vaccine provided, whether donated through WHO or through bilateral assistance or produced locally in the endemic countries.
Testing Samples of Vaccine
Between 1959 and 1967 the testing of samples of vaccine donated to WHO was a lengthy process, sometimes more than a year elapsing between the submission of a sample and the sending of a report on its potency to the producer. Two factors contributed to this delay. First, the testing arrangements were entrusted as additional work to the small Biological Standardization unit of WHO, whose normal duties involved the staff in frequent absences from Geneva; samples for testing and reports on potency had to await their return from duty travel. Secondly, the testing was carried out for WHO by the State Serum Institute in Copenhagen, which produced smallpox vaccine on a relatively small scale and for only a brief period each year; to rationalize its work, it usually tested samples from WHO at the same time as its own local production batches. This delay, which was unacceptable if the assays of vaccine quality were to be of any use, was eliminated when the responsibility for testing was transferred to the WHO reference centres for smallpox vaccine and the handling of samples and reports in Geneva was taken over by the Smallpox Eradication unit.
After 1968, all producers who donated vaccines to the eradication program or who produced vaccines for national eradication programmes were requested to submit vaccine samples periodically for testing by WHO. In addition, laboratories developing the capacity to produce freeze-dried vaccine were encouraged to submit samples to WHO for testing, so that they could be advised, if necessary, on how to improve the quality of their vaccine. Between 1967 and 1984, 27 countries donated freeze-dried vaccine to WHO (see Table 11.15). Samples of all these donations were sent to WHO reference centres for testing. At later stages of the programme, when samples from producers consistently met WHO requirements, donations were accepted without advance testing, although samples were tested after the donation had been received, to confirm that it was of the desired quality.
For the quality control of vaccine produced locally in endemic countries, WHO smallpox eradication staff working for national programmes were actively involved in collecting and dispatching vaccine samples and, if the results of the assays carried out by the national laboratory and the WHO International Reference Centre were in agreement, the batches of vaccine from which the samples had been taken were dispatched for use in the field.
Plate 11 .8. Nelja N . Maltseva (b . 1934), a member of the WHO collaborating centre in the Moscow Research Institute for Viral Preparations, USSR, was active in laboratory diagnosis and research and worked as a consultant on vaccine production in several countries.
Visits by Consultants
Vaccine producers having problems in ensuring that vaccine quality met WHO requirements, starting new methods of production or modifying traditional production methods were encouraged to benefit from the advice of visiting WHO short-term consultants. Although many laboratories were engaged in vaccine production, only a few of the personnel concerned were sufficiently experienced to be able to suggest realistic improvements in production methods, since instruments and working procedures had to be adapted to the practical realities in developing countries. The 15 consultants and the countries they assisted are listed in Table 11.9. Over the period 1967-1979, they visited more than 20 laboratories.
Freeze-driers manufactured by Edwards High Vacuum, Crawley, Sussex, England, were widely used and, at the request of the Smallpox Eradication unit, technicians from this company visited producers to maintain and service equipment.
Reference Vaccine
The International Reference Preparation of Smallpox Vaccine was established in 1962 (Krag & Bentzon, 1963) and is held in the custody of the International Laboratory for Biological Standards, State Serum Institute, Copenhagen, Denmark. One ampoule contains 14 mg of freeze-dried vaccine. This International Reference Preparation was designed to be used for standardizing producers’ own reference vaccines, which could then be used whenever testing was carried out. However, many producers, especially in developing countries, were unable to produce satisfactory working reference vaccines. The WHO International Reference Centre for Smallpox Vaccine therefore produced a special batch of freeze-dried vaccine, No. 6713-18, which had been titrated in parallel with the International Reference Preparation and had a titre of 108 pock-forming units per ml when reconstituted. A number of these ampoules were kept in Geneva at—15 °C and the rest at the WHO International Reference Centre; they were provided as required to vaccine producers in developing countries for use as reference vaccines.
Seed Lots of Vaccine
It was recommended in the document Methodology of Freeze-dried Smallpox Vaccine Production that the seed lot system should be used (see box)—i.e ., a reasonably large freeze-dried or frozen primary seed lot was to be maintained, from which secondary seed lots to be used in production runs were to be derived. These secondary seed lots were to be no more than 5 passages removed from the primary seed lot. However, in many laboratories the history of primary seed lots was unknown, and many were of low potency (less than 108.7 pock-forming units per ml) or were heavily contaminated so that it was extremely difficult for certain producers to use the system.
In 1968, responding to a request from the Smallpox Eradication unit, the WHO International Reference Centre for Smallpox Vaccine overcame these difficulties by the production of a large secondary seed lot (Li2 K2 G) from the Lister strain of vaccinia virus. This consisted of a large number of ampoules of freeze-dried virus, each of which after reconstitution contained 10 ml of virus with a titre of about 109 pock-forming units per ml . The Lister strain of virus used in this seed lot had been received from the Lister Institute in 1961 as a sheep lymph preparation, and had been passed twice on calf skin . Second-passage material was lyophilized in 10-ml amounts as seed virus for the production of smallpox vaccine . Thus, the vaccine in the seed lots prepared for international distribution was the second passage on calf skin, and had a high viral content. These seed lots were distributed on request to producers and often used immediately for the inoculation of vaccinifers so that the production process was accelerated. The availability of this material was partly responsible for the widespread use of the Lister strain of vaccinia virus from 1969 onwards (see Table 11.21).
These two products, the working reference vaccine and the secondary seed lot ampoules, greatly simplified procedures and assisted producers who were encountering difficulties. When supplied, they were always accompanied by special instructions on the potency testing of smallpox vaccine, as outlined below in the section on assay technique.
The Seed Lot System for Vaccine Production
"The seed virus system is one of the procedures necessary to ensure that each production lot of vaccine has the same desirable biological characteristics as the parent strain . . . [It] requires that primary and several secondary seed lots be produced and dispensed in sufficient numbers of containers to ensure an adequate supply of virus for inoculation for long periods of time. The secondary seed virus used for production is not to exceed the fifth serial passage of the primary seed virus ; thus, each production lot of vaccine will not be more than six passages removed from the primary seed virus.
"The size of a seed virus lot is dependent upon the requirements of the production laboratory. Units requiring large volumes of seed virus during a short interval of time may find it necessary to prepare several passages of secondary seed lots in order to obtain the required volume of inoculum for production lots of pulp. Smaller production units may be able to utilize the second passage of the primary seed as the inoculum in the production of vaccine pulp.
"The primary and secondary seed virus lots should pass the standard tests for identity, safety and bacterial content . . . The potency of the primary and secondary seed virus should be as high as is practicable and assayed periodically (every three months) to ensure adequate potency following long-term storage . Seed virus with a potency less than 5 x 108p.f.u. per ml should not be used.
"Ideally, the primary and secondary seed lots should be freeze-dried in ampoules and stored at 4°C or lower . However, an adequate supply of potent seed virus can also be maintained by the use of freeze-dried primary seed virus and the preparation of secondary seed virus as glycerolated suspensions (50% glycerol in 0.004 M Mcllvaine's buffer) which will retain adequate potency for one year when stored at—20°C." (From Methodology of Freeze-dried Smallpox Vaccine Production Methodology; SE/68.3 Rev.2.)
Evaluation of Testing Methods
Rapid heat-stability test
During the first 3 years of the Intensified Smallpox Eradication Programme, substantial efforts were made to ensure the flow of adequate supplies of vaccine to national smallpox eradication programmes. All vaccine, however, was tested in order to ensure that its quality was satisfactory. The bottleneck in the standard testing procedures was the heat-stability test, which took not less than a month, because of the need to assay the vaccine after 28 days at 37°C. Cross et al. (1957), however, had demonstrated the feasibility of determining stability by assaying potency before and after heating at 100°C for 1 hour.
In 1969, Arita, in collaboration with the WHO International Reference Centre for Smallpox Vaccine, compared the results of heat-stability testing of 139 batches of vaccine by the conventional 4-week test at 37°C and the 1-hour boiling test (Fig. 11 .1). Preparations with an initial titre of over 10 8.5 pockforming units per ml and a titre of more than 107.5 pock-forming units per ml after boiling always met the standard requirements for heat stability (Arita, 1973). Accordingly, after 1969, the testing procedures at the WHO International Reference Centre were modified. Vaccine samples were first tested for stability by incubation at 100°C for 1 hour; if the vaccine met the requirements noted, it was regarded as acceptable. Vaccine which failed to pass this screening test was further tested by the conventional heat-stability test.
This approach greatly speeded the testing procedures. Between 1969 and 1972, of 337 batches tested by this method by the WHO International Reference Centre, 224 (67%) were found to be acceptable. This method was also used in India (Sehgal et al .,1969; Sehgal & Singha, 1972). For reasons unknown, vaccine produced in Bangladesh in vials rather than ampoules and Iranian vaccine prepared in ampoules consistently failed the boiling test, although these vaccines were usually acceptable by the standard heatstability test. They were therefore selectively tested only by the standard heat-stability method.
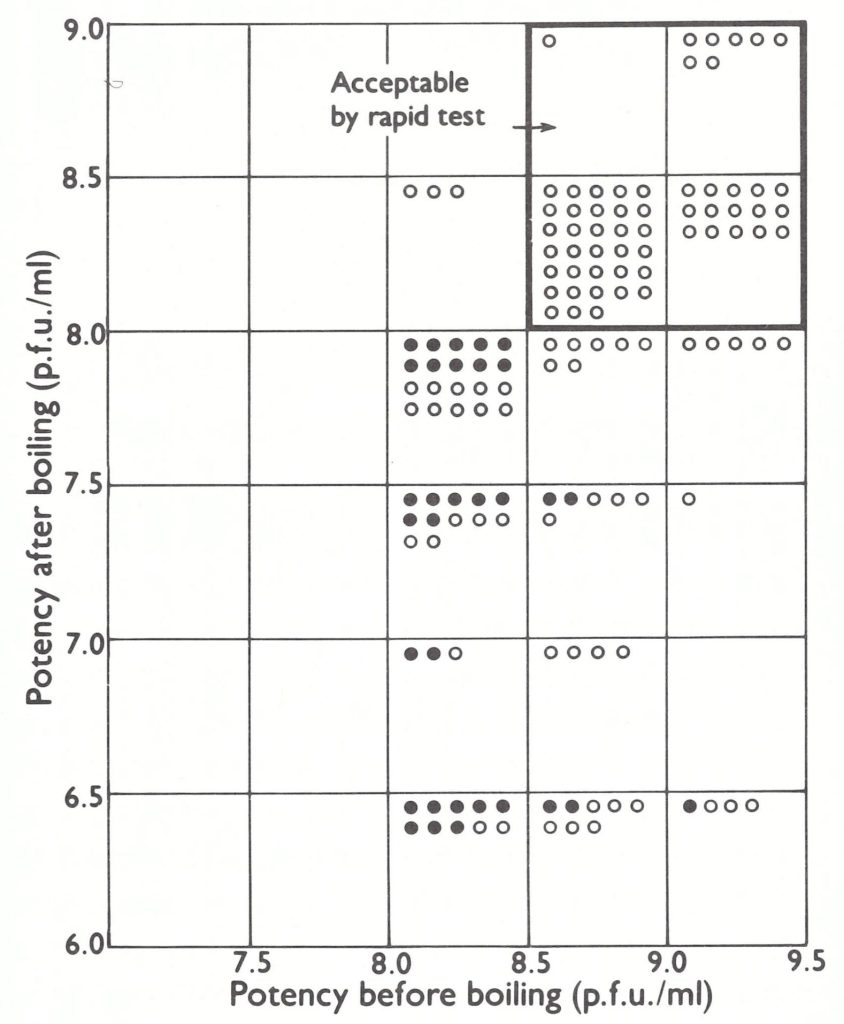
Fig. 11.1. Comparison of the rapid screening test (100°C for 60 minutes) and the conventional heat stability test (37 °C for 4 weeks). Open circles: acceptable by conventional test; closed circles: unacceptable by conventional test. (From Arita, 1973.)
Discrepancies between test results
As the testing services developed, the Smallpox Eradication unit took great pains to determine the causes of discrepancies sometimes encountered between the results of potency tests carried out in the WHO International Reference Centre and in individual production laboratories. These investigations often led to improvements in the manufacturers’ potency testing procedures and also helped to maintain their confidence in the results obtained by the centre. When a discrepancy occurred, the test was repeated in order to determine whether the difference in titres was, in fact, significant. Use of the centre’s reference vaccine, which was distributed to all producers on request, also helped to solve such problems.
Assay technique
Some discrepancies resulted from apparently minor differences in the procedures used for assaying vaccine on the CA membrane. For instance, it was recommended (WHO Expert Group on Requirements for Biological Substances, 1966) that: “At least ten chick embryos, each of about 12 days’ incubation, shall be divided into two equal groups. To the chorioallantoic membrane of each embryo of the first group, 0.1 ml or 0.2 ml of a suitable dilution of the vaccine shall be applied.” In this context, some producers were using 12-day-old embryonated eggs and some 13-day-old eggs, or 11-,12- or 13-day-old eggs as available. Some producers used an inoculum of 0.1 ml, others one of 0.2 ml.
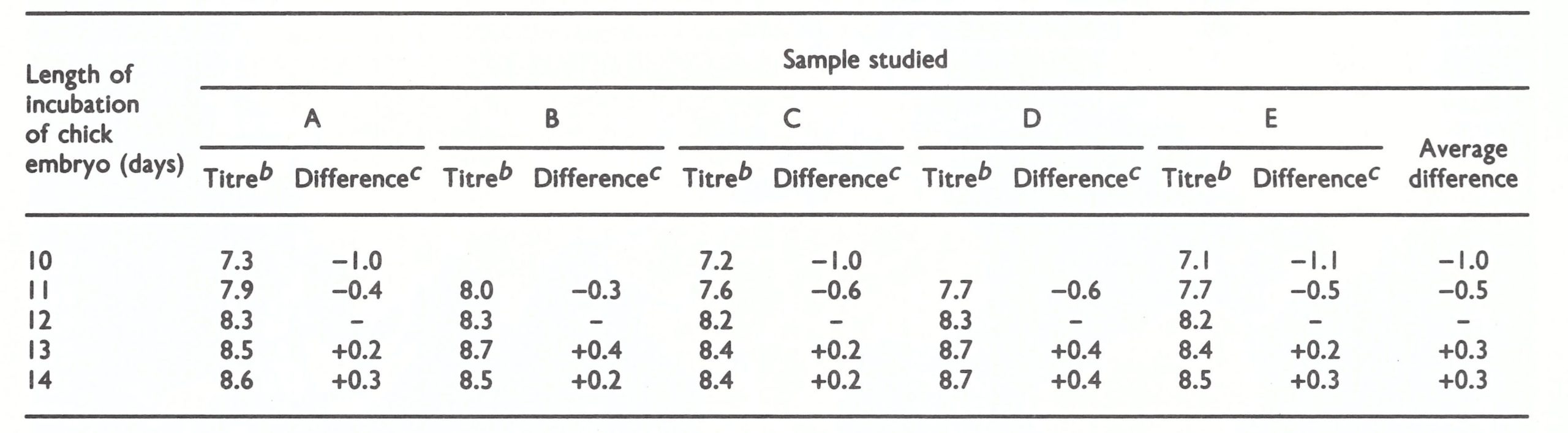
Dr Alan Bernstein (Wyeth Laboratories) studied the titre of smallpox vaccine as a function of the number of days of incubation of the embryonated eggs (10, 11, 12, 13 or 14 days) . The younger the embryo used for the assay, the lower was the apparent titre of the vaccine (Table 11.10). The use of eggs only 1 day younger or 1 day older than 12 days changed the titre by as much as-0.6 to + 0 .4 log unit respectively. Differences were also observed when inocula of 0.1 and 0.2 ml were used (Slonim et al., 1967). Because of these findings, an inoculum of 0.1 ml and an incubation period of 12 days for the eggs were accepted as standard.
It was also suggested that the quality of the eggs from different geographical areas might affect the assays. For reasons unknown, vaccines assayed in developing countries often gave titers 0.2-0.3 log units lower than when tested by the WHO reference centres, a not unwelcome circumstance, as it ensured with greater certainty that the vaccine used in the field was fully potent.
To ensure comparability of results, it has been decided that assay by pock counting on the CA membrane will be used for as long as it is necessary to test samples from the vaccine reserves held in Geneva and Lausanne (see Chapter 28).
Discrepancies between test results
As the testing services developed, the Smallpox Eradication unit took great pains to determine the causes of discrepancies sometimes encountered between the results of potency tests carried out in the WHO International Reference Centre and in individual production laboratories. These investigations often led to improvements in the manufacturers’ potency testing procedures and also helped to maintain their confidence in the results obtained by the centre. When a discrepancy occurred, the test was repeated in order to determine whether the difference in titres was, in fact, significant. Use of the centre’s reference vaccine, which was distributed to all producers on request, also helped to solve such problems.
Assay technique
Some discrepancies resulted from apparently minor differences in the procedures used for assaying vaccine on the CA membrane. For instance, it was recommended (WHO Expert Group on Requirements for Biological Substances, 1966) that: “At least ten chick embryos, each of about 12 days’ incubation, shall be divided into two equal groups. To the chorioallantoic membrane of each embryo of the first group, 0.1 ml or 0.2 ml of a suitable dilution of the vaccine shall be applied.” In this context, some producers were using 12-day-old embryonated eggs and some 13-day-old eggs, or 11-,12- or 13-day-old eggs as available. Some producers used an inoculum of 0.1 ml, others one of 0.2 ml.
Dr Alan Bernstein (Wyeth Laboratories) studied the titre of smallpox vaccine as a function of the number of days of incubation of the embryonated eggs (10, 11, 12, 13 or 14 days) . The younger the embryo used for the assay, the lower was the apparent titre of the vaccine (Table 11.10). The use of eggs only 1 day younger or 1 day older than 12 days changed the titre by as much as-0.6 to + 0 .4 log unit respectively. Differences were also observed when inocula of 0.1 and 0.2 ml were used (Slonim et al., 1967). Because of these findings, an inoculum of 0.1 ml and an incubation period of 12 days for the eggs were accepted as standard.
It was also suggested that the quality of the eggs from different geographical areas might affect the assays. For reasons unknown, vaccines assayed in developing countries often gave titers 0.2-0.3 log units lower than when tested by the WHO reference centres, a not unwelcome circumstance, as it ensured with greater certainty that the vaccine used in the field was fully potent.
To ensure comparability of results, it has been decided that assay by pock counting on the CA membrane will be used for as long as it is necessary to test samples from the vaccine reserves held in Geneva and Lausanne (see Chapter 28).
Moisture content
Although residual moisture appeared to be an important factor influencing the stability of freeze-dried vaccine, it was difficult to establish criteria for the moisture content. Sparkes & Fenje (1972) studied the decline with time in the potency of freeze-dried vaccine with moisture contents ranging from 0.36% to 6.7%, at temperatures of 4°C, 24°C, 37°C and 100°C (Fig. 11.2). They concluded that a residual moisture content of less than 1% was essential for the satisfactory storage of freeze-dried smallpox vaccine at ambient temperatures . However, the test for moisture content was too expensive and technically too difficult to use as a routine, and samples with too high a moisture content rarely passed the heat-stability test. The moisture content was sometimes assayed with batches of low heat stability to determine whether this was due to inadequate freeze-drying.
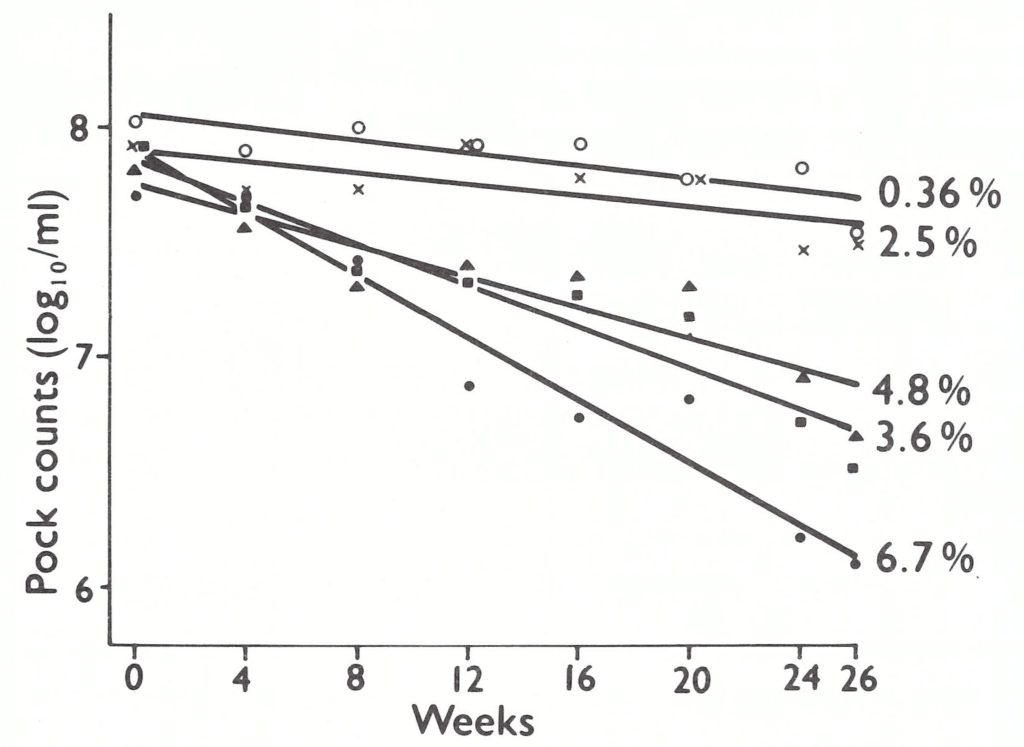
Fig. 11.2. Decline with time in titres of samples of smallpox vaccine of different moisture contents kept at 24°C. The relative effects were comparable at 37 ° C, but the decline in titre at all moisture contents higher than 0.36 % was greater. (From Sparkes & Fenje, 1972.)
Improvements in Vaccine Quality
General
The Intensified Smallpox Eradication Programme marked the first time in the history of WHO that an effective world-wide quality control programme for biological products had been established (Fig. 11.3). Between 1967 and 1980, 2578 production batches were tested, the annual number of samples ranging from 392 in 1973 to 46 in 1980 (Table 11.11) . From 1969 onwards there was a considerable improvement in vaccine quality. Between 1967 and 1972 initial potency was unsatisfactory in 47% of instances, and heat stability in 43% and bacterial count in 10%. From 1973 onwards the initial potency was usually satisfactory but heat stability remained an occasional problem.
Of 1842 batches tested between 1967 and 1976 and classed as satisfactory, an average of 10% were of “borderline” potency after heating-i.e., the potency was between 107.8 and 108.0 pock-forming units per ml. Only one borderline batch was found after that date.
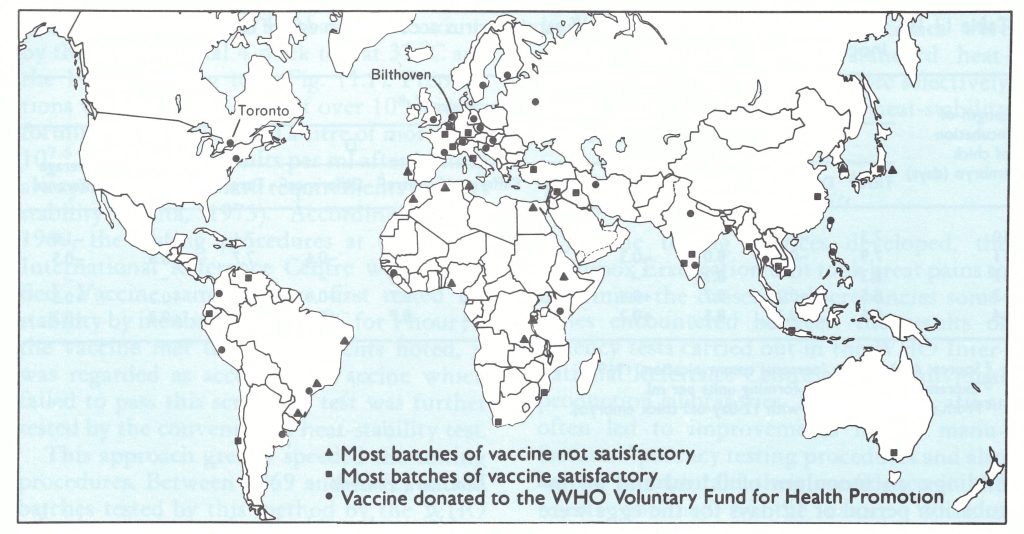
Fig. 11.3. Locations of various categories of producers of freeze-dried smallpox vaccine, 1967-1979; and of the WHO Reference Centre for Smallpox Vaccine for the Americas (Toronto) and the WHO International Reference Centre for Smallpox Vaccine (Bilthoven).
In addition to this regular quality control of vaccine provided by production laboratories, staff of national smallpox eradication programmes sent samples of vaccine from the field for testing if the expiry dates had passed, or if shipments had been delayed or mishandled so that their potency might have been adversely affected (Table 11.12). If the samples tested met the standards of potency and stability, the expiry dates for that batch were extended for another year. After 1970 plies were being more carefully handled in the countries in which they were used.
Results in different regions
By 1971 the quality control operation covered 41 laboratories producing vaccine for national eradication campaigns, for donation to WHO or for bilateral assistance to endemic countries. Although there was a distinct overall improvement, problems continued to occur. Table 11.13 shows the results of quality control tests by WHO reference laboratories of batches produced during the period 1971- 1974.
In South America, Brazil was the only country in which smallpox was still endemic in 1967, but the quality of the vaccine produced in 3 of its 4 production laboratories, most of which was grown in eggs, often failed to meet WHO standards, especially for heat stability (see Chapter 12). Smallpox was nevertheless eradicated from the country, primarily because of the care exercised by staff in the storage and transportation of vaccine.
Producers in North America supplied large amounts of vaccine of good quality to the global programme. However, during the early stages of the programme there were problems of lower potency with some batches produced in the USA for jet injectors. In 1974, some batches of vaccine from Canada were also found to be of unsatisfactory potency . However, these were very occasional failures and the quality was quickly improved when the deficiencies were drawn to the attention of the producers.
Table 11.13: WHO quality control of vaccine in use for the Intensified Smallpox Eradication Programme: results of tests carried out, by WHO region
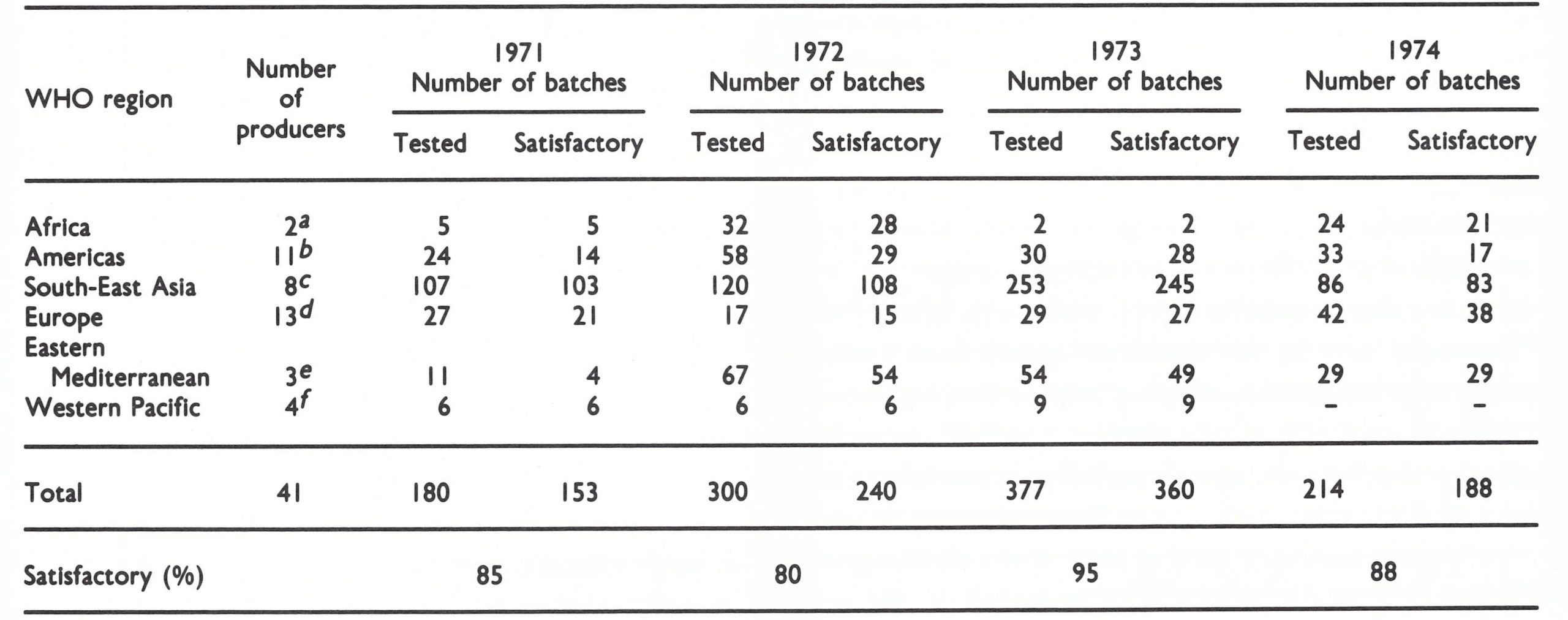
a Guinea, Kenya.
b Argentina, Brazil (4), Canada, Colombia, Ecuador, Peru, Venezuela, US.
c Bangladesh, Burma, India (4), Indonesia, Thailand.
d Belgium, Czechoslovakia, Finland, France, German Democratic Republic, Federal Republic of Germany, Hungary, Netherlands, Sweden, Switzerland, USSR, United Kingdom, Yugoslavia.
e Iran, Iraq, Syrian Arab Republic.
f China (Province of Taiwan), New Zealand, Philippines, Viet Nam.
In Africa, vaccine from Guinea and Kenya was of good quality and was used in their national programmes, some also being donated to other African eradication programmes . Production facilities that had been established in Ethiopia, Rwanda and Zaire soon discontinued production because of unsatisfactory results in tests of vaccine samples and an assessment by consultants that the production problems could not be readily overcome . Efforts were made to improve the quality of vaccine produced in the Nigerian laboratory, but these proved unsuccessful.
In the WHO Eastern Mediterranean Region, excellent vaccine was produced in Iran from 1972 onwards and was used both for the national vaccination programme and in Pakistan, through donation to WHO. Samples of good-quality vaccine were also received from the Syrian Arab Republic, beginning in 1973, but no other laboratories in this region were successful in producing satisfactory freeze-dried vaccine.
In South-East Asia, excellent progress was made in 8 production laboratories in 4 countries, the results of testing after 1971 being generally satisfactory.
Samples from the 4 laboratories in the Western Pacific Region which produced vaccine for their own use or for donation were all satisfactory.
All the European producers enumerated in Table 11.13 produced vaccine for donation to WHO, a sample of each batch being sent to the WHO International Reference Centre for confirmation of their own assay results. A few batches from the USSR tested before 1971 failed to meet WHO requirements, as did a few batches sent from Switzerland in 1972. Since the 2 laboratories in question were major suppliers of vaccine to the global smallpox eradication programme, the failure of these batches to meet the requirements was of concern to both WHO and donor governments. Following an intensive review of procedures, the quality of vaccine supplied by both laboratories improved so that WHO requirements were regularly met.
From 1973 onwards there was enough good-quality vaccine, produced locally or donated to WHO, both to cover adequately the needs of endemic countries and to extend the supply to adjacent countries at risk, as well as countries in which maintenance vaccination was continuing.
Experience with this quality control programme provides some useful lessons. First, even sophisticated laboratories in the industrialized countries produced substandard vaccines— albeit infrequently— indicating that all vaccines must be subject to quality control. Secondly, with adequate technical advice, certain laboratories in endemic countries were successful in producing high-quality vaccine in large quantities. Lastly, quality control contributed to the decision by a number of governments to discontinue production when it became apparent that their vaccine failed to meet WHO standards.

Plate 11 .9. Anton C . Hekker (b. 1928) was head of the WHO International Reference Centre for Smallpox Vaccine at the National Institute of Public Health, Bilthoven, Netherlands, established in 1967. He provided invaluable help in ensuring effective quality control of vaccine used in the global smallpox eradication programme and in helping manufacturers in endemic countries to produce high-quality vaccine.
VACCINE PRODUCTION IN ENDEMIC COUNTRIES
In the interests of self-sufficiency, many endemic countries wished to embark on the production of freeze-dried smallpox vaccine. For all or even most to do so would have been uneconomical in scale of production. Thus, it was necessary to develop a policy based on population size, so as to limit the number of countries to which WHO assistance would be provided.
“If a laboratory is suitable for upgrading to enable it to make freeze-dried smallpox vaccine, it should be equipped to produce at least 500 000 containers a year, each containing 0 .25 ml of vaccine. This is equivalent to about 125 liters [12.5 million standard doses]. Countries not planning to produce this quantity of vaccine annually would be ill-advised to initiate production .” (Methodology of Freeze-dried Smallpox Vaccine Production; SE/68 .3 Rev.2.)
For example, vaccine production in a country with a population of less than 10 million would be uneconomic, since in such a country only 2 or 3 million doses of vaccine would be required annually, an amount that would ordinarily be produced in 10 or 15 production batches, in a few months. Such a production effort would not justify the necessary investment of manpower, equipment and WHO training resources.
However, local production in the larger endemic countries was of the utmost importance, since such large amounts of vaccine were needed. For example, the combined population of Bangladesh, India and Indonesia in 1972 was estimated to be about 762 million—i .e., roughly half the population of all the countries in which smallpox was endemic in the late 1960s. Both WHO and UNICEF provided substantial assistance to these countries, each of which produced large quantities of vaccine and became self-sufficient (Table 11.14). Indeed, in the later stages of the campaign, India became a vaccine donor (see Table 11.15).
During the mid-1960s, a number of laboratories in Africa produced liquid vaccine and so me endeavored to convert to the production of freeze-dried vaccine, but only a few were successful. WHO provided support to 3—Guinea, Kenya and Nigeria—but only the first 2 were successful in consistently producing satisfactory vaccine. In addition, Mozambique and South Africa produced satisfactory freeze-dried vaccine.
Of the WHO-supported efforts, that of Kenya was the most successful . The laboratory in Kenya had been producing liquid vaccine for its own use and for sale to other countries in eastern Africa since the 1930s. The Smallpox Eradication unit, thinking that it might be possible to develop regional centres for vaccine production, took Kenya as a possible model. In practice, an unforeseen economic problem arose in developing the laboratory as a regional resource. To encourage the use of freeze-dried vaccine, it was WHO’s policy to provide it free of charge to endemic countries. However, Kenya needed to recover the set-up and production costs of vaccine it supplied to other countries. Yet these countries could hardly now be asked to buy freeze-dried vaccine from Kenya when other countries received their vaccine free through WHO.
The problem was solved by supplying Kenya with materials needed to produce all of its freeze-dried vaccine, thus offsetting needed expenditures for additional vaccine production for donation to other African countries. Between 1967 and 1977 some 28 million doses were produced in Kenya, of which over half was donated to WHO.
Because of its small population (4.1 million in 1972), Guinea did not meet the criteria for WHO assistance for vaccine production, but the WHO Regional Office for Africa was persuaded by the government of Guinea to arrange for substantial WHO assistance, in terms of visits by consultants, freeze-driers, vaccine containers and reagents. Some 1.8 million doses produced in Guinea were eventually donated to the Intensified Programme in 1974 and 1975, but to all intents and purposes production ceased in 1971, when the WHO technical officer working with the project left the country after completion of his assignment.
Mozambique, while still an overseas province of Portugal, consistently produced satisfactory freeze-dried vaccine, and South Africa began producing a satisfactory freeze-dried vaccine in 1970.
In the South-East Asia Region, WHO consultants frequently visited the 4 Indian producers (in Belgaum, Hyderabad, Guindy (Madras) and Patwadangar) as well as those in Bangladesh, Burma, Indonesia and Thailand (see Table 11.9). UNICEF joined with WHO in supplying equipment, spare parts and other supplies, and by 1971 all were making vaccine that met WHO requirements.
In western Asia, efforts in Pakistan (then West Pakistan) to develop national vaccine production were not successful and the eradication programme there relied on donated vaccine. After 1972, good-quality vaccine was produced in Iran, which donated 26 million doses to WHO between 1973 and 1979. Enough vaccine for national needs was produced in the Syrian Arab Republic by 1973.
VACCINE PRODUCTION IN ENDEMIC COUNTRIES
A total of 465 million doses of vaccine, worth US$8 .5 million, were donated by 27 countries to the Intensified Smallpox Eradication Programme between 1967 and 1984 (Table 11.15). Some 100 million doses remained at the end of the Programme, and are now kept as part of the WHO emergency vaccine reserve for the post-eradication era (see Chapter 28).
The amounts of vaccine distributed annually to 70 countries or organizations between 1967 and 1979 have been given in Chapter 10, Table 10 .9 . Between 15 and 45 million doses of vaccine were dispatched annually to the endemic countries, in addition to the very substantial quantities that were produced locally or donated through bilateral aid programmes.
The problems of effectively distributing the small amounts of vaccine that were donated to WHO between 1958 and 1966 were resolved by centralizing distribution through the Smallpox Eradication unit, which rented cold-storage space in Geneva. Each country that had pledged donations of vaccine was asked to send such donations to the unit as soon as they were available. Charts assessing current and future vaccine requirements for all countries were drawn up. When a request was received from a country, the vaccine required was immediately dispatched by airfreight. During holiday periods, such as Christmas or Easter, special arrangements were made by WHO supply services to ensure that vaccine, if urgently required, could be sent on the earliest possible flight.
Despite these arrangements, the quantity of the vaccine held in stock in Geneva was always small, and often inadequate to meet unexpectedly large demands. It was often possible to send to a country only enough vaccine for 2-3 months’ operations, and often donated vaccine was dispatched as soon as it was delivered to Geneva. The situation was especially serious in 1972, when smallpox spread across Iran, Iraq and the Syrian Arab Republic, the demand for vaccine from these countries alone reaching 17 million doses. The situation became acute when the outbreak spread to Yugoslavia, which did not have enough freeze-dried vaccine to deal with the problem. Emergency appeals for donations were made, and the half million doses of vaccine in WHO cold storage were all dispatched to Yugoslavia. Only from 1974 onwards was a sufficient quantity of vaccine able to be held in Geneva.
Most vaccine was dispatched through Geneva, but in some areas it proved practical and more economical to send the vaccine direct from the producer to the recipient country. Thus donations from Kenya were sent direct to other African countries, from Iran direct to Pakistan, and from India direct to Bangladesh, Nepal and Sri Lanka. Several countries in South America assisted each other, as the need arose.
Table 11.15: Smallpox vaccine contributed to the WHO Voluntary Fund for Health Promotion, Special Account for Smallpox Eradication,a 1967-1984

a Donations made in kind.
b Liquid vaccine not used for the eradication programme but shipped to a country in the temperate zone.
c 100 million doses of multipuncture vaccine donated by Belgium, the German Democratic Republic, India, Iran, the Netherlands, and the USSR, which were not required for the eradication programme, are kept in the WHO vaccine reserve in Geneva and Lausanne. Unused vaccine for let infection has not been retained.
NEW VACCINATION TECHNIQUES
The traditional techniques of vaccination as practiced in the early 1960s have been described in Chapter 7. Two new vaccination techniques were introduced in the Intensified Programme: intradermal inoculation by the jet injector in 1967 and multiple puncture inoculation with the bifurcated needle in 1968.

Plate 11 .10. Production of freeze-dried vaccine, as illustrated in postage stamps issued by Guinea in 1973 to celebrate the 25th anniversary of WHO.
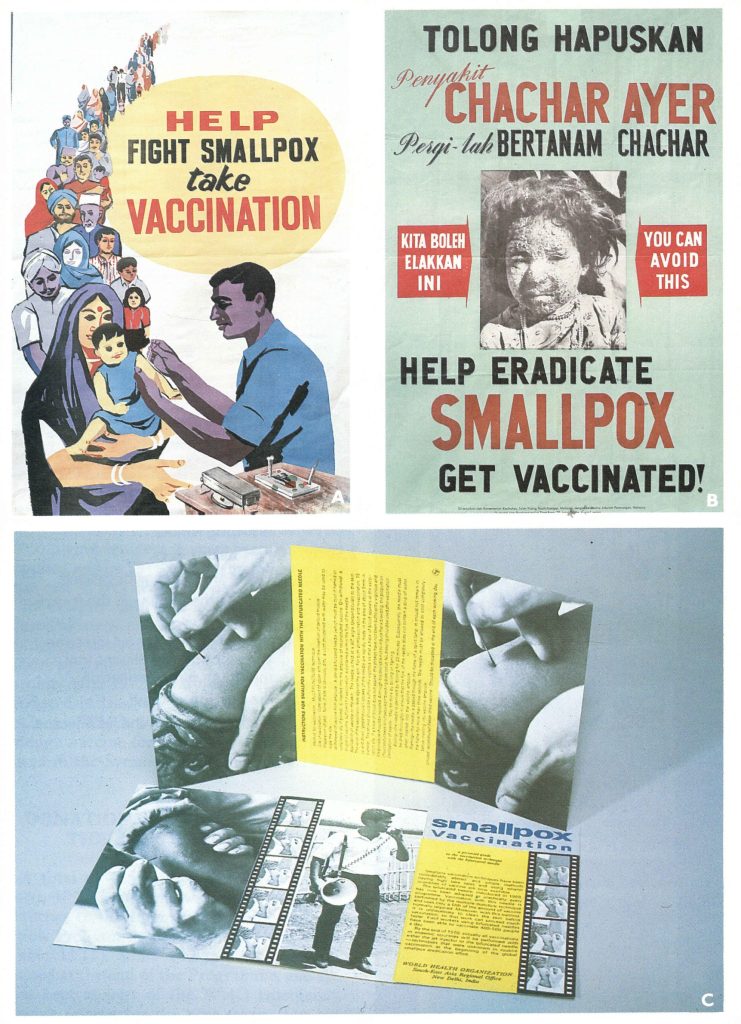
Plate 11.11. Posters promoting vaccination during the Intensified Smallpox Eradication Programme. A: India. B: Indonesia. C: WHO brochure illustrating the technique of vaccination with the bifurcated needle, which was widely distributed.
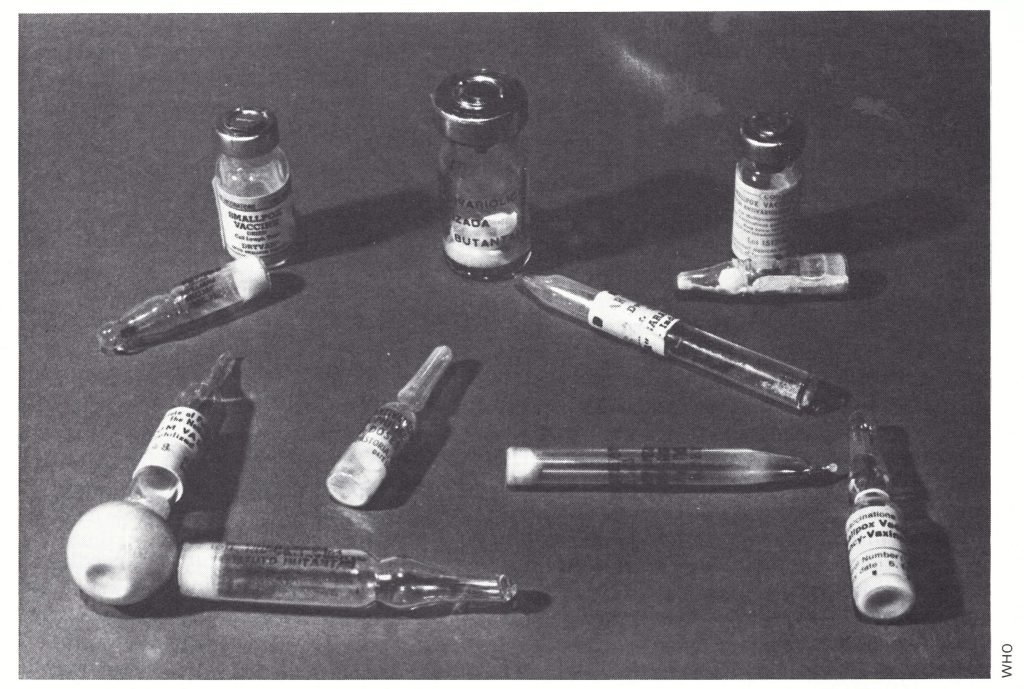
Plate 11.12. Vials and ampoules of freeze-dried smallpox vaccine made in Brazil, Canada, Guinea, India, Netherlands, Thailand, USA, and USSR, and used in the Intensified Smallpox Eradication Programme.
The Bifurcated Needle
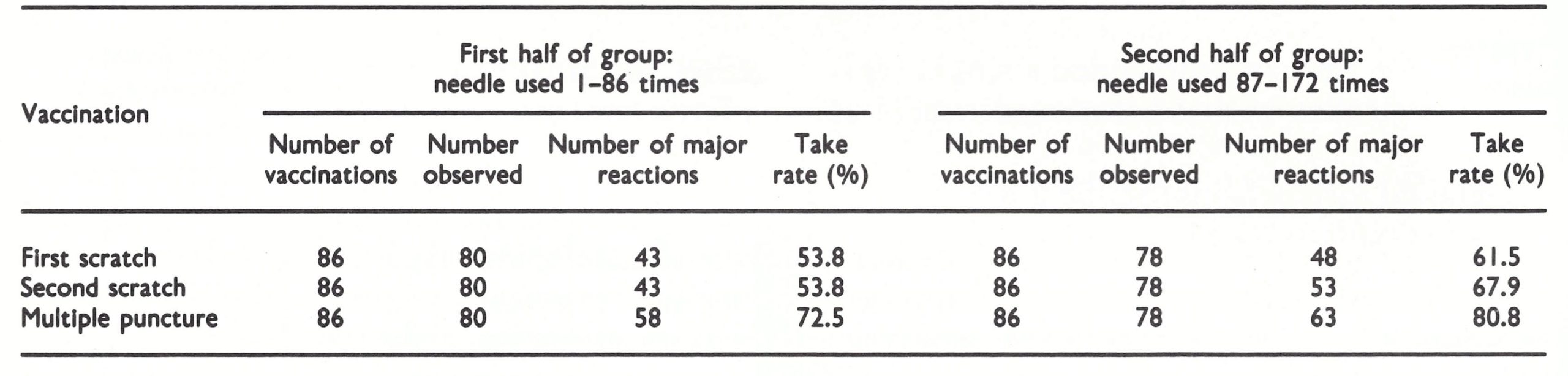
Soon after its development and testing, the bifurcated needle (see Plates 11.14 and 11.15) became the standard instrument for vaccination in the global smallpox eradication programme. Its use simplified vaccination procedures, reduced the quantity of vaccine used and gave a better take rate than earlier vaccination techniques.
History
Liquid vaccine was usually dispensed in sealed capillary tubes containing either one or several doses of vaccine. With single-dose capillaries, the ends were broken and the vaccine applied directly to the inoculation site. With multiple-dose capillaries, the ends were broken and the vaccine was usually put on a plate, from which it was taken up by a glass rod or the vaccination instrument itself and applied to the skin, to be followed by vaccination by scarification (scratch inoculation) or multiple pressure.
When manufacturers produced freezedried vaccine in the 1950s, it was packaged in multidose vials or ampoules, in which the vaccine was reconstituted when required by the addition of sterile diluent. A glass rod was dipped into the container and a droplet of the reconstituted vaccine transferred to the surface of the skin, after which vaccination by scarification or multiple pressure was carried out with a lancet or needle. Since this procedure was rather complicated for use in the field, several manufacturers, including Wyeth Laboratories, Philadelphia, the major producers of vaccine in the USA, sought a better method for transferring vaccine from the vial to the skin. Many types of instrument were tested (see box), resulting ultimately in the development of the bifurcated needle, which was found to produce successful results even when used by vaccinators with little training.
The bifurcated needle was invented by Dr Benjamin A . Rubin of Wyeth Laboratories, and tested in the field by Dr M. Z . Bierly, who used the conventional multiple-pressure method in order to evaluate its efficacy. The needles were patented under United States Patent No. 3 194 237 on 13 July 1965, but Wyeth Laboratories waived all royalties for needles manufactured under contract with WHO.
Use of bifurcated needles in the global smallpox eradication programme
The simplicity of the bifurcated needle as a means of transferring vaccine to the skin and carrying out the vaccination was most attractive but, before recommending its use, WHO organized several studies to ensure that it would be effective in the variety of circumstances met with in various national smallpox eradication programmes. Ladnyi, then a WHO intercountry smallpox adviser in Nairobi, Kenya, Dr H. Mayer, a WHO intercountry smallpox adviser in Monrovia, Liberia, Dr E . Shafa, the regional smallpox adviser in the Eastern Mediterranean Region, and Henderson and Arita from the Smallpox Eradication unit at WHO Headquarters arranged to undertake these studies (SE/72.5). The most important development was the introduction of a new method of vaccination, by multiple punctures rather than multiple pressures.
Vaccination by multiple puncture. Traditionally, vaccine was introduced into the Malpighian layer of the epidermis either by scarification with a needle or lancet or by multiple pressure inoculation with a straight surgical needle (see Chapter 7, Fig . 7.2) . Multiple puncture was impossible with a sharp surgical needle because it would penetrate too deeply ; moreover, there was a belief that if blood was drawn vaccination was less efficacious . However, the flat prongs of the bifurcated needle prevented too deep an entry into the dermis. Experiments soon showed that the multiple puncture method, in which the bifurcated needle was held at right angles to the skin, which was then punctured several times with the prongs (Plate 11.14), was very efficient and very easy even for an illiterate vaccinator to learn. It became the standard method of vaccination throughout the world.
It was observed that, if certain vaccinators recorded more vaccination failures than expected, it was often because they were too gentle, partly because of the above-mentioned belief that bleeding at the inoculation site reduced the take rate. However, experience showed that this belief was unfounded, and vaccinators were advised to use the multiple puncture method with the bifurcated needle with enough force to cause slight bleeding.
Take rate. Tests carried out in Egypt, Kenya and Liberia (Table 11.16) showed that take rates by the multiple puncture method were in the range of 98-100% in primary vaccinations and that reactions specified as major (see Chapter 7) occurred in 56-82% of revaccinations . In experiments by Dr Shafa in Egypt, in which the same individuals were inoculated on opposite arms by different methods the take rates for revaccination by scarification were slightly lower than those obtained with the bifurcated needle.
Amount of vaccine. The amount of reconstituted vaccine taken up by the bifurcated needle was tested in the Smallpox Eradication unit in Geneva and the results were confirmed in the field. It was estimated that one dip of the needle point lifted an average of 0.0025 ml of reconstituted vaccine between the prongs. Since the amount of reconstituted vaccine used in conventional vaccination was about 0.01 ml, vaccination with the bifurcated needle saved vaccine, permitting 4 times as many vaccinations to be administered with a given quantity of vaccine.
Design of the bifurcated needle. The original needle developed by Wyeth Laboratories was designed to be used once only and then discarded . However, for the global eradication programme it was essential to be able to reuse the needles several times and to make them as cheaply as possible. In collaboration with a metallurgical firm, WHO investigated methods of increasing the carbon content so as to produce the hardest possible steel that would not rust. When this steel was used, metallurgical testing showed that there was no change in the “hardness index” after a bifurcated needle was flamed in a spirit lamp up to 50 times, for 3 seconds on each occasion, but that the index decreased considerably if a needle was flamed for 5 seconds on 25 or more occasions. Furthermore, since the major element in the cost of the needles was the amount of steel contained in each, their length was reduced from 65 mm to 50 mm, and they were made somewhat thinner than the original Wyeth needle.
Durability of needles as modified by WHO. Field tests were carried out on the durability of the smaller, hardened needles. Working in Egypt, Dr Shafa (Table 11.17) observed the frequency of take rates when a single needle was used for 172 successive revaccinations with a vaccine having a titre of 108.6 pock-forming units per ml. The needle was flamed before SMALLPOX AND ITS ERADICATION each vaccination by passing it 3 times through the flame of a spirit lamp, and allowed to cool before being dipped into the vaccine vial. Three insertions were performed in each of 172 previously vaccinated individuals; 2 by the scratch method (6-7 mm in length) on one arm, and 1 by multiple puncture (15 strokes within an area 3-5 mm in diameter) on the other arm. The responses were examined on the 6th or 7th day after revaccination. Comparison of the first 86 members of the group with the remainder showed that there were no significant differences in take rates between them but that multiple puncture inoculation gave consistently better results than scratch vaccination. Primary vaccinations performed on 93 people, using the same needle, sterilized by boiling, 46 or 47 times, produced takes in all but one person.
Number of puncture sites. The amount of vaccinia virus entering the epidermis could be varied by altering either the number of strokes or the number of insertion sites. Both variations were tested. Dr M. A. Rahman, of the Public Health Institute in Dhaka, Bangladesh (personal communication, 1967), showed that, after the revaccination of individuals who had been vaccinated within the previous 3 years, major reactions were somewhat more common after 30 than after 15 strokes, whereas 5 strokes sufficed to produce takes in the great majority of primary vaccinations.
Possibly as a result of the use of low-titre liquid vaccine, there was a long-standing tradition in many countries that the chance of vaccination failure could be reduced by inoculating in 2 sites, especially in revaccination. During the late 1960s, several groups of workers studied this problem. Using vaccine that met WHO standards for potency (10 8 pock-forming units per ml) Pattanayak et al. (1970) showed that there was little advantage in using 2 insertion sites (Table 11.18), and that the bifurcated needle was superior to the rotary lancet for both primary vaccination and revaccination.
Lane et al. (1970a) revaccinated 334 subjects using the multiple puncture method with bifurcated needles, 161 of them at a single vaccination site and 173 matched subjects at 2 sites. There was little difference between the 2 groups’ intake rates, and no significant difference in titres of neutralizing antibody. On the other hand, Nyerges et al. (1973) found that neutralizing antibody titres were somewhat higher after insertions at 2 sites and suggested that immunity might be more persistent after vaccination in this way.
However, there was general agreement that it was much more important to administer a potent vaccine than to use 2 insertion sites or to increase the number of punctures . With the improvements in freeze-dried vaccines, it was a great deal easier to ensure that the vaccine was potent than to change the method of vaccination. To make the technique in the field as simple as possible, the Smallpox Eradication unit recommended a single regimen: 15 strokes with a bifurcated needle at 1 site.
Detailed instructions (SE/68.2 Rev.1) were widely circulated and it was pointed out in them that, with the multiple puncture method, a trace of blood indicated that the punctures were likely to have introduced the virus into the epidermis.

Plate 11.13. Benjamin Arnold Rubin (b . 1917) invented the bifurcated needle while working at Wyeth Laboratories, Philadelphia, Pennsylvania, USA.
Plate 11.14. Vaccination with the bifurcated needle. The requisite amount of reconstituted vaccine is held between the prongs of the needle and vaccination done by multiple puncture: 15 strokes, at right angles to the skin over the deltoid muscle, in an area about 5 mm in diameter.
Use of Bifurcated Needles
WHO procured about 50 million bifurcated needles between 1967 and 1976, of which 5 million are being retained in Geneva together with a smallpox vaccine reserve for emergency purposes (see Chapter 28) . The 1970 price of the needles was US$5 per 1000 needles. The other 45 million needles were distributed during the Intensive Smallpox Eradication Programme to practically all the endemic countries in which WHO-assisted smallpox eradication programmes were operating. Additionally, other countries which wanted to strengthen their vaccination programme or had to deal with smallpox epidemics also received the needles . These countries were Democratic Kampuchea, Lao People's Democratic Republic, Malaysia, the Philippines, Sri Lanka and Viet Nam in the Western Pacific and South-East Asia Regions ; Bahrain, Iran, Oman and Saudi Arabia in the Eastern Mediterranean Region, and Argentina, Bolivia, Chile, Colombia, Cuba, Mexico, Peru and Venezuela in the Region of the Americas . If the population of the 31 endemic countries in 1967 is included, the total population of countries in which bifurcated needles were used for vaccination programmes would have been about 2000 million .
Containers for bifurcated needles
The package of smallpox vaccine prepared by Wyeth Laboratories contained freeze-dried vaccine, fluid for reconstitution, and bifurcated needles in a disposable plastic container. Disposable appliances were, however, too expensive to be used in the global smallpox eradication programme, and early in 1968, the Smallpox Eradication unit sought a plastic container which would hold about 100 needles, would be cheap to produce and could be sterilized by boiling. The containers eventually used were designed by Dr Shafa, who was successful in stimulating local producers in Bangladesh and Pakistan to produce the container illustrated in Plate 11.15. After the conical end had been unscrewed, needles were placed in the containers with the prongs towards the base, and were sterilized by placing the closed container in boiling water. The bottom of the container was provided with a few holes so that the water could be drained or shaken off after boiling. In the field, needles could be removed aseptically from the container one at a time through the hole at the apex of the conical lid, and placed in an empty container after use, for sterilization next day. These procedures greatly simplified the vaccinators’ work. Subsequently the WHO Regional Office for South-East Asia produced similar containers, and after 1971 they were widely used in all WHO-assisted national smallpox eradication programmes. It is interesting to note that local producers in Bangladesh, India and Pakistan, in which smallpox was endemic, devised a plastic container made of high-density polyethylene which withstood boiling, whereas efforts made by the Smallpox Eradication unit at WHO Headquarters to persuade plastics manufacturers in Switzerland and the USA to produce such containers were unsuccessful.
Plate 11.15. WHO bifurcated needles and plastic containers. The top of one container has been unscrewed so that it can be packed with needles. Holes are provided in the bottom to allow excess water to be removed after sterilization by boiling. Sterile needles are shaken one at a time through the hole in the conical lid.
Packaging and instruction sheets
Initially the packaging and instruction sheets provided with donated vaccine varied, since they followed the regulations of the donor countries. Usually the multiple pressure or scarification method was described, instructions were given about sterilizing the skin and the numerous and complex contraindications to vaccination were listed.
With the introduction of the bifurcated needle, a simple set of instructions in English and French was included in each box of vaccine. In addition, the contraindications to the use of vaccine in endemic countries were simplified in accordance with the policy of the Smallpox Eradication unit, as follows: “In endemic smallpox regions or in areas geographically proximate only individuals who are severely ill are not vaccinated.” This contraindication was included to avoid vaccination being blamed if the patient died. Later some countries making donations on a bilateral basis—for example, India—prepared their own instruction sheets along the same lines.
As the programme progressed, manufacturers were encouraged to provide the vaccine and the diluent in separate packages, so that only the vaccine needed to be stored in refrigerated space, something that was always in short supply in developing countries.
Jet Injectors
Jet injectors (trade name Ped-o-Jet) played an important role during the initial phase of the Intensified Smallpox Eradication Programme. They were used to ensure rapid vaccination coverage with satisfactory take rates in national smallpox eradication programmes in Brazil, Zaire, countries in western and central Africa, and to a small extent in several other countries. In all these countries bifurcated needles were also used, especially in rural areas and where only small numbers of people required vaccination. In Brazil, the programme was highly organized and there was no shortage of skilled maintenance and repair staff for the jet injectors. In western and central Africa there was a special reason for using the jet injectorr—namely, the simultaneous programmes of vaccination against smallpox and tuberculosis in Zaire and against smallpox and measles in western Africa. In other areas, in which only smallpox vaccine was being administered, the bifurcated needle, by virtue of its simplicity and advantages in field use, had been universally adopted by 1969 and had replaced jet injectors where these had earlier been used experimentally.
History
In immunization campaigns in developed countries, undertaken to control the diseases of childhood, immunizing antigens other than smallpox vaccine were administered with syringes and needles . At that time, before disposable equipment had been invented, sterilization between vaccinations was time-consuming, but necessary if serum hepatitis was to be avoided. The jet injector overcame this difficulty. It consisted, in essence, of a piston which forced a measured dose of vaccine, under high pressure, through a narrow orifice, thereby achieving subcutaneous inoculation without the need for syringe and needle.
Hingson et al. (1963) had used jet injection to administer local anaesthetics, insulin and various antibiotics since 1947, and after 1954 the procedure had been used on military recruits for the subcutaneous injection of influenzavirus and poliovirus vaccines. By 1962, vaccination against cholera, DPT (diphtheria, pertussis and tetanus), typhoid and yellow fever was being carried out with jet injectors in Central and South America. All the antigens concerned were introduced subcutaneously by jet pressure, each being given at a different site.
Smallpox vaccination, however, required intradermal deposition of the virus, and in 1962 Dr Aaron Ismach, of the United States Army Research and Development Command, modified the nozzle to enable it to deposit vaccine intradermally (see Millar et al ., 1969).
Developmental studies
In July 1962, Henderson and Dr J . D. Millar, using this nozzle attached to a Ped-o-Jet (Plate 11 .16), inoculated 41 previously vaccinated young adults with diluted smallpox vaccine; 32 of them showed satisfactory takes (major reactions). These preliminary results prompted a group of scientists in the Communicable Disease Center (CDC-later the Centers for Disease Control) in the USA to conduct a series of developmental studies on the efficacy and safety of intradermal jet injection for smallpox vaccination. Millar et al . (1969) vaccinated 156 volunteers, of whom 16 were unvaccinated and 140 had been vaccinated more than 5 years before, either with undiluted smallpox vaccine by the multiple pressure technique or with 0 .1 ml of various dilutions of smallpox vaccine by jet injector, using the newly developed nozzle. Cutaneous and serological responses in revaccinated persons revealed that jet injection of diluted vaccine with a titre of 106.1 pockforming units per ml1was as effective as multiple pressure inoculation of undiluted vaccine (107.6 pock-forming units per ml). Among the small number of subjects undergoing primary vaccination, jet injection of diluted vaccine with a titre of only 105 .1 rock forming units per ml appeared as effective as multiple pressure inoculation of undiluted vaccine, which suggested that use of the jet injector would result in considerable savings in vaccine . No complications of vaccination, either local (at the inoculation site) or general, occurred in this small series.
Roberto et al..(1969) then conducted studies on cutaneous and serological responses to primary vaccination in 625 children in Jamaica, comparing the jet injection of diluted smallpox vaccine with multiple pressure inoculation of undiluted vaccine. For jet vaccination with diluted vaccine, the take rate depended on the titre as follows:
1Assays of viral infectivity in this study were carried out by titration in primary rhesus monkey kidney cells using half-log dilution steps, and were expressed as TCID50 per ml . Since viral titres elsewhere in this book are expressed as pock-forming units per ml, the published results have been converted from TCID50 per ml to pock-forming units per ml, using a factor provided by J.H.Nakano (personal communication, 1982) . The tissue culture end-points were about 0.9 log unit higher than the titres on the CA membrane.
These results can be compared with the take rate of 96% found in persons vaccinated by multiple pressure with undiluted vaccine (107.6 pock-forming units per ml). Subjects who developed Jennerian vesicles usually developed neutralizing antibody (2 failures out of 105 subjects). Seroconversion was not found in those who failed to develop such vesicles. Vesicles and scars were generally smaller in the jet-vaccinated subjects than in those vaccinated by the multiple pressure technique. Vaccinial complications did not occur in any of the 625 subjects, and infants tolerated jet vaccination satisfactorily. Roberto et al. (1969) concluded that intradermal jet injection of 0.1 ml of vaccine with a titre of 105.4 pock-forming units per ml or higher was a very effective method of achieving successful primary smallpox vaccination.
These studies showed that intradermal jet injection was satisfactory both for primary vaccination and for the revaccination of individuals who had been vaccinated many years before. Neff et al. (1969) followed up this work with an evaluation of the performance of jet injectors, as compared with the multiple pressure technique, in a wellvaccinated prison population, using serial dilutions of vaccine; they concluded that vaccination by jet injection with a vaccine containing 106.1 pock-forming units per ml was at least as efficacious as vaccination by the standard multiple pressure technique.
Freeze-dried vaccine produced by Wyeth Laboratories was employed in all these studies. As used in the field in western Africa, the vials contained a freeze-dried vaccinia virus suspension which was reconstituted with 10 ml of Hanks’ solution, so that 0.1 ml, the volume administered by jet injection, contained at least 106.5 pock-forming units—considerably more than was required, according to the experimental work.
However, since the improvements in vaccine production procedures, outlined in the earlier part of this chapter, ensured that highly potent vaccine was available, titres in excess of those shown to be necessary in trials were recommended so as to provide a safety margin to allow for the effect of the problems in the handling of vaccine that would inevitably occur in the field at some time or another.
Application
Provided that the instruments could be properly maintained, jet injection, using the special nozzle for intradermal injection, had obvious attractions for mass smallpox vaccination campaigns. In 1965 Millar and his colleagues in CDC undertook a pilot study of jet injection and multiple pressure vaccination in Brazil, the results of which stimulated that country to undertake a national smallpox eradication programme (Millar et al ., 1971). They reported that between 27 January and 15 February 1965,47 926 residents of Amapa Territory, Brazil, living in both urban and rural areas, were vaccinated, all by jet injection. For purposes of comparison, the traditional multiple pressure method was used in Mazagao, where 34 vaccinators, 2 supervisors and 2 local staff vaccinated 911 persons in 3 hours. In Amapa town, in contrast, 2 jet vaccinators, 1 recorder and 2 local staff vaccinated 1335 persons in 6 hours (Table 11.19).
Using data obtained in part of this trial and those collected in a conventional mass vaccination campaign in Belem, Millar et al. (1971) analysed the relative costs of the two methods (see Chapter 12, Table 12.5) . Jet injection was found to reduce manpower, transport and vaccine requirements and appeared to be more efficient . In urban campaigns it was estimated that jet injection costs were about one-third of those of conventional techniques. They concluded that jet injection, if intelligently applied, could significantly increase the speed, efficacy and efficiency of national mass smallpox vaccination programmes.
CDC launched a smallpox eradication and measles control programme in 20 countries of western and central Africa in 1967, as part of the WHO global smallpox eradication programme (see Chapter 17) . The use of the jet injector was regarded as the key to this programme, since it not only reduced the needs for manpower, transport, the quantity of vaccine and the chances of unsuccessful vaccination but also made possible simultaneous vaccination against smallpox and measles . In 1966, when the Director-General of WHO submitted a report to the Nineteenth World Health Assembly on the organization of the global programme, he specifically referred to the usefulness of intradermal jet injection for vaccination when large groups could be assembled. WHO provided some 100 jet injectors to the national programmes in Pakistan, the Sudan and Zaire in 1967 and 1968. Some 10 jet injectors were also kept in the Regional Offices for the Eastern Mediterranean, Africa and South-East Asia and in the Smallpox Eradication unit, for emergency use. However, largely because of problems with their maintenance and repair, they were little used except in Brazil, western Africa and Zaire, and briefly in India and Indonesia.
Scars after Vaccination with jet Injectors
During the Intensified Smallpox Eradication Programme, vaccination scar surveys were often carried out to determine the vaccination coverage of populations . Since the experimental studies with jet vaccination had shown that the resulting lesions and scars were somewhat smaller than those produced by other methods of vaccination, there was some question whether the scars would persist for as long. Dr D. R . Hopkins, of the Communicable Disease Center (CDC) in the USA, studied the persistence of vaccination scars when he was working in Sierra Leone in 1967-1968 and concluded that there was essentially no difference in persistence, whether scars were produced by jet vaccination or by multiple puncture. In 1982, Dr A . Gromyko, while working for the Smallpox Eradication unit, carried out further investigations in Sierra Leone and Cote d'Ivoire and confirmed that, in 80% of subjects, vaccination scars persisted for more than 12 years.
Vaccine for jet injectors
The vaccine to be used in jet injectors had to meet special requirements, relating both to bacteriological sterility and to potency. Because the vaccine was administered parenterally, it was essential that it should have a low to nil bacterial count when tested in the laboratory. This recognized the fact that bacterial sterility could not be achieved unless the vaccine was produced in eggs or tissue culture (see box opposite), and no laboratories at that time produced such a vaccine which met other WHO standards of potency and heat stability. Although the vaccine had inevitably contained a few non-pathogenic bacteria, no unusual complications due to bacterial infection had been observed during the extensive field trials of the jet injector. As a working standard, a WHO Scientific Group on Smallpox Eradication (1968) noted that vaccine containing up to 5 non-pathogenic bacteria per ml had been extensively used for jet injection without untoward effects.
In the smallpox eradication programmes carried out with United States assistance in 20 countries of western and central Africa, freeze-dried calf lymph vaccine produced by Wyeth Laboratories was used. Low or zero bacterial counts per ml were achieved with this vaccine by frequent and meticulous cleansing of the animal skin from the time of inoculation to that of harvest. Producers in Switzerland and the USSR were likewise successful, as were some others, in providing a vaccine of low bacterial content, and this vaccine was used in Zaire. For Brazil, the vaccine was produced in Brazilian laboratories, and although many batches of the vaccine contained more than 5 non-pathogenic bacteria per ml, and sometimes some pathogenic bacteria, no adverse consequences were detected during the eradication programme in Brazil.
The other feature of the vaccine used for jet injection was that the recommended titre of the reconstituted vaccine was lower than that required for multiple pressure vaccination—namely, 106.5 instead of 108.0 pock-forming units per ml—since all the virus in the injected dose was introduced into the skin, rather than a minute fraction as in the multiple puncture method. Wyeth Laboratories produced a special vaccine vial which, when reconstituted with 50 ml of saline (a quantity sufficient to vaccinate 500 persons), gave this titre. When other vaccines were used, a dilution factor was chosen to give the same final concentration. For example, the vial used for vaccine produced in the USSR contained the freeze-dried residue of 0 .2 ml of vaccine with a minimum concentration of vaccinia virus of 108.0 pock-forming units per ml. One ampoule of this vaccine diluted with about 6.6 ml of saline therefore gave a final concentration of about 10 6.5 pock-forming units per ml. However, since in practice the vaccine had a virus concentration of 2 or 3 x 108 pock-forming units per ml, the vaccine in 2 ampoules was usually diluted with 25 ml of saline (a lot size easily available on the market) for intradermal jet injection. In places in which the Wyeth vaccine and the corresponding diluent were not available, WHO provided national eradication programmes with special vials containing 25 ml of saline together with instructions for dilution. Sometimes, by mistake, distilled water was used to reconstitute the freeze-dried vaccine. Vaccine so reconstituted caused a sharp pain when injected, which was not the case when saline was used as the suspending fluid, but it proved satisfactory otherwise.
Manuals in English, French and Portuguese, describing the maintenance and repair of jet injectors, were developed by the Communicable Disease Center and produced for national vaccination campaigns in which this instrument was used.
Discontinuation of use ofjet injectors
A few disadvantages of jet injectors emerged during the campaigns in which they were used . In contrast to the simplicity of bifurcated needles, the jet injector required meticulous care and maintenance and considerable repair skills, which could not always be provided despite all the efforts to prepare a detailed, profusely illustrated manual . Furthermore, the instrument was expensive and heavy . With the introduction of bifurcated needles in 1968, it became apparent that these were far more functional, and in well-organized campaigns it was found that individual vaccinators could vaccinate 1000-1500 persons per day . At the same time, experience with jet injectors showed that vaccinations teams were seldom able to assemble more than 2000-3000 persons per day, on average, hence the jet injectors offered little advantage. Thus their use was largely confined to the programmes begun in 1967-1968-in Brazil, countries of western and central Africa, and Zaire.
Other jet injectors
In 1968 and 1969, Arita coordinated investigations of several other devices developed by various manufacturers: the Press-o-Jet, a hand-operated jet injector produced in the USA, the Dermojet (modified as the Vaccijet) produced in France, and the Porton needleless injector produced in the United Kingdom. Some experimental models were sent to Kenya and their suitability was tested in the field by Ladnyi. None of these instruments proved as satisfactory as the better-established Ped-o-Jet with regard to take rates, mechanical reliability or general convenience. By 1969, the advantages of the bifurcated needle had been fully recognized, and the studies were discontinued.
Bacterial Counts of Vaccine Used in Jet Injectors
Bacteriologically sterile smallpox vaccine can be produced on the CA membrane or in cultured cells. However, the vast majority of producers used animal skin, and the resulting vaccine always contained some bacteria. The number of viable bacteria could be reduced by treatment with phenol, but it was impossible to produce bacteriologically sterile vaccine which remained potent in terms of its viral content. The term "bacterial count zero" (see Table 11.7) does not mean "bacteriologically sterile" but only that no viable bacteria were found in the samples tested.
A conflict arose in the framing of standards for vaccine for use in jet injectors because of the insistence by experts on biological standardization that all vaccine designated for parenteral use in man should be bacteriologically sterile and the pragmatic view expressed by the WHO Scientific Group on Smallpox Eradication (1968) that freeze-dried vaccine containing up to 5 non-pathogenic bacteria per ml of reconstituted vaccine had been used for jet injectors in recent extensive trials without untoward effects . In fact, no new standards were drawn up by WHO. However, in 1969 the United States health administration proposed that the vaccine for jet injectors should pass the same sterility test as that used for other vaccines for parenteral use (Federal Register, 1969). At that time Wyeth Laboratories were supplying the jet injector vaccine, and although the bacterial count was often zero it was not bacteriologically sterile . Hence the concern of Dr J.H. Brown of Wyeth Laboratories, Henderson of WHO, and Dr D. Millar of CDC, all of whom believed that the requirement was academic and would hamper the progress of the eradication campaign in western Africa, sponsored by the USA, where jet injectors were a major tool. Discussions took place with the United States health administration and eventually the requirement was not imposed .
MODIFICATION TO VACCINATION PROCEDURES
The principal modification to previously used procedures related to the cleansing of the skin prior to vaccination. It was generally believed that bacteria on the surface of the skin might cause infection if introduced during vaccination. For smallpox vaccination, it was usual to cleanse the skin with acetone or 70% alcohol. With the latter, such cleansing, to be effective, had to continue for about 15 seconds, although even then there was no assurance that the skin would be free of sporeforming bacteria. However, if vaccine was deposited on the skin before it was completely dry, the potency could be reduced by the residual alcohol.
According to the Memorandum on Vaccination against Smallpox (England and Wales, Ministry of Health, 1962), “Many doctors use nothing at all if the arm is reasonably clean, and there is no evidence to condemn this practice”. Subsequently, Dann (1966) reported that, when 1078 intradermal, subcutaneous, intramuscular and intravenous injections were done without preparation of the skin, there was no subsequent infection. He concluded that “at best, then, pre-injection skin preparation reduces the risk of infection and probably in practice it has no useful effect whatsoever”. The Handbook for Smallpox Eradication Programmes in Endemic Areas (SE/67.5 Rev.1) stated that no pretreatment of the skin was necessary for vaccination but that, if the site was obviously dirty, the skin should be wiped with a cloth moistened with water. This minor change from the traditional method greatly facilitated vaccination programmes, since it dispensed with the need for additional material such as cotton swabs and antiseptics and speeded up the operation . It was subsequently discovered that, in the smallpox eradication programme in Iran in 1953-1961, glycerolated lymph had been administered by the scratch method to over 30 million persons in whom the site was not cleansed, since the use of alcohol and even soap was not recommended for fear of inactivating the virus: no serious local infections were reported (WHO/SE/78 .120).
Neonatal Vaccination
As has been noted in Chapter 7, in the early 1960s Rao had introduced the practice of neonatal vaccination in hospitals, first in Madras and subsequently throughout urban areas in south India. The WHO Expert Committee on Smallpox (1964) recommended that in endemic areas primary vaccination should be carried out as early as possible, preferably in the neonatal period, and repeated about 12 months later. This view was reinforced in 1967 (WHO Scientific Group on Smallpox Eradication, 1968) and again in 1971 (WHO Expert Committee on Smallpox Eradication, 1972).
During the intensified eradication programme in India, neonatal vaccination was widely practised in municipalities and corporations, where the majority of births occurred in health institutions and maternity centres whose staff were provided with vaccine and bifurcated needles and trained in their use. Similar procedures were adopted in certain African countries, where births occurred in health institutions or were assisted by experienced midwives. However, in the rural areas of the Indian subcontinent and in most parts of Africa, neonatal vaccination was not possible, and efforts were therefore made to vaccinate infants at the first health examination, where such facilities existed.
THE SEARCH FOR NEW VACCINES
As has been mentioned earlier in this chapter, at the outset of the Intensified Smallpox Eradication Programme in 1967 the Smallpox Eradication unit reached the conclusion that smallpox could be eradicated by the effective use of the vaccine then available . Not only did it see no need for a new vaccine but it also feared that the trials that such a product would have to undergo would act as a brake on the global eradication programme . Since, despite the tremendous amount of work involved in coordinating this programme, the unit at its largest consisted of only 6 professionals and 4 supporting staff, extremely careful attention had to be given to the determination of priorities.
However, advanced industrial nations which had eliminated smallpox decades earlier saw the problem from a different perspective. Health officials and the public alike were concerned by the sickness, occasional complications and, rarely, death that followed the administration of existing smallpox vaccines. Virology had advanced a long way since 1798, when vaccination had first been introduced, and many workers, especially in Europe, Japan and the USA, sought a vaccine that would be associated with milder lesions after primary vaccination and especially with less likelihood of complications.
Methods of improving smallpox vaccine, from the point of view of reducing complications, were extensively discussed; first, in 1969, at a symposium on smallpox organized in Zagreb by the Yugoslav Academy of Sciences and Arts, (Gusic, 1969) and again in 1972, when a special session on smallpox vaccination was convened in Bilthoven by the International Association of Biological Standardization (Regamey & Cohen, 1973). Three approaches were adopted in the studies designed to develop less reactogenic vaccines : (1) selection of the least reactogenic strains from among those currently being used for vaccine production ; (2) development of an attenuated strain; and (3) use of inactivated vaccine. The second and third approaches included attempts to develop a method in which attenuated or inactivated vaccine was first used to provide an initial immunological stimulus and thus partial protection, followed by vaccination with the usual smallpox vaccine, a procedure which should in theory reduce complications. A fourth series of investigations, largely independent of the issue of vaccinial complications, was aimed at developing a tissue culture vaccine which, unlike vaccine of animal skin origin, would be sterile. None of these attempts resulted in an alternative vaccine which could be widely used for the global smallpox eradication programme . Nevertheless, the efforts of laboratory investigators and epidemiologists in these once important research activities are significant for the historical record. If attempts to immunize human beings against a variety of diseases by the incorporation of designated foreign genes in vaccinia virus are successful, there will be renewed interest in methods of reducing the incidence of severe complications.
Selection of Vaccinia Virus Strains of Low Pathogenicity
Polak et al. (1963) reported on the pathogenicity to man of vaccines made with the Bern, Copenhagen, Ecuador and Lister strains of vaccinia virus. The generalized responses, in terms of the degree of morbidity (the ratio of number of bed-patients to number of successful vaccinations), high fever, and prolonged fever in bed-patients, were recorded. The Lister strain produced the mildest response, followed by the Ecuador strain. The Copenhagen and Bern strains were similar in their effects and of greater pathogenicity than the other two. Thus, when a sound evaluation method was used with adequate controls, it was demonstrated that vaccinia strains differed in their pathogenicity to man. In addition, the study suggested that the potency of the vaccine (in terms of its titre) seemed to have no bearing on the course of illness following vaccination. These results supported the views held by many epidemiologists that different strains of vaccinia virus were associated with different frequencies of complications (see Chapter 7). For example, the Bern strain, once used in Austria, Germany, Switzerland and Yugoslavia, had been associated with much higher complication rates (especially of postvaccinial encephalitis) than those reported in the United Kingdom (in which the Lister strain was used) or the USA (in which the New York City Board of Health strain was used). By 1971, these countries, with the exception of Yugoslavia, had changed to the Lister strain for vaccine production, and from that time on the complication rates decreased.
In the 1960s, Dr Marennikova and her colleagues in Moscow collected vaccinia strains from different vaccine producers and studied their pathogenicity, as determined by inoculation by various routes into rabbits, mice and irradiated rats. Table 11 .20 sum marizes the results obtained for a number of different strains (Marennikova et al., 1969). Subsequent investigations (Marennikova, 1973) showed that the Tashkent strain was associated with much higher levels of reported postvaccinial complications (46 cases per million doses distributed, with 18 per million of encephalitis) than the B-51 or especially the EM-63 strain (17 per million doses with 7 per million cases of encephalitis for EM-63). Likewise, the Wyeth (New York City Board of Health) strain, of low pathogenicity for experimental animals, was associated with a relatively low rate of postvaccinial complications (see Chapter 7).
This work, which was presented at two major conferences of vaccine producers, influenced health authorities’ decisions as to the choice of a strain, particularly as additional epidemiological data became available. For example, the Tashkent strain, which according to Dr Marennikova was highly pathogenic for animals, had been used in the USSR for the production of smallpox vaccine for local use and for donation to India and other Asian countries up to 1966. It caused considerable concern among Indian health workers because of the severe local reactions (Goyal et al.,1969), and after 1966 its production was discontinued in the USSR.
Table 11.21, based on data from WHO surveys in 1968 and 1971, shows how vaccine producers in several countries changed from the strains they had previously been using to the less reactogenic Lister strain, which was distributed by the WHO International Reference Centre both as a reference vaccine for potency assays and as seed lots for vaccine production. Of 71 vaccine producers in 49 countries or areas, 42 (59%) were using the Lister strain in 1971 compared with 22 (31 %) in 1968.
In Japan, complications of smallpox vaccination caused substantial public concern in the early 1970s and the health authorities reevaluated their traditional Ikeda vaccinia strain in comparison with the Lister strain. Country-wide studies of vaccinial reactions, such as fever, the size of lesions, and the need for hospitalization, showed that the Lister strain was less reactogenic, and all 6 Japanese production laboratories changed to it in 1971.
Attenuated Strains
In 1931, Dr T. M . Rivers of the Rockefeller Institute of Medical Research, New York, initiated a series of passages of the New York City Board of Health strain of vaccinia virus through minced chick embryo cells, primarily in order to obtain a bacteria-free vaccine (Rivers, 1931. Passage in rabbit testes was included, initially to obtain a bacteria-free preparation for passage in tissue culture and later to restore the pathogenicity of the virus for rabbit skin. In the process, the virulence of the virus became attenuated.
Two strains were used in subsequent studies; their passage history has been summarized by Barker (1969). Strain CVI-78 had been passed 124 times in chick embryo explants and 19 times on the CA membrane; strain CVII had been carried for a total of 235 passages in chick embryo explants. Rivers & Ward (1935) showed that CVII was suitable for human vaccination by intradermal inoculation. Noting that this tissue culture vaccine consistently produced less severe reactions in rabbits and humans than did the New York City Board of Health calf lymph vaccine, Rivers et al . (1939) carried out further tests, which showed that primary vaccination by intradermal injection of the high-passage tissue culture virus produced only red papular lesions; pustules did not develop and there were few constitutional symptoms. However, they believed that such vaccination would not give complete protection against smallpox, and they recommended that revaccination with the calf lymph vaccine should be carried out 6 months to 1 year later to produce solid and lasting immunity.
The Rivers strains attracted the attention of Dr C. H. Kempe, of the University of Colorado Medical Center, Denver, USA, who was interested in the use of a less reactogenic vaccine in order to reduce the severity of reactions in children suffering from eczema or with other contraindications to vaccination. Using CVI-78, Kempe et al. (1968) vaccinated 1009 patients suffering from eczema (879 primary vaccinations ; 130 revaccinations), 326 by the multiple pressure method and the rest by subcutaneous inoculation of graded doses. Local reactions and temperature elevations were much milder than those seen in children vaccinated with standard vaccine and, except for 2 cases of mild erythema multiforme, no patients suffered from virus dissemination or other complications. The mean neutralizing antibody titre of 162 subjects given primary vaccination with CVI-78 by multiple pressure was very similar to that obtained in 45 controls vaccinated with calf lymph vaccine.
Vaccine produced on the CA membrane from the CVII strain of Rivers was used over a period of 5 years for the primary vaccination of more than 60 000 army recruits in the Netherlands to evaluate whether such a strain would reduce complications, as compared with the conventional calf lymph vaccine, which at that time was produced with the Lister strain (Noordaa et al., 1967). All of the recruits received, in addition, 2 ml of vaccinia- immune globulin. The local reaction after primary vaccination with the CVII strain was milder than that with other vaccines and there was only 1 mild case of postvaccinial encephalitis in the 60 000 vaccinations, but the neutralizing antibody titres measured a year after vaccination were also somewhat lower than usual. The research workers in the Netherlands concluded that the use of the CVII strain would probably reduce complications, but that, to produce sufficient protection against smallpox, it should be followed by revaccination with standard calf lymph vaccine 12 months later.
Tint (1973) summarized experience with primary vaccination with the CVI-78 strain in 9000 subjects (3500 of whom had eczema or other skin diseases) in England, Japan and the USA. He suggested that vaccination with this attenuated strain on its own was probably not sufficient to provide protection against smallpox, but that its use in eczematous children as a preliminary to vaccination with standard vaccine would substantially lower the risks of eczema vaccinatum. Such a regimen might be feasible in industrialized countries, but clearly a two-step schedule was out of the question for vaccination in the global smallpox eradication programme.
Because of increasing concern about the morbidity and mortality associated with smallpox vaccination, the National Institute of Allergy and Infectious Diseases, National Institutes of Health, USA, sponsored a study of the reactogenicity and immunogenicity of 4 vaccines: calf lymph and egg vaccine made from the New York City Board of Health strain, egg vaccine made from CVI-78, and Lister sheep vaccine, administered at several dosages by percutaneous and subcutaneous routes (Galasso, 1970). The results were published in 1977 in 6 papers in the Journal of Infectious Diseases. Primary vaccination by the subcutaneous route, while accompanied by lower rates of fever, led to unsatisfactorily low antibody responses both initially and after standard percutaneous revaccination (Galasso et al.,1977). In a comparison of the 4 vaccines, it was concluded that:
“For percutaneous primary vaccination the CVI strain was 10-fold less infectious than the other three vaccines. CV-I also differed from the other three vaccines in that it produced smaller skin lesions and vaccination was not associated with a febrile response. Only 30% of recipients of primary percutaneous CV-I vaccination with primary type skin responses developed neutralizing antibody; in contrast neutralizing antibody occurred in 82%-85% of the recipients of the other three vaccines.
“After standard challenge vaccination, those children with previous successful percutaneous CV-I vaccination were more likely to have a primary-type skin response. CV-I vaccinees also tended to have larger skin lesions after revaccination, but fever and minor complications were not more frequent. One month after revaccination, neutralizing antibody was present in 93%-96% of those with “takes” on primary vaccination with NYC-CL, NYC-CAM, or Lister vaccines, in contrast to only 75% in CV-I vaccinees.”
Thus the Rivers attenuated strains, which had been studied most extensively (Galasso et al.,1977), appeared to be insufficiently immunogenic for use in vaccination against smallpox.
Workers in several other countries developed attenuated vaccines during the 1970s since it was believed at that time that smallpox vaccination would have to be maintained for many years after eradication and that an attenuated vaccine would then be necessary. In the Federal Republic of Germany, Stickl and his collaborators (Hochstein-Mintzel et al.,1975) produced a highly attenuated strain of vaccinia virus (MVA) by 572 serial passages of the Ankara strain in chick embryo fibroblasts. In the process, the molecular weight of the viral DNA was diminished by 9% and the strain was shown to have greatly reduced virulence for the chick embryo, laboratory animals and man (Mayr et al.,1978), but its immunogenicity was never adequately tested. Stickl et al . (1974) proposed that it should be used : (1) routinely as pre-immunization for primary vaccinations, when it should be followed by conventional vaccine; and (2) for both primary vaccination and revaccination of all individuals at special risk (e.g., with eczema or under immunosuppression). Its probable safety in immunosuppressed individuals was suggested by experiments in irradiated rabbits reported by Werner et al. (1980).
In Japan, Tagaya et al.(1961) recovered an attenuated strain (DIs) from the standard Japanese vaccine strain Dairen by passage in 1-day-old chick embryos. It produced very small pocks on the CA membrane and was not pathogenic for mice, guinea-pigs or rabbits, but produced small skin lesions and induced antibody production after scarification of the skin of cynomolgus monkeys . The immunity produced in rabbits was poor but antibody levels in monkeys were similar to those produced by the parent strain (Kitamura et al.,1967). Tagaya et al. (1973) concluded that DIs was not suitable for use as a smallpox vaccine, but it might have a role if “prevaccination” became an accepted practice.
Stimulated by Tagaya’s results, Hashizume (1975) deliberately sought attenuated variants of the Lister strain by serial passage in rabbit kidney cells at 30 °C and subsequent selection of a small pock from the CA membrane. The variant most extensively studied, LC16m8, was much less pathogenic after intracerebral inoculation in monkeys than CVI, EM-63, Lister or the New York City Board of Health strains (Hashizume et al.,1973), but produced a satisfactory immune response (haemagglutinin-inhibiting and neutralizing antibody) in humans as well as in vaccinated animals (Hashizume, 1975).
in vaccinated animals (Hashizume, 1975) . In 1974 the Smallpox Vaccine Committee, Ministry of Health, Tokyo, organized a large scale field evaluation of a number of vaccines, including one produced in rabbit kidney cells with the LC16m8 strain, more than 40 000 persons being vaccinated (Japan, Ministry of Health, 1975). There were no notifiable severe complications among these subjects, those receiving LC16m8 vaccine being closely followed. While the take rates with the LC16m8 vaccine were not significantly different from those of the other vaccines tested, the fewest general responses and smallest local responses were obtained with this vaccine (Table 11 .22). Among those receiving LC16m8 vaccine, 1 case of eczema vaccinatum, 3 cases of convulsions and 8 cases of generalized vaccinia were discovered, all of which were mild. It was not clear whether the 3 cases of convulsions were related to the vaccine. The examination of 142 vaccinated subjects by electroencephalography showed that the number of temporary anomalies was lowest in those vaccinated with the LCI6m8 vaccine (Table 11.23).
Tests for immunogenicity were carried out in 138 persons vaccinated with LC16m8 vaccine by challenging them with Lister vaccine 12 months later. Major reactions were seen in 18.8%, a rate similar to that observed in 714 persons vaccinated with the Ikeda strain, and challenged with that strain a year later. Thus it appeared that the immunogenicity of the LC16m8 vaccine was similar to that of other vaccines. Although no field experience was available to provide evidence of the protective effect of this vaccine against smallpox, freeze-dried LCI 6m8 strain virus grown in rabbit kidney cells is being held as part of a reserve stock of vaccines in Japan. Because of its low reactogenicity and because it can be produced in tissue culture, the LC1 6m8 strain might be a good candidate for use as a vector for other antigens should this procedure become a practical proposition. The average loss of titre of this vaccine after 4 weeks at 37°C was estimated to be about 100.4 pock-forming units per ml, which is comparable to that of calf lymph vaccine of acceptable heat stability (T. Kitamura, personal communication, 1984).
In 1970 workers in China produced an attenuated variant (G-9) of the Temple of Heaven strain by inoculating children with plaque-purified material and selecting from among those reacting with small skin lesions. The strain produced smaller pocks than the parental strain on the CA membrane and has been used experimentally for the primary vaccination of several million children, but no reports on its immunogenicity or of complications following its use are available.
Inactivated Vaccines
Prior to 1960 only a few live vaccines were available—namely, those for smallpox, tuberculosis and yellow fever—whereas many vaccines consisted of inactivated antigens—pertussis, typhoid, cholera, diphtheria toxoid, tetanus toxoid, etc. Although there are theoretical reasons, discussed in Chapter 3, why inactivated vaccinia virus vaccines, without a follow-up with live virus vaccine, are unlikely to be effective, several research workers undertook developmental studies of inactivated smallpox vaccine in the hope that it might reduce the complications of vaccination.
The earliest study was that reported by Janson (1891), who found that subcutaneous injections of heat-killed vaccine gave equivocal results in children. Many other methods of inactivation were employed (Kaplan, 1962, 1969; Turner et al .,1970) including the use of formaldehyde, ultraviolet irradiation and photodynamic inactivation. Although some of the resulting inactivated vaccines produced neutralizing antibodies in rabbits, none was satisfactory for the primary vaccination of human subjects.
Because complications, especially postvaccinial encephalitis, were very much less common after revaccination than after primary vaccination, several attempts were made to “pre-immunize” subjects with inactivated virus, before inoculation with standard vaccine 1-2 weeks later. Herrlich (1959, 1964) prepared an inactivated vaccine by treatment with formaldehyde, which was used on a small scale in the Federal Republic of Germany and in the German Democratic Republic from the late 1950s as a form of pre-immunization designed to reduce the risk of vaccinial complications. The procedure was to use inactivated “vaccinia-antigen” first and then, 2-3 weeks later, to give conventional live virus vaccine as a booster. In some cases in which pre-immunization was used, the second vaccination produced a large swollen area of erythema and induration surrounding the site of inoculation. In any case, the procedure did not completely eliminate complications . “Vaccinia-antigen” plus vaccinia-immune gamma-globulin, but without follow-up live virus vaccine, was given to 2 elderly patients involved in the Meschede outbreak of smallpox (see Chapter 4) 10-14 days before their exposure, but did not protect either from smallpox, from which one of them died (Wehrle et al.,1970).
A two-step procedure, using gamma-irradiated vaccine for the priming dose, was carried out in a field trial in eastern Europe in 1977 (Marennikova et al ., 1978c). One case of postvaccinial encephalitis occurred in a child with congenital macrocephaly, among some 23 000 vaccinated subjects, all of whom were over 3 years of age (S. S. Marennikova, personal communication, 1985).
Production of Vaccine in Eggs and Tissue Culture
Vaccine production on the CA membrane
Soon after the demonstration by Goodpasture et al. (1932) that vaccinia virus would grow on the CA membrane, Goodpasture & Buddingh (1935) published a detailed analysis of the suitability of eggs for the large-scale production of vaccine. The advantages were that production methods were relatively simple and that bacteriologically sterile vaccine could be obtained. Glycerolated egg vaccines were in use in the state of Texas, USA, from 1948 (Cook et al., 1953) and in New Zealand and Sweden from the 1960s.
Freeze-dried vaccine derived from the CA membrane was produced on an experimental scale by Jackson et al. (1956) and on a commercial scale in Sweden (Hedstrom, 1970). Freeze-dried vaccine prepared on the CA membrane largely replaced calf skin vaccine in Brazil in 1958 (Clausell, 1963) and was used on a very large scale throughout the smallpox eradication programme in Brazil (Voegeli, 1973). Unfortunately, many batches of the Brazilian vaccine did not meet WHO standards for heat stability (see Chapter 12).
Although there has been no evidence of complications or adverse effects caused by avian leukosis viruses in the millions of subjects vaccinated against yellow fever with virus grown in eggs, these agents became a matter for concern when, in about 1967, it was discovered that they were commonly present in eggs. Early in 1970 Swedish producers changed to eggs from flocks free from avian leukosis for smallpox vaccine production.
Vaccine production in cultured cells
As has already been described in relation to the development of the CVI and CVII strains of attenuated vaccinia virus, production in cultured cells began even earlier than in eggs (Rivers, 1931). Over the next 3 decades efforts were made periodically to produce vaccine in cultured cells, at first from chick embryos and later in cell monolayers derived from a variety of sources. However, as with egg vaccine, tissue culture vaccine was not widely adopted, mainly because production in animal skin was simple and cheap and yielded a vaccine that was heat-stable when freeze dried and was known to protect against smallpox. Furthermore, the seed lot virus system had not been well established when cultured cells first became available for the commercial production of smallpox vaccine, and it was feared that serial passage of vaccinia virus in tissue culture might lead to its attenuation. After 1967, when the Intensified Smallpox Eradication Programme was initiated, WHO did not promote the production of vaccine in cultured cells because it was realized that the success of the eradication campaign was heavily dependent on the production of large amounts of vaccine in laboratories in developing countries, which were unlikely at that time to be able successfully to produce tissue culture vaccine.
In the late 1960s, the WHO International Reference Centre for Smallpox Vaccine initiated a development study with Lister vaccine grown in primary rabbit kidney cells. At the time this was the only tissue culture vaccine comparable with conventional calf lymph vaccine in terms of heat stability, immunogenicity and reactogenicity (Hekker et al ., 1973a). The production method was simple and cheap, the vaccine was sterile and free from mycoplasmas and other adventitious agents, and it maintained its potency for up to 8 weeks at 37°C. The actual reductions in titre, ranging from 100.2 to 100.4 pock-forming units per ml after 8 weeks at 37°C, were smaller than those of the calf lymph vaccine tested as a control . The immunogenicity of this vaccine was tested by measuring the neutralizing antibody titres in subjects 1 year after primary vaccination, a control group having been vaccinated with conventional calf lymph vaccine . All the subjects produced neutralizing antibody and there was no significant difference in the results obtained, as between tissue culture and calf lymph vaccines. (Hekker et al .,1973b). Measurement of the antibody titre 2 months after revaccination with the tissue culture vaccine showed an adequate booster effect compared with the calf lymph control group.
Because of these successful results, a field trial on a large scale in Lombok, Indonesia, was jointly organized in 1973 by the Smallpox Eradication unit, the WHO Regional Office for South-East Asia and the government of Indonesia. A total of 45 443 children under the age of 15 years were vaccinated with tissue culture vaccine and the results compared with those for 9061 children of a similar age and sex distribution who had been vaccinated with the standard Lister strain calf lymph vaccine (Hekker et al ., 1976). The success rate with tissue culture vaccine reached 97% in primary vaccination and 75% in revaccination (Table 11.24), results comparable with those obtained with the calf lymph vaccine.
The children were carefully followed up for vaccination complications. The only suspected complication was a fatal case of possible encephalitis in a 5-month-old girl who had been vaccinated with tissue culture vaccine, but in Lombok there were many other possible causes of this disease.
Both this tissue culture vaccine and the LC16m8 strain of Hashizume, which was also produced in primary rabbit kidney cells, met the WHO requirements for safety, potency and stability, and were comparable to calf lymph vaccine in effectiveness for both primary vaccination and revaccination. These 2 strains are the only tissue culture vaccines which have been thoroughly and systematically investigated in the laboratory and also to a limited extent in the field. In the Netherlands, the Lister tissue culture vaccine is kept as the vaccine stock for emergency use, both locally and for supply to other countries ; the LC16m8 vaccine is kept in Japan as a local reserve stock. Either of these vaccines should be suitable for use in the future, should the need arise, when production methods using animal skin are unlikely to be acceptable.
Avian Leukosis and Egg Vaccine
The WHO requirement for smallpox vaccine from chick embryos (revised in 1965 ; WHO Expert Group on Requirements for Biological Substances, 1966) indicated that "only eggs from flocks known to be free from disease, including avian leukosis, shall be used". In 1967 a Swedish laboratory proposed to donate egg vaccine to WHO. The vaccine was sterile and thus suitable for jet injector use, but it was produced from flocks which had not been tested for avian leukosis, whose pathogenicity for man was then unknown. In Sweden, egg vaccine had been used for a long time, and Espmark (1969) reported that there had been no increase in leukaemia in the population . Yellow fever vaccine was also being produced in eggs from untested flocks . Despite arguments advanced by Dr Holger Lundbeck, Director of the National Bacteriological Laboratory, Sweden, and by Henderson, the Chief of the Biological Standardization unit of WHO did not agree to accept the donation from Sweden . The Swedish laboratory finally produced a vaccine from leukosis-free flocks and donated vaccine to WHO . However, in Brazil, smallpox vaccine from eggs produced from flocks which had not been tested for avian leukosis, but were almost certainly carriers of the virus, was used throughout the eradication programme, with no known ill effects.
Silicone Ointment Vaccine
Trials were carried out with freeze-dried vaccine suspended in an ointment, so that it could be squeezed on to the skin in the same way as toothpaste from a tube. An ointment vaccine produced by mixing liquid vaccine lymph with lanoline had been produced at the Lister Institute and in Nigeria in the 1960s, but was no more stable than liquid vaccine. In the early 1970s, the Vaccine and Serum Institute, Berne, Switzerland, developed a similar vaccine, in which freeze-dried vaccinia virus was mixed with silicone ointment. The Smallpox Eradication unit recognized the potential value of such preparations for field use and undertook a development study together with the producer and the WHO reference centres. The titration of this preparation presented a problem, since the vaccine was not readily dispersed. After this difficulty had been solved, the WHO International Reference Centre for Smallpox Vaccine discovered that the heat stability was less than that of the standard freeze-dried vaccine. The manufacturers tried unsuccessfully to improve the stability, and the vaccine was never used in the global smallpox eradication programme.
EFFICACY OF VACCINATION
The most persuasive evidence that vaccination was effective in preventing smallpox was the progressive decrease in the incidence of the disease that followed its introduction at the beginning of the 19th century, the progressive elimination of the disease from Europe and North America in the middle years of the 20th century and final global eradication of smallpox in 1977. All this happened without the benefit of a controlled trial of the kind that would now be used for evaluating the efficacy of a newly developed vaccine.
Until the Smallpox Eradication unit carried out in 1967-1968 the work described earlier in this chapter, neither the potency of vaccines nor the methods of inoculation were standardized, so that the difficulty of evaluating vaccine efficacy and the duration of protection was often compounded by the use of vaccine of low potency or by unsatisfactory techniques. Even during the Intensified Smallpox Eradication Programme it was surprisingly difficult to obtain accurate and reliable data on the level of protection against variola major provided by vaccination. The reason for this is that allocation of an individual to the “vaccinated” category has always been made on the basis of the presence of a scar attributed to smallpox vaccination . Prior to the improvements in vaccine achieved after the Intensified Smallpox Eradication Programme was launched, such a scar was sometimes due to bacterial infection rather than the replication of vaccinia virus, especially in the Indian subcontinent, where the rotary lancet was widely used. In addition, studies in Pakistan by Heiner et al.(1971a) showed that many vaccinated persons living in endemic areas experienced subclinical attacks of smallpox, thus augmenting the protection conferred by vaccination.
The best available information, which almost certainly underestimated the protection afforded by vaccination, came from several sets of data on secondary attack rates among vaccinated and unvaccinated family contacts of smallpox cases in Bangladesh, India and Pakistan reviewed in Chapter 4 (see Table 4.12) . Cases involving substantial numbers of contacts (100 persons) were selected and the rates of protection afforded by vaccination were calculated (Table 11 .25) ; they varied between 90.7% and 97.1%. In these field studies neither the lapse of time since vaccination nor the potency of the vaccine as administered was known, so that it was impossible to determine the reasons for the occurrence of cases of smallpox among vaccinated contacts. The most likely reasons were the use of unsatisfactory vaccines many years earlier or a long period of time since primary vaccination in areas in which revaccination was uncommon.
When confronted with an outbreak of smallpox, it was usual for public health workers to vaccinate or revaccinate all close contacts of index cases (see Chapter 10) . Some of these contacts would not then have been infected with variola virus, but others were probably incubating the disease. The studies just described provide some data on the protection provided by post-exposure vaccination (Table 11 .26). Although the numbers are small and not statistically significant in some individual studies, all the analyses showed a lower rate of occurrence of smallpox in previously unvaccinated family contacts who were vaccinated after exposure. The level of protection was greater when previously vaccinated subjects were included (Heiner et al ., 1971a). Even when post-exposure vaccination did not prevent the occurrence of smallpox, it often mitigated its severity. Rao (1972), for example, recorded that 8.8% of cases of smallpox occurring in subjects who underwent primary vaccination after exposure were of the modified type, compared with 1.1 % among those never vaccinated and 24.7% among persons with scars of primary vaccination, but not vaccinated after exposure.
Mack et al .(1972) succinctly summarize their views on the determinants of infection with smallpox as follows:
“For contacts of any given vaccination status, the rates for protection did not vary with the age, sex or disease severity of the introducer, or with the age or sex of the contacts . Neither did they vary by season, by the closeness of the kinship between contact and introducer (i.e ., siblings, cousins, uncle-nephew, etc.), by their housing relationship (i.e., same or different house within the compound), by caste, by occupation of the household head or by the house construction material.”
Thus vaccination status was overwhelmingly the most important factor in determining the occurrence of infection.
Even though controlled trials were never carried out with smallpox vaccine, it is apparent that it was very effective in preventing smallpox. Clearly, a potent vaccine was required, so that protection was ensured, although such protection declined with the passage of time. Smallpox vaccination had the great advantage over most other types of immunization that it was very easy to determine when a successful take had been achieved, especially after primary vaccination, and the resulting scar provided permanent evidence of it, The general medical opinion, on which the International Health Regulations for smallpox vaccination were based, was that successful vaccination or revaccination within the previous 3 years provided virtually certain protection against samllpox.